How Many Valence Electrons Does Niobium Have
Hey there, coffee buddy! So, we're diving into the world of Niobium today, huh? Super exciting stuff, I know. Who knew chemistry could be this… sparkly? We're going to talk about something called "valence electrons." Sounds fancy, right? But don't worry, it's not like we need a PhD in rocket science to figure this out. Think of it like this: these valence electrons are the little guys on the outside of an atom, the ones that are basically itching to go play with other atoms. They're the life of the party, the social butterflies of the atomic world. And understanding them tells us a whole lot about how an element is going to behave. Kind of like knowing if your friend is a homebody or always up for an adventure!
So, Niobium. Ever heard of it? Probably not unless you're, like, super into fancy kitchen knives or, you know, the bleeding edge of technological innovation. It's one of those elements that's hiding in plain sight, doing all sorts of cool stuff behind the scenes. It's a metal, for starters. A transition metal, to be exact. That means it's got a bit of an attitude, a bit of a rebellious streak. Transition metals are notoriously tricky when it comes to their electron configurations. They're not as straightforward as, say, your alkali metals, which are practically begging to give away an electron. Niobium is a little more… enigmatic.
Now, the big question: how many valence electrons does Niobium actually have? Drumroll, please! It's actually a little bit of a trick question, and I love a good trick question! See, it's not as simple as just looking at the group it's in on the periodic table and