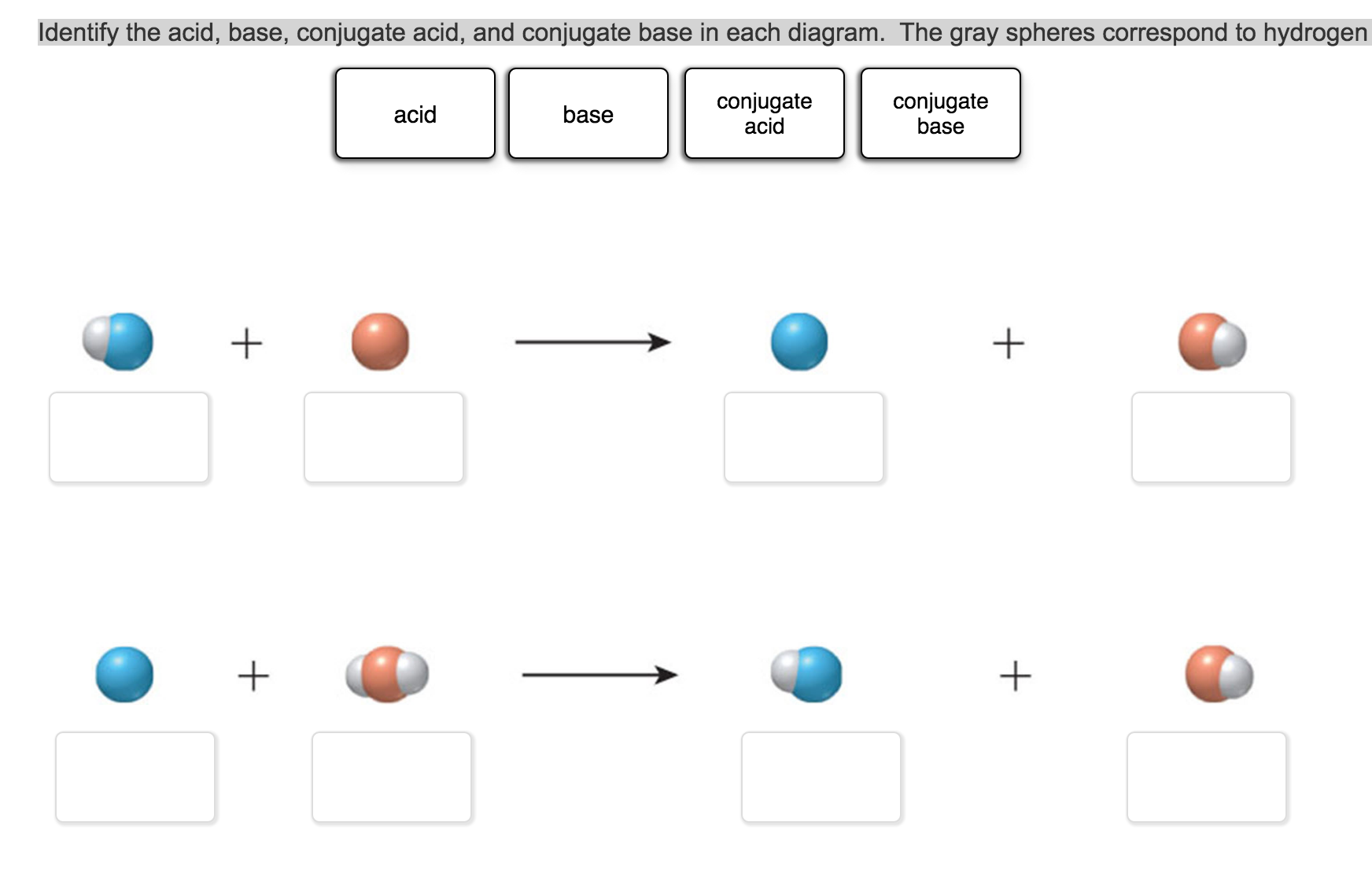
Identify The Conjugate Base For Each Acid.

So, picture this: I’m in my first year of chemistry, brimming with enthusiasm (read: absolute terror). My professor, a wonderfully eccentric soul with perpetually ink-stained fingers, declared one day, "Today, we’re talking about acids and bases. And not just the ones that make your stomach hurt, but the ones that give and take." My brain, already a little foggy from trying to decipher molecular structures, did a collective shrug. Give and take? Like, sharing is caring, but with protons?
He went on to explain this whole Brønsted-Lowry theory thing, which, honestly, sounded like it belonged in a medieval alchemist’s grimoire. But then, it clicked. It’s all about a little chemical handshake, a transfer of a proton. And the star of our show today is identifying the conjugate base for each acid. Don't worry, it's not as scary as it sounds. Think of it as figuring out who's left holding the bag after a proton has been politely (or not so politely) passed along. 😉
You see, in the world of chemistry, acids are like generous givers. They have a proton (that’s just a hydrogen atom that’s lost its electron, you know, the positively charged H+ thingy) that they’re eager to donate. Bases, on the other hand, are the eager receivers. They’re waiting, practically with open arms (or electron pairs, if we want to get technical), to snatch up that proton. And when an acid gives away its proton, what’s left behind? That’s our conjugate base!
