Is A Displacement Reaction Endothermic Or Exothermic

Ever watched a science experiment that made you go "Whoa!"? That's kind of what happens with displacement reactions. They’re like the drama queens of chemistry, always putting on a show. And the big question is, do these shows get hot or cold? In other words, are they endothermic or exothermic? Let's dive in and find out what makes these reactions so captivating!
Think of a displacement reaction like a dance-off. You've got a group of dancers (atoms or ions) and one dancer wants to cut in on a pair. So, one dancer steps in and takes the place of another. Pretty straightforward, right? But here's where the magic happens: this dance can either heat up the dance floor or cool it down! It's this energy change that makes them so fascinating.
Now, let's talk about the two main ways these reactions can go. We have the exothermic reactions, and then we have the endothermic ones. It’s like choosing between a fiery salsa and a chilling waltz.
Must Read
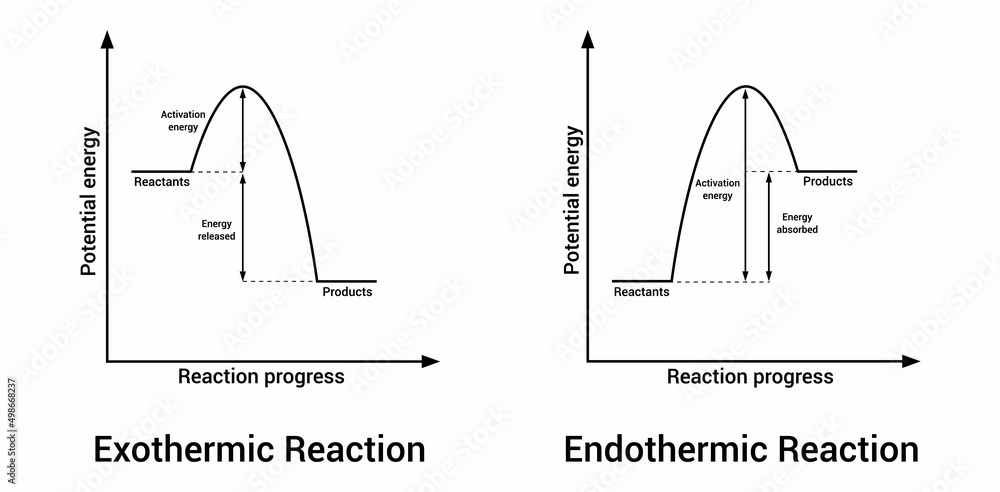
An exothermic displacement reaction is the energetic one. It’s the type that gives off heat. Imagine you’re at a party, and a really exciting song comes on. Everyone starts dancing, the energy level rises, and the room gets warmer. That’s an exothermic reaction in a nutshell! It’s releasing energy, usually in the form of heat, and you can often feel it. It’s like a chemical high-five, giving off its extra energy to the surroundings. This is what makes them so visually striking and sometimes even a bit dramatic. You might see steam rising, a flame flicker, or just feel the beaker get surprisingly warm to the touch. It's a clear sign that something energetic has just happened!
It's like a chemical party, where the energy is bursting out!
exothermic and endothermic reactions ppt | PPT
On the other hand, we have the endothermic displacement reaction. These are the ones that suck up energy from their surroundings. Think of it like stepping into a cool, shaded spot on a really hot day. You feel that immediate relief as the heat is absorbed. An endothermic reaction does the same thing. It needs energy to happen, so it pulls it from wherever it can – usually the air around it, or the liquid it’s in. This means that if you're performing an endothermic displacement reaction, the container might feel cold! It's like the reaction is taking a deep, cool breath, and in doing so, it cools everything down. This can be just as intriguing as the heat-releasing ones, albeit in a different way. The lack of heat can be a subtle but powerful indicator of what’s going on at the molecular level.
It's like a chemical chill, taking energy away.
Ncert class 10 - science - chapter 1 - chemical reactions and equations
So, which is it? Are displacement reactions always hot, always cold, or a mix? Well, it’s a bit of a mix! The outcome, whether it’s endothermic or exothermic, depends entirely on the specific players involved in the reaction. It’s like picking dancers for a duet – some pairings are naturally going to generate a lot of heat, while others might require a bit of warming up. The strength of the bonds being broken and the bonds being formed are the real deciding factors here.
For instance, if the new bonds that form in the reaction are stronger and more stable than the bonds that were broken, then energy is released. This is the recipe for an exothermic reaction. The system is happy to let go of that extra energy. It’s like finding a more comfortable position; you don’t need any extra effort, you just settle in and release any tension.

Conversely, if the bonds that need to be broken are really strong, and the new bonds formed aren't as robust, then the reaction needs a little help. It needs to absorb energy from the outside to get those stubborn bonds to break. This is where endothermic reactions come into play. The system is essentially saying, "I need a boost to make this happen!"
The beauty of displacement reactions lies in this variability. You can have a reaction where a metal like zinc jumps into a solution of copper sulfate. This one is typically exothermic! You can often see the copper metal forming, and the solution might warm up a bit. It's a classic, energetic exchange.

But then you might have other scenarios where the energy demands are different. The specific metals and the compounds they are displacing all play a role. It’s like a chemical game of rock-paper-scissors, where the outcome is determined by the inherent properties of each element. Some elements are just naturally more eager to give up their electrons or bond with others in a way that releases energy, while others are more reluctant and require energy input.
This is why studying displacement reactions is so engaging. You never quite know if you're going to witness a fiery display or a cooling phenomenon. It keeps you on your toes, wondering what the next reaction will bring. It’s this element of surprise, combined with the fundamental chemical principles at play, that makes them so captivating for chemists and anyone curious about the world around them.
So, next time you hear about a displacement reaction, remember it’s not just a simple swap. It's a chemical event that can either heat things up or cool things down, all depending on the unique personalities of the atoms and ions involved. It’s this dynamic interplay of energy that makes the world of chemistry so wonderfully entertaining and endlessly surprising. It's a little bit of everyday magic, happening right before your eyes!