Ionic And Covalent Bonds Virtual Lab Answers
Ever found yourself staring at a glass of water and wondering, "What's really going on in there?" Or maybe you've tinkered with a battery, curious about the invisible forces at play. These everyday marvels are all thanks to the fundamental building blocks of matter: chemical bonds. And for anyone who loves a good puzzle or a bit of scientific exploration, diving into the world of ionic and covalent bonds can be surprisingly fun and incredibly illuminating. It's like unlocking a secret language that explains how everything around us is held together.
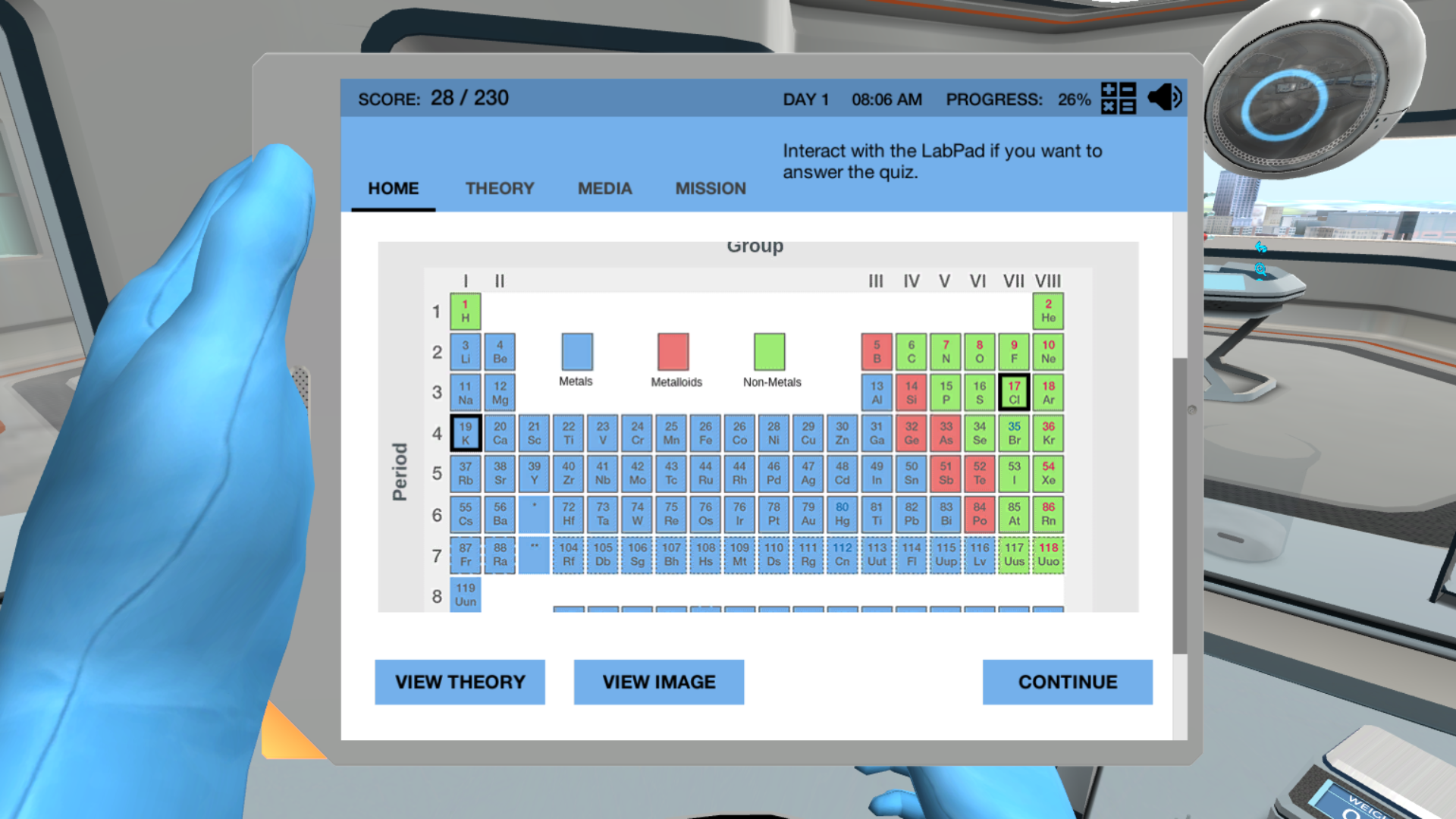
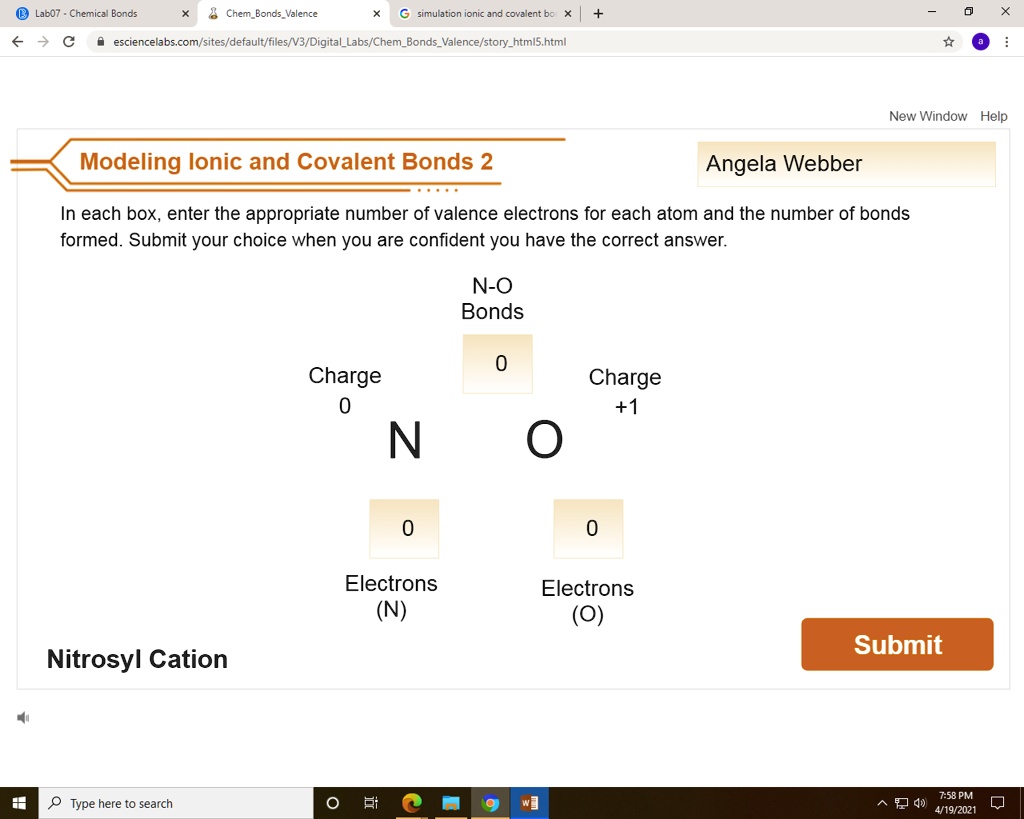
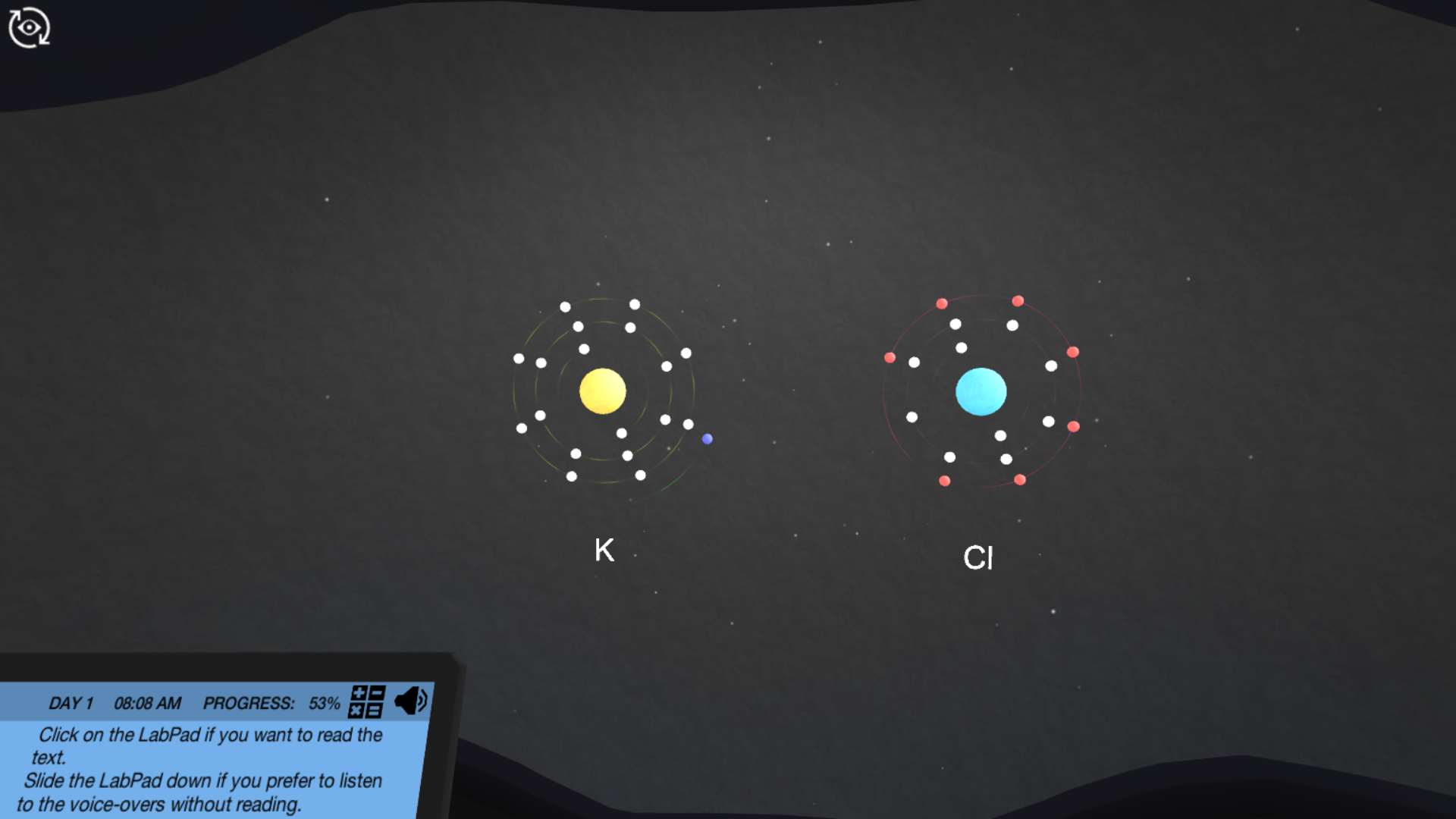
Now, you might have stumbled upon something called "Ionic And Covalent Bonds Virtual Lab Answers." Don't let the "answers" part make you think this is just about memorizing facts. Think of it more like having a digital sandbox to play in. The purpose of these virtual