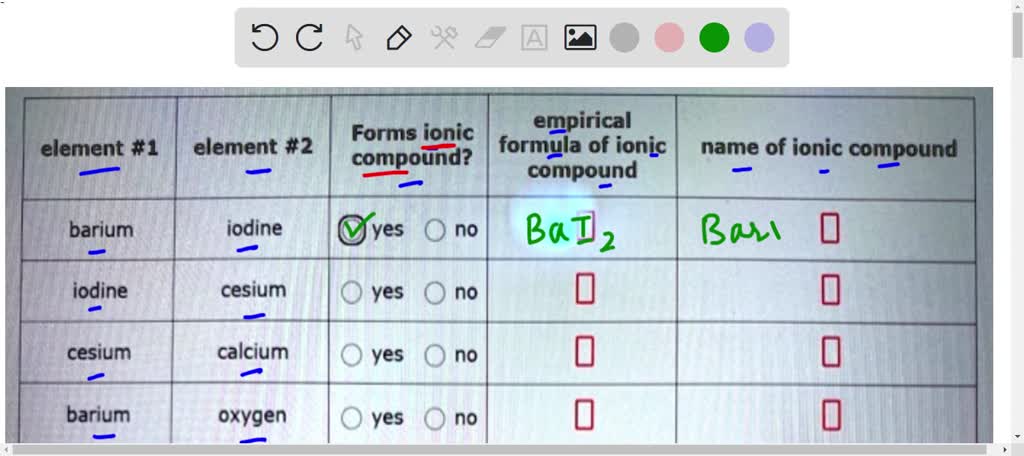
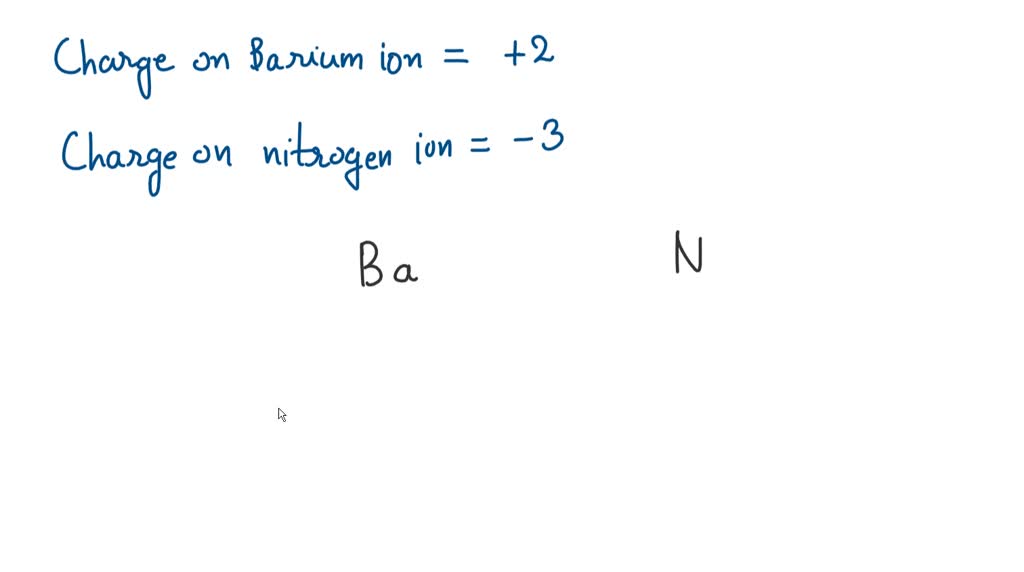Ionic Compound Made Of Barium And Nitrogen

Ever wondered what happens when elements get together and form new compounds? It's like a cosmic matchmaking service where atoms find their perfect partners to create something entirely new and often surprisingly useful! Today, we're diving into the exciting world of barium and nitrogen and the fascinating ionic compound they can create: barium nitride. Now, you might think "chemistry talk," but trust me, this is more like discovering a secret ingredient that pops up in some pretty cool places. It's a compound that might not be a household name like salt or sugar, but its unique properties make it a star player in specific industries.
Unlocking the Potential of Barium Nitride
So, what's the big deal about barium nitride? Well, it's all about its incredible ability to withstand extreme conditions and its unique electronic properties. Imagine a material that can handle a scorching hot environment or is a key player in creating vibrant displays. That's where this ionic compound shines! Its formation is a classic example of how elements with very different personalities – a reactive metal like barium and a non-metal like nitrogen – can combine to form a stable and functional substance.
When barium, which loves to give away electrons, meets nitrogen, which is eager to grab them, they form a strong ionic bond. This means barium atoms become positively charged (cations), and nitrogen atoms become negatively charged (anions), and these opposite charges attract each other like tiny magnets, forming a crystal lattice. This lattice structure is incredibly robust, giving barium nitride its impressive durability and high melting point.
Must Read
The purpose of understanding and utilizing barium nitride lies in harnessing these specific characteristics. One of its most significant benefits is its role in the creation of advanced materials. For instance, in the realm of electronics, barium nitride is being explored for its potential in developing new types of semiconductors and superconductors. These are the unsung heroes behind everything from faster computers to more efficient energy transmission. Think about it: a compound that helps make our technology smarter and our energy usage greener!
Beyond electronics, barium nitride has also found its way into specialized applications where resistance to heat and chemicals is paramount. It can be used in certain types of ceramics, which are materials known for their toughness and ability to endure high temperatures. This makes it valuable in industries that deal with extreme environments, such as aerospace or high-performance manufacturing. It's like having a material that's virtually unbreakable when things get tough.

The unique way barium and nitrogen interact to form this compound also opens doors for research into new catalytic processes. Catalysts are substances that speed up chemical reactions without being consumed themselves. Finding efficient catalysts is crucial for developing more sustainable and cost-effective ways to produce a vast array of chemicals, from pharmaceuticals to plastics. Barium nitride, with its specific electronic structure, shows promise in this area, hinting at future innovations that could impact our daily lives in ways we haven't even imagined yet.
What makes it particularly engaging is the sheer diversity of its potential applications. From the microscopic world of semiconductors to the robust demands of industrial manufacturing, barium nitride demonstrates how fundamental chemical principles can lead to groundbreaking technological advancements. It’s a testament to the power of combining elements, each with its own unique properties, to unlock entirely new functionalities.

Furthermore, studying compounds like barium nitride helps us refine our understanding of chemical bonding and material science. Every new compound we discover and characterize adds another piece to the intricate puzzle of how matter behaves. This knowledge is foundational for future scientific exploration and drives innovation across countless fields. It’s like learning a new language – the more you know, the more you can express and create.
So, the next time you hear about barium and nitrogen, remember that they’re not just random elements on a chart. Together, they can form a powerful ionic compound with the potential to revolutionize technology and improve our world. It's a reminder that even in the seemingly abstract world of chemistry, there are tangible benefits and exciting possibilities waiting to be discovered!