Is Ester Electron Donating Or Withdrawing
Ever wondered what makes molecules behave the way they do? It’s like a secret handshake between atoms, and understanding it can unlock a whole world of chemical wonders. Today, we're diving into a concept that might sound a little intimidating at first, but trust me, it's actually super cool and incredibly useful: whether an ester group is electron-donating or electron-withdrawing.
Think of molecules as tiny teams of atoms. To work together effectively, they need to share or pass around their electrons, which are like the team's energy and communication tools. Sometimes, an atom or a group of atoms in a molecule is really good at holding onto its electrons, kind of like a player who keeps the ball close. These are called electron-withdrawing groups. On the other hand, some groups are happy to share their electrons, pushing them towards other parts of the molecule, like a great passer on the field. These are electron-donating groups.
Now, where does the ester group fit into this picture? The ester group has a bit of a split personality, and that's what makes it so fascinating! Chemically speaking, an ester group looks like this: -COO-. It's made of a carbon atom double-bonded to one oxygen atom and single-bonded to another oxygen atom, which is then attached to the rest of the molecule.
Must Read
The magic, or rather the chemistry, happens because of the electronegativity difference between carbon and oxygen. Electronegativity is basically an atom's "pulling power" for electrons. Oxygen is much more electronegative than carbon. This means the oxygen atoms in the ester group have a strong grip on the electrons.
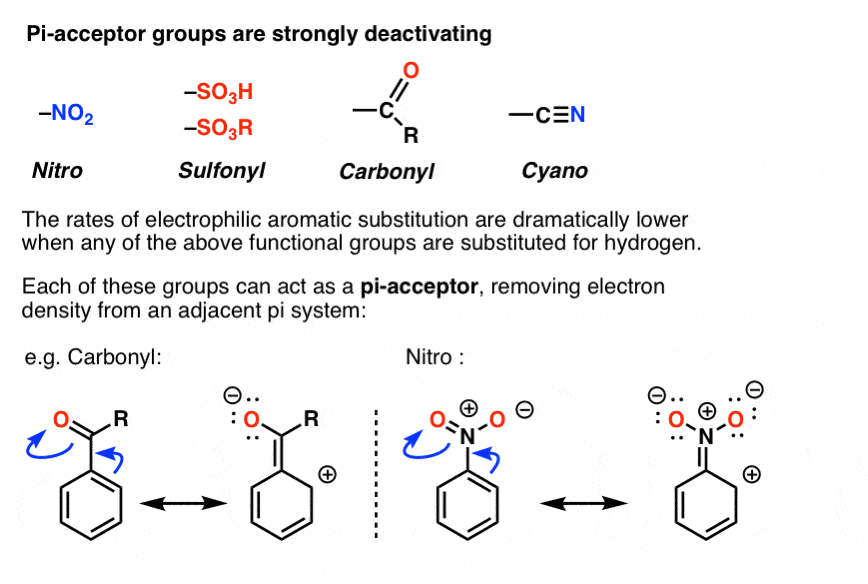
The Electron-Withdrawing Side of Ester
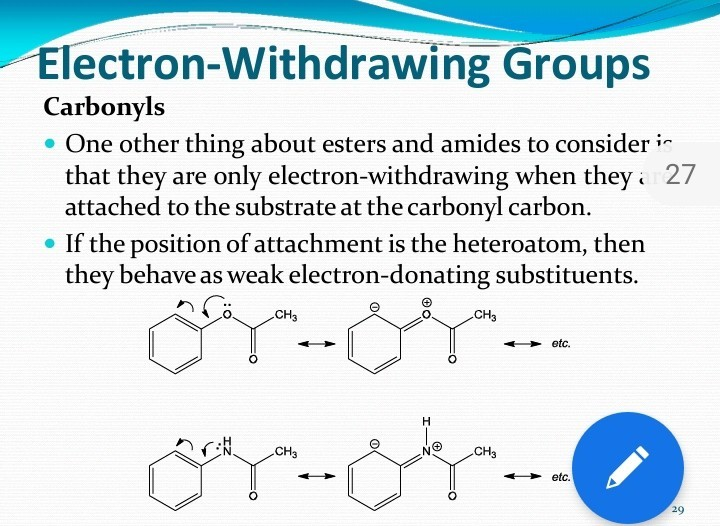
Let's focus on the carbon atom that's double-bonded to an oxygen. Because that oxygen is so electronegative, it pulls electrons strongly towards itself. This pull is so significant that it makes the carbon atom in the ester group slightly electron-deficient, or electron-poor. This means the ester group, particularly through its carbonyl carbon (the C=O part), tends to withdraw electron density from the rest of the molecule it's attached to. Imagine that oxygen saying, "Mine!" and yanking electrons towards itself.

This electron-withdrawing effect is a really important characteristic of esters. It influences how reactive the molecule is, how acidic or basic it might be, and even its physical properties like boiling point and solubility.
The Electron-Donating Side of Ester
But wait, there's more! The story isn't quite as simple as just one action. The oxygen atom that's single-bonded to the carbonyl carbon (the -OR part, where R is the rest of the molecule) has lone pairs of electrons. These lone pairs can actually be donated, or pushed, into the system of the molecule. This is particularly true if the ester is attached to a system that can accept these donated electrons, like a benzene ring (an aromatic ring).
This phenomenon is called resonance. The lone pairs on the single-bonded oxygen can participate in resonance, spreading out electron density and effectively donating electron density into certain parts of the molecule. It's like that single-bonded oxygen saying, "Here, take some electrons, we've got plenty!"
So, Which is it?
This is where it gets really fun! The ester group is often described as being moderately electron-withdrawing, primarily due to the strong pull of the carbonyl oxygen. However, it also exhibits electron-donating character through resonance from the single-bonded oxygen.
Think of it like a tug-of-war. The double-bonded oxygen is pulling hard, trying to withdraw electrons. But the single-bonded oxygen is pushing back with its lone pairs, donating electrons. The overall effect depends on what the ester is attached to and which effect is stronger in a particular situation.

This dual nature is incredibly useful in organic chemistry. It allows chemists to fine-tune the properties of molecules. For example, in pharmaceuticals, understanding whether an ester group is donating or withdrawing can help predict how a drug will interact with biological systems.
It also plays a role in the behavior of flavors and fragrances, many of which are esters. The subtle electronic differences can influence how they interact with our smell receptors. So, the next time you catch a whiff of a fruity perfume or taste a delicious dessert, you can give a little nod to the fascinating electron-donating and withdrawing dance happening within those ester molecules!
Understanding this concept helps us predict reaction pathways, design new molecules with specific properties, and even appreciate the intricate beauty of the chemical world around us. It’s not just about memorizing facts; it’s about understanding the fundamental forces that shape matter. Pretty neat, right?