Must Read
The Proton Party: Acids Giving and Bases Taking
Let’s break it down. When an acid (let’s call it HA, where H is the proton and A is the rest of the molecule) bumps into a base (let’s represent it as B), a little chemical drama unfolds. The acid HA, being the proton donor it is, will release its H+. So, HA becomes A-. This A- is the conjugate base of the original acid HA.
Meanwhile, the base B, being the proton acceptor, grabs that H+. So, B becomes BH+. This BH+ is the conjugate acid of the original base B. It’s like a whole acid-base couple dancing! We’ve got the acid (HA) and its partner, the conjugate base (A-), and the base (B) and its partner, the conjugate acid (BH+).
The key thing to remember is that the conjugate base is formed after the acid has donated its proton. It’s the leftover bit. Easy peasy, right? Well, usually. Sometimes the formulas look a bit intimidating, but the principle is always the same: remove one H+ from the acid to get its conjugate base.
Spotting the Conjugate Base: It's All About the Missing Proton!
So, how do we actually identify this elusive conjugate base? It’s like a game of "spot the difference," but with hydrogen atoms. For any given acid, you just need to look at its formula and imagine it saying "Adios, H+!" and then see what’s left.
Let’s take a classic example: hydrochloric acid (HCl). This is a strong acid, meaning it’s very good at donating protons. When HCl acts as an acid, it gives away its proton (H+). What’s left? Well, it’s the chlorine atom with a negative charge, Cl-. So, Cl- is the conjugate base of HCl. Simple as that!
Think of it like this: HCl is the proud parent, always ready to let go of its little proton child. Once the child is gone, what remains is the parent, now a bit lonelier and with a slight negative vibe (hence the minus charge!).

The Water Waltz: H₂O as Both Acid and Base
Now, things can get a little more interesting when we involve molecules like water (H₂O). Water is a superstar in chemistry because it can act as both an acid and a base. It’s the ultimate diplomatic molecule!
When water acts as an acid, it donates a proton. So, H₂O loses an H+ and becomes OH- (hydroxide ion). In this case, OH- is the conjugate base of water (when acting as an acid). See? We just removed an H+ from H₂O.
But wait, there’s more! When water acts as a base, it accepts a proton. So, H₂O gains an H+ and becomes H₃O+ (hydronium ion). In this scenario, H₃O+ is the conjugate acid of water (when acting as a base). This is where the "giving and taking" really shines, showing water’s dual personality!
It's pretty neat, isn't it? This ability of water to be both an acid and a base is fundamental to why aqueous solutions behave the way they do. It’s like the social butterfly of the molecular world!
More Acidic Adventures: Let's Try Some Others!
Let’s get our hands dirty with a few more examples. The more you practice, the more intuitive this becomes. You’ll be spotting conjugate bases faster than you can say "proton transfer."
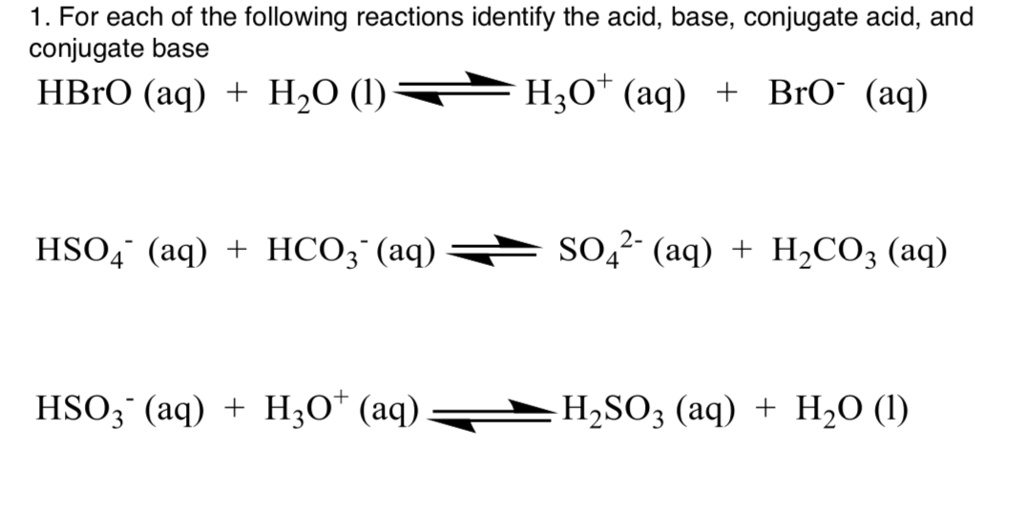
Sulfuric Acid (H₂SO₄)
Sulfuric acid is another familiar acid. When H₂SO₄ acts as an acid and donates a proton, what’s left? We take away one H+, and we’re left with HSO₄- (the hydrogen sulfate ion or bisulfate ion). So, HSO₄- is the conjugate base of H₂SO₄.

