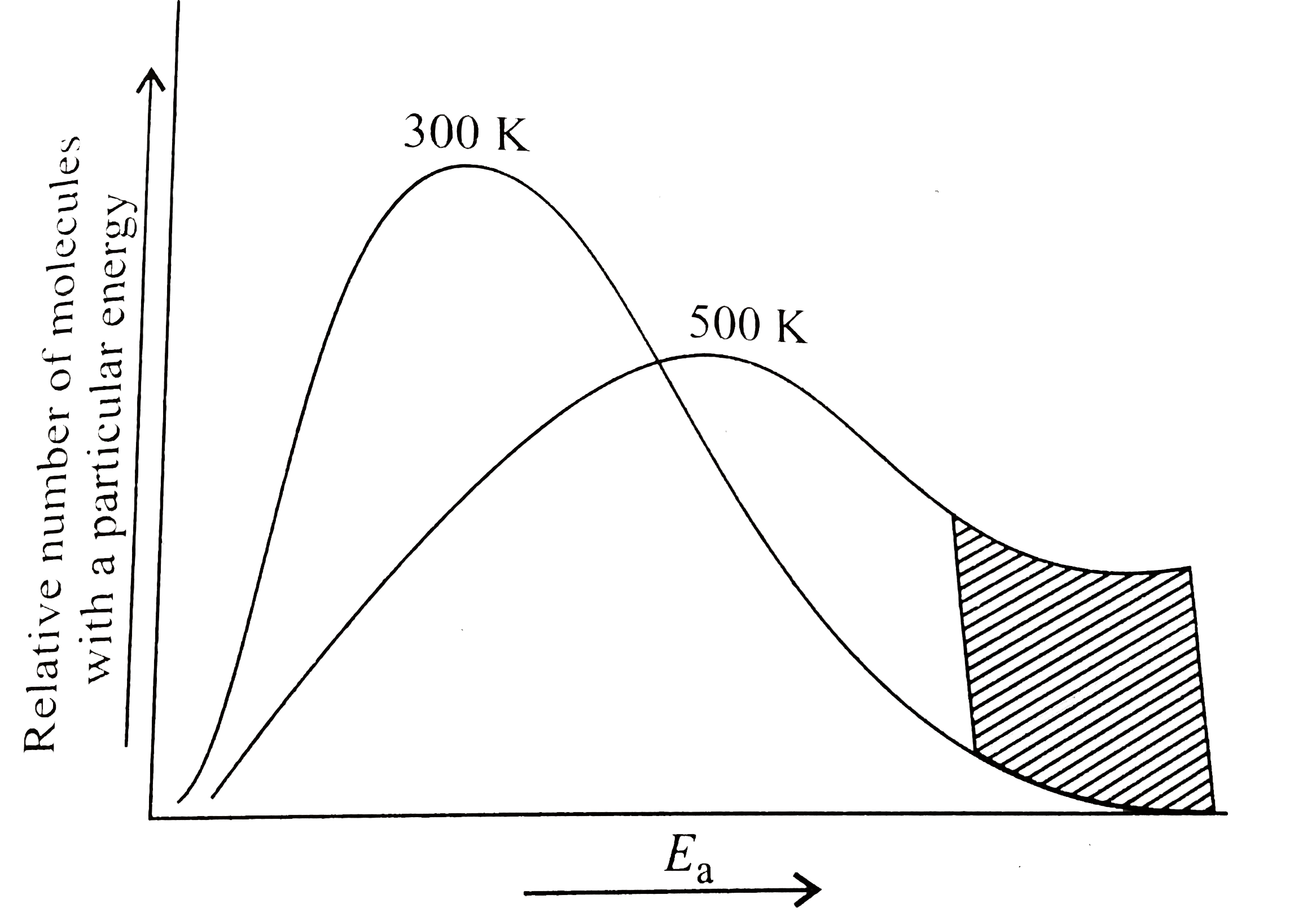
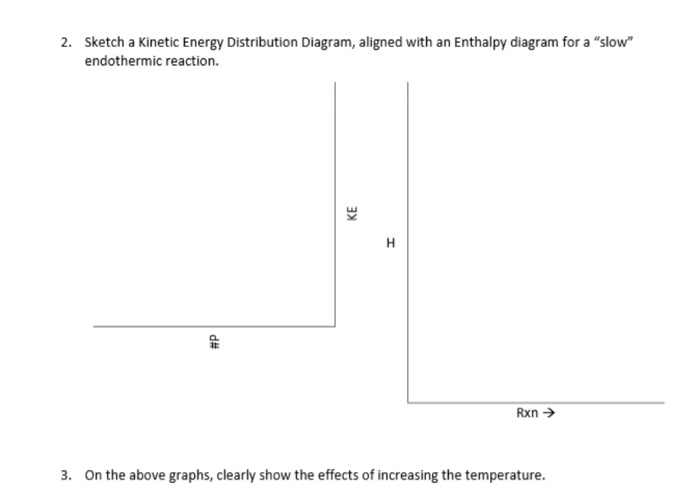
Kinetic Energy Distribution Diagram

Imagine a bustling party where everyone is trying to dance. Some people are doing the cha-cha, others are doing the worm, and a few are just awkwardly shuffling. This is a bit like what’s happening inside everyday objects, from your steaming cup of coffee to the air you breathe.
