How To Calculate The Rf Value Chromatography
Ever looked at a colorful science experiment, maybe in a movie or a particularly engaging episode of your favorite educational show, and thought, "Wow, that looks cool! But how on earth do they do that?" Well, get ready to pull back the curtain because we're diving into the magical world of chromatography and, more specifically, how to calculate something called the Rf value. Don't let the fancy name scare you; it's actually as fun and accessible as making a surprisingly vibrant tie-dye shirt!
Think of chromatography as a high-stakes, microscopic race. You've got a mixture of different "runners" (your chemical compounds) and they're all trying to get from point A to point B on a special "track" (the stationary phase, like a piece of paper or a thin layer of silica). What makes this race interesting is that each runner has a slightly different personality – some are a bit lazy and don't go very far, while others are super energetic and zoom ahead. The Rf value is basically your way of scoring this race, giving each runner a definitive number that tells you just how far they got compared to the furthest anyone could possibly go.
So, why should you care about calculating an Rf value? Because it’s not just for lab coats and microscopes! Understanding this simple calculation can unlock a whole new appreciation for the colorful world around you. Think about the vibrant dyes in your clothes, the natural pigments in plants, or even the way different flavors are extracted for your favorite foods. Chromatography, and the Rf value that quantifies its results, is the unsung hero behind so much of the sensory joy in our lives.
Must Read
Let’s get down to the nitty-gritty, shall we? Calculating the Rf value is remarkably straightforward. You don't need a PhD, just a ruler, a bit of patience, and a successful chromatography experiment. First things first, you need to have run your chromatography. This usually involves placing a tiny spot of your mixture at the bottom of your "track" (often called the stationary phase) and then letting a "mobile phase" (a solvent or mixture of solvents) travel up the track. As the mobile phase moves, it carries your mixture's components with it, separating them based on their differing affinities for the stationary and mobile phases.
Once the solvent has traveled as far as it's going to go (or you decide to stop the race), you'll see distinct spots or bands representing your separated compounds. Now for the measuring part! You'll need to grab that trusty ruler.
The Big Calculation: It's Easier Than You Think!
Here's the magic formula, drumroll please:
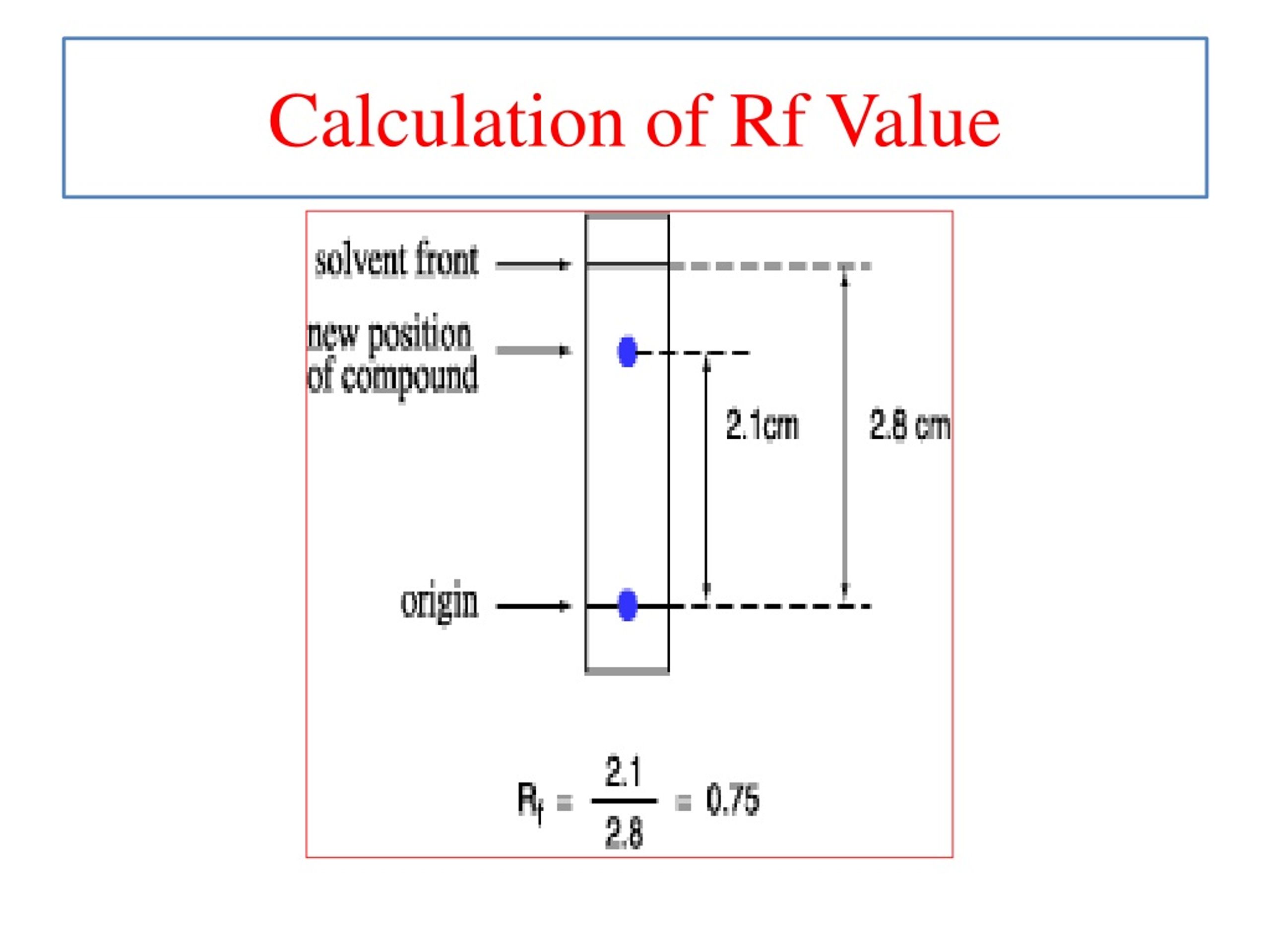
Rf value = (Distance traveled by the spot) / (Distance traveled by the solvent front)
See? Not so scary, right? Let's break down what each part means.

