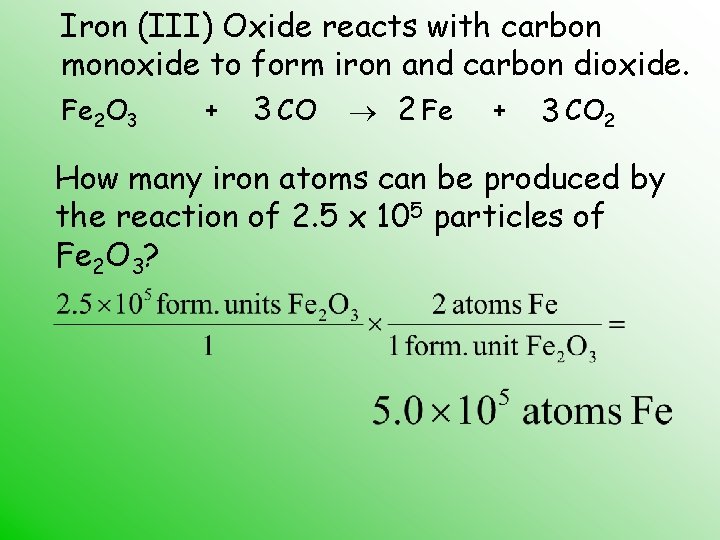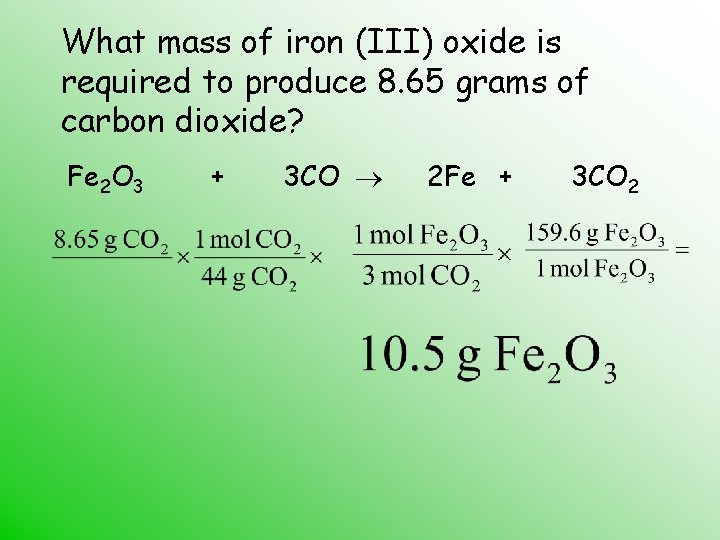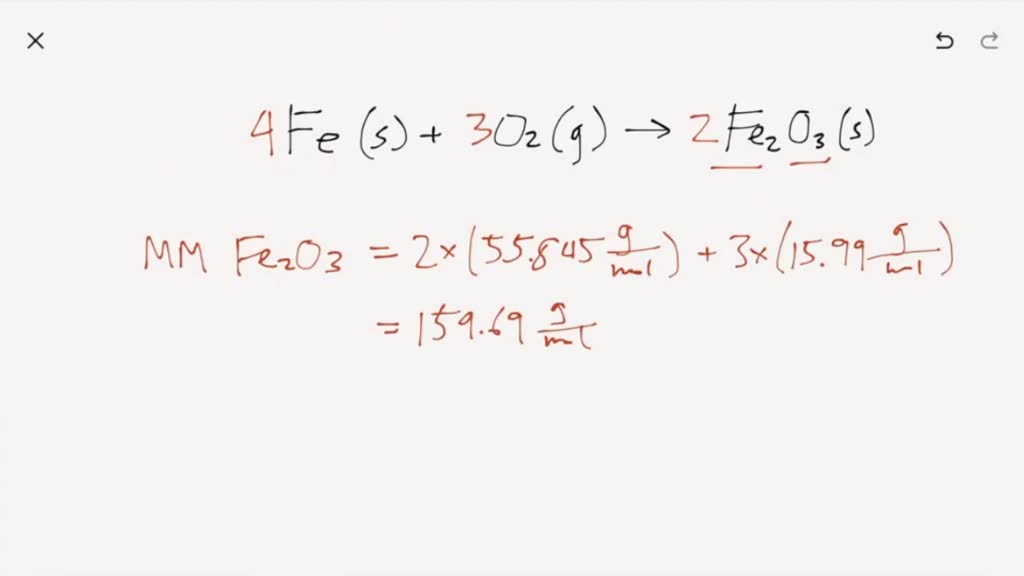Iron Iii Oxide And Carbon Balanced Equation
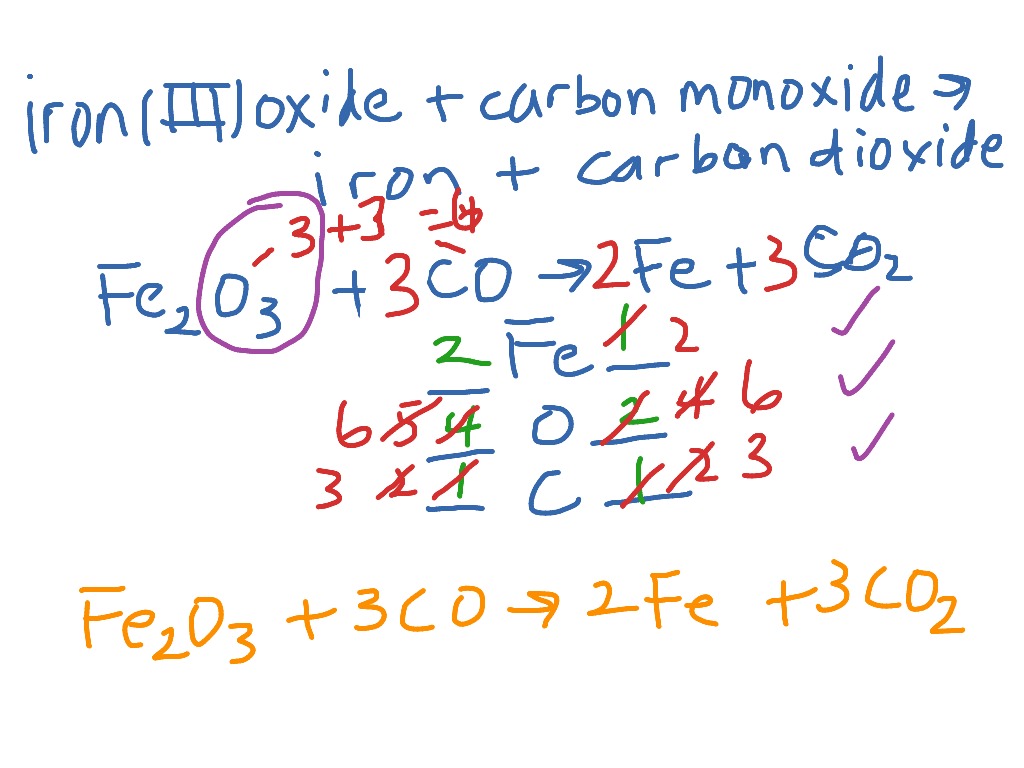
Ever wondered what happens when rust meets charcoal? Or perhaps you've seen a vibrant red pigment and thought, "What's that made of?" Well, get ready for a little bit of chemical magic because we're diving into the fascinating world of Iron(III) Oxide and Carbon! It might sound like something out of a mad scientist's lab, but this particular reaction is surprisingly useful and incredibly cool. Think of it as a tiny chemical dance where ingredients transform into something new and exciting.
So, what's the big deal with Iron(III) Oxide and Carbon? At its heart, this is all about making things happen in the world of chemistry. We're talking about a chemical reaction where a common substance, Iron(III) Oxide (which is basically rust, Fe₂O₃), gets a makeover with the help of Carbon (think the stuff in charcoal, C). This isn't just for show; it has some really neat applications. One of the most significant benefits is its role in metallurgy, the science of metals. Specifically, this reaction is a key part of how we get pure iron out of its ore. Imagine all those strong metal structures, the cars we drive, the tools we use – they all start with processes like this!