At the heart of this party is something called kinetic energy. It’s basically the energy of motion. The faster something moves, the more kinetic energy it has. Think of a car zooming down the highway versus a snail inching along.
Now, here’s where it gets interesting. In any group of particles – be it atoms, molecules, or even tiny dust motes – they’re not all moving at the same speed. Some are lazy bums, barely wiggling. Others are hyperactive toddlers on a sugar rush, zipping around like crazy.
Must Read
This difference in speed leads to a difference in their kinetic energy. And this is where our special diagram comes into play. It’s like a snapshot of the party, showing us how many guests (particles) are dancing at different speeds.
The "Many" and the "Few"
The diagram, often called a Maxwell-Boltzmann distribution (don’t let the fancy name scare you!), typically looks like a lopsided hill. On one side, we have the shy wallflowers, the particles moving very slowly. There are usually quite a lot of these guys.
As we move up the hill, we find particles with more and more speed. The number of particles at each speed level starts to decrease. It’s like fewer people are willing to attempt the moonwalk compared to just swaying to the music.
At the very top and then trailing off on the other side, we find the speed demons. These are the particles with the highest kinetic energy, whizzing around at incredible speeds. There are usually only a handful of these extreme movers.
It’s a heartwarming reminder that diversity exists even at the microscopic level! Some particles are content to boogie slowly, while others are born to sprint.
What Makes the Hill Change Shape?
So, what influences the shape of this energy party diagram? The most important factor is temperature. Think of temperature as the party’s DJ.
When the DJ turns up the heat (increases the temperature), everyone starts dancing faster. The whole hill shifts to the right, meaning more particles have higher kinetic energy. The party gets wilder!
Conversely, if the DJ turns down the music (decreases the temperature), things slow down. The hill flattens and shifts to the left. The party becomes more of a gentle sway.
It's fascinating how a simple change in temperature can so dramatically alter the behavior of countless tiny dancers. Your coffee is less energetic on a cool morning than on a scorching summer day, at the molecular level!
Surprising Connections to Your World
This isn’t just abstract science; it has real-world implications that are surprisingly cool. For instance, this diagram helps us understand how reactions happen. For a reaction to occur, particles need to bump into each other with enough energy.
This minimum energy is called the activation energy. On our diagram, it’s like a bouncer at the door, deciding who’s cool enough to enter the reaction dance floor. Only particles with enough kinetic energy can overcome this barrier.
This is why heating things up (increasing temperature) makes reactions go faster. You're essentially giving more particles the energy to get past the bouncer and join the party! Baking a cake, for example, relies heavily on this principle.
Think about your car engine. It needs to reach a high temperature for the fuel to combust efficiently. The kinetic energy distribution is crucial for this ignition.
A Touch of Whimsy
Sometimes, the diagram can look a bit like a little humpbacked creature, happily existing in its energetic world. It’s a visual representation of the chaos and order that coexist within matter. Each little bump and curve tells a story.
It’s also a reminder that even the most seemingly stable things are in constant motion. That solid table you’re leaning on? Its atoms are still vibrating, a constant, albeit slow, dance.
The distribution curve even has a peak, representing the most probable speed. This is the speed at which you'll find the largest group of particles. It's the most popular dance move at the party, if you will.
Understanding this distribution helps scientists predict how gases will behave, how quickly substances will evaporate, and even how quickly your ice cream will melt on a warm day. It’s the secret sauce behind so many everyday phenomena.

The Bottom Line: A Lively Picture
So, the next time you see a diagram showing how particles distribute their kinetic energy, don’t just see lines and curves. See a vibrant, energetic party. See the lazy shufflers, the energetic movers, and the super-speedy outliers.
It’s a snapshot of a microscopic world in constant, dynamic motion. A world where temperature is the DJ, and every particle is doing its own unique dance. It’s a surprisingly simple yet profound way to look at the universe, one energetic shuffle at a time.
It’s a fundamental concept that underlies so much of chemistry and physics, and it’s a beautiful illustration of how even in the smallest scales, there’s a whole lot of movement and variety. It's a lively picture that brings the unseen world to life.
So next time you’re enjoying a hot cup of tea, remember the energetic dance happening within each molecule!