Measuring the Spot
First, you need to measure the distance that your particular spot (the one you're interested in) traveled. You'll measure from the original spot line (where you first put your mixture) all the way up to the center of that specific spot. This is your "distance traveled by the spot." It’s like measuring how far your favorite runner made it from the starting line. Remember to be as precise as you can – a millimetre here or there can make a difference, especially if you're trying to identify a specific compound!
Measuring the Solvent Front
Next, you need to measure how far the solvent front traveled. The solvent front is simply the furthest point the mobile phase reached on your track. Imagine it as the finish line that the fastest runner could have reached. You measure this distance from the original spot line all the way up to that solvent front. This is your "distance traveled by the solvent front." Think of it as the maximum possible distance any runner could have covered in this particular race.
Once you have both of those measurements, you simply divide the distance your spot traveled by the distance the solvent front traveled. And voilà! You have your Rf value.

Why is This Little Number So Important?
The Rf value is like a unique fingerprint for each compound under specific chromatographic conditions. Different compounds will have different Rf values because they interact differently with the stationary and mobile phases. Some compounds might be really attracted to the stationary phase and barely move (low Rf value), while others might be perfectly happy hitching a ride with the mobile phase and travel almost all the way to the solvent front (high Rf value, approaching 1).
This is where the fun really begins. Imagine you're trying to figure out what makes a specific flower's scent so unique. By performing chromatography on its extracted oils and calculating the Rf values of the separated components, you can compare them to known Rf values of common fragrant compounds. It's like being a detective, piecing together clues to identify the mystery ingredients!
Or consider the vibrant reds and oranges in autumn leaves. Chromatography can help us understand the different pigments present. Each pigment will have its own characteristic Rf value, allowing scientists (and you, with a little practice!) to identify chlorophyll, carotenes, and other natural colorants. Isn't that just marvelous?

Important Note: The Rf value is only consistent if you use the exact same conditions every time. That means the same stationary phase, the same mobile phase, the same temperature, and the same type of chromatography. If you change even one of these, your Rf value might change too! It's like playing a game with slightly different rules each time – the scores might not be comparable.
So, the next time you see a colourful separation in a lab or even a simplified illustration, you'll know the secret behind identifying those magical components. You’ll understand that behind every vibrant hue and distinct smell, there's a little bit of science, a bit of measurement, and a whole lot of fun in calculating that Rf value.
This simple calculation isn't just a scientific tool; it's a gateway to understanding the intricate chemistry that shapes our world. It empowers you to see beyond the surface and appreciate the hidden complexities that make everyday life so fascinating. So, don't be shy! Experiment with it, explore it, and let the world of chromatography and its intriguing Rf values spark your curiosity. You might just discover a newfound passion for the colorful, aromatic, and utterly amazing science that surrounds us!