labs is to give you a hands-on, risk-free way to experiment with different atoms and see how they interact. You can mix and match, change conditions, and observe the results in real-time, all without needing a bulky lab coat or worrying about making a mess!
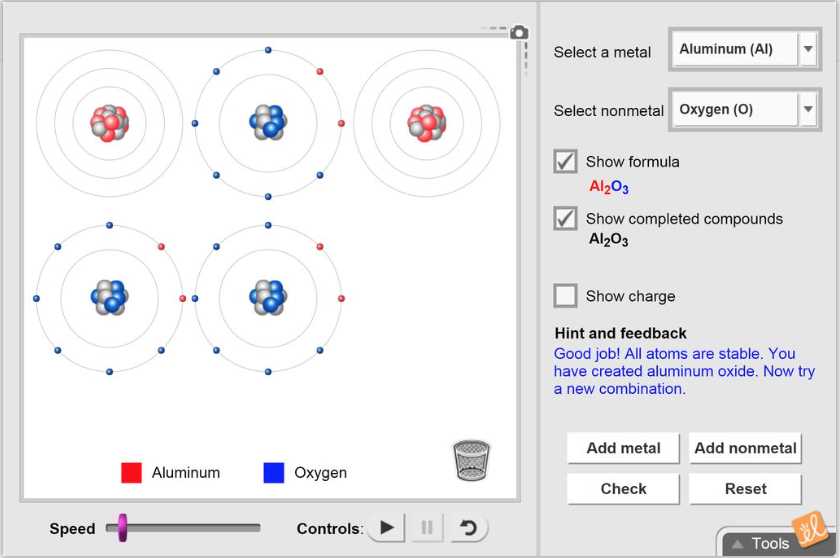
The benefits are pretty fantastic. For students, it's a game-changer. Instead of just reading about abstract concepts like electronegativity or electron sharing, you can visualize and manipulate them. It makes learning about how substances form – like table salt (ionic) or water (covalent) – much more intuitive and memorable. Beyond the classroom, understanding these bonds is key to appreciating so many things in our daily lives. Think about the plastic in your phone, the metals in your car, or even the medicines you might take. All of these are products of intricate atomic arrangements driven by ionic and covalent bonding.
Must Read
So, how can you get in on this? If you're a student, your teacher might already be using virtual labs in your chemistry class. Ask them about it! If you're just curious, a quick online search for "ionic covalent bonding virtual lab" will reveal a treasure trove of free resources. Many educational websites offer interactive simulations that let you build molecules, predict bond types, and even explore the properties of the resulting compounds. It’s a wonderfully accessible way to satisfy your scientific curiosity.
Even without a formal lab, you can start exploring. Grab some everyday items and ponder their composition. Think about why a piece of metal conducts electricity (free-moving electrons in metallic bonds, related to covalent) while a piece of plastic doesn't. Consider the difference between dissolving sugar (covalent) in water versus dissolving salt (ionic). These are all gentle nudges towards understanding the powerful forces that shape our world. So, go ahead, experiment virtually, and discover the fascinating dance of atoms!