But it's not just about making bulk metal. The balanced equation for the reaction between Iron(III) Oxide and Carbon is a prime example of a reduction-oxidation reaction, often called a redox reaction. Don't let the fancy terms scare you! In simple terms, it's like a chemical swap meet. Carbon is a fantastic "reducing agent," meaning it's really good at taking away oxygen from other substances. In this case, it's a bit of a chemical bully, snatching oxygen atoms away from Iron(III) Oxide. This leaves behind the pure, elemental iron that we find so useful.
Must Read
The "balanced equation" part is where things get precise and elegant. Think of it like following a recipe. You need the right amounts of each ingredient to get the perfect outcome. A chemical equation shows us which substances are involved and how they rearrange. A balanced equation ensures that we have the same number of atoms of each element on both sides of the reaction. This is a fundamental law of chemistry: atoms aren't created or destroyed in a chemical reaction; they just find new partners. So, for Iron(III) Oxide and Carbon, the balanced equation tells us exactly how much Carbon is needed to "reduce" a certain amount of Iron(III) Oxide into iron and carbon dioxide.

Let's peek at this important equation. You'll often see it written as:
Fe₂O₃(s) + 3C(s) → 2Fe(s) + 3CO(g)
Whoa, what does all that mean? Let's break it down in a friendly way.
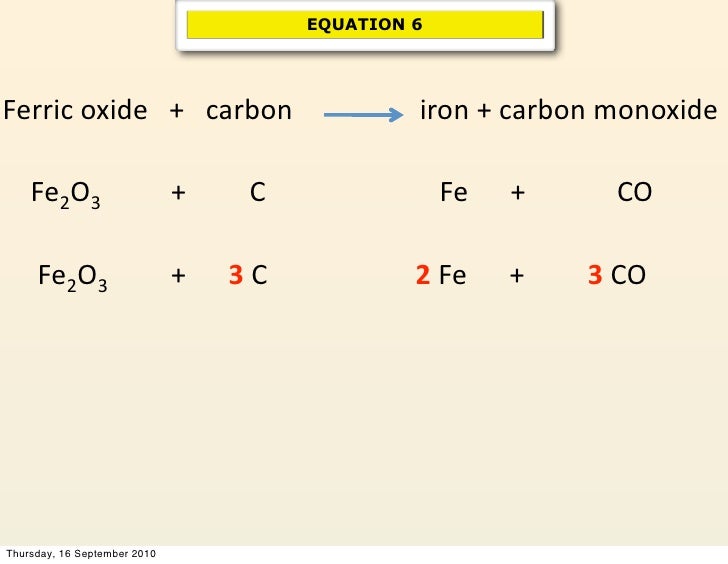
- Fe₂O₃(s): This is our Iron(III) Oxide, the rust. The '(s)' just means it's a solid.
- 3C(s): This is Carbon, our reducing agent, also a solid. Notice the '3' in front? That's the balancing act! It tells us we need three carbon atoms for every one molecule of Iron(III) Oxide.
- →: This arrow means "reacts to produce" or "yields."
- 2Fe(s): This is the prize! Pure, elemental iron, again as a solid. And see the '2'? It means we get two atoms of iron out of this reaction.
- 3CO(g): This is carbon monoxide, a gas. The '3' here shows that the three carbon atoms have combined with three oxygen atoms from the iron(III) oxide, forming three molecules of carbon monoxide.
The beauty of this balanced equation is that it's not just theoretical. It's the blueprint for industrial processes. In blast furnaces, where we make most of our iron, this very reaction happens on a massive scale. Huge amounts of iron ore (which is rich in Iron(III) Oxide) are heated with coke (a form of carbon). The carbon performs its oxygen-snatching duty, and out comes molten iron. It's a testament to how understanding basic chemical principles can lead to incredible feats of engineering and production.
But wait, there's more! While the primary use is in iron production, understanding this reaction helps us grasp other chemical transformations. It illustrates how different elements have different affinities for oxygen. It also shows how simple, readily available materials like rust and charcoal can be used to create something as valuable and fundamental as iron. It’s a perfect example of turning something seemingly mundane into something incredibly useful. So, the next time you see a metal object, or even just a rusty bit of metal, remember the elegant dance of atoms that might have been involved in its creation – the powerful, yet simple, reaction of Iron(III) Oxide and Carbon!

+oxide+reacts+with+carbon+monoxide+to+form+iron+and+carbon+dioxide..jpg)