Now, here’s a little twist for the curious minds: HSO₄- can also act as an acid! If it donates another proton, it becomes SO₄²- (the sulfate ion). So, in a different reaction, SO₄²- is the conjugate base of HSO₄-. It’s like a proton cascade!
Nitric Acid (HNO₃)
Nitric acid is pretty straightforward. When HNO₃ loses a proton, it forms NO₃- (the nitrate ion). Therefore, NO₃- is the conjugate base of HNO₃.
Notice a pattern here? Many common acids end up with a negative charge after donating a proton. This makes sense, right? They started neutral (or positive in some complex cases), and they gave away a positive particle.
Acetic Acid (CH₃COOH)
This one looks a little more complex, but the rule is the same. Acetic acid, the acid in vinegar, has a structure where one hydrogen atom is attached to the oxygen atom in the -COOH group. That's the proton that’s typically donated. So, when CH₃COOH donates a proton, it becomes CH₃COO- (the acetate ion). Hence, CH₃COO- is the conjugate base of CH₃COOH.
The other hydrogens in the CH₃ group are generally not acidic; they’re tucked away quite happily. So, focus on the acidic proton!
Phosphoric Acid (H₃PO₄)
Phosphoric acid has three potential protons to donate. If it donates one, it becomes H₂PO₄- (the dihydrogen phosphate ion). So, H₂PO₄- is the conjugate base of H₃PO₄.
If H₂PO₄- donates another proton, it becomes HPO₄²- (the hydrogen phosphate ion). Thus, HPO₄²- is the conjugate base of H₂PO₄-.
And if HPO₄²- donates its final proton, it becomes PO₄³- (the phosphate ion). So, PO₄³- is the conjugate base of HPO₄²-. It’s like a series of proton farewells!
Why Does This Matter? The Chemical Ecosystem
You might be thinking, "Okay, I can take off a proton. Big deal. What’s the point?" Ah, my friend, this is where the magic happens! Understanding conjugate bases is crucial for understanding chemical reactions. It helps us predict how acids and bases will interact.
The strength of an acid is directly related to the strength of its conjugate base. A strong acid (like HCl) has a very weak conjugate base (like Cl-). This means Cl- is not good at grabbing protons back. It’s happy to stay as Cl-.
Conversely, a weak acid (like acetic acid, CH₃COOH) has a relatively strong conjugate base (like CH₃COO-). The acetate ion is much more likely to pick up a proton than the chloride ion is.
This relationship is super important when we talk about buffers – those wonderful chemical systems that resist changes in pH. Buffers are usually made of a weak acid and its conjugate base (or a weak base and its conjugate acid).

For example, a buffer system in our blood often involves carbonic acid (H₂CO₃) and its conjugate base, bicarbonate (HCO₃-). When we produce too much acid, the bicarbonate ion picks up the excess protons, preventing our blood from becoming too acidic. When we produce too little acid, the carbonic acid can donate a proton to help raise the acidity. It’s a delicate balancing act, and it relies entirely on the interplay between acids and their conjugate bases!
Ammonia (NH₃) – A Base with a Conjugate Acid
So far, we've focused on acids and their conjugate bases. But let’s not forget about bases! When a base accepts a proton, it forms its conjugate acid. Remember our earlier chat? This is the flip side of the coin.
Let's take ammonia (NH₃). Ammonia is a classic base. When it accepts a proton (say, from water), it forms NH₄+ (the ammonium ion). So, NH₄+ is the conjugate acid of ammonia (NH₃). See? We added an H+ to NH₃.
The rule is simple: add one H+ to the base to get its conjugate acid. Just like removing a proton gives you the conjugate base, adding one gives you the conjugate acid.
The Takeaway: It’s All About the Proton!
So, the next time you see an acid, just imagine it saying goodbye to a proton. Whatever’s left is its conjugate base. It’s like peeling off a layer. The acid is the whole onion, and the conjugate base is the onion without its outermost skin.
Remember these key points:
- An acid donates a proton (H+).
- A base accepts a proton (H+).
- The conjugate base is formed when an acid loses a proton. You get it by removing one H+ from the acid's formula.
- The conjugate acid is formed when a base gains a proton. You get it by adding one H+ to the base's formula.
- Water can act as both an acid and a base.
- The strength of an acid is inversely related to the strength of its conjugate base.
Don't get bogged down by complex formulas. Focus on the proton. That little positively charged H+ is the star of this entire acid-base show. Once you master identifying the conjugate base (and acid!), you've unlocked a whole new level of understanding in chemistry. It’s a fundamental concept that pops up everywhere, from biology to environmental science. So, embrace the proton transfer, and happy conjugate base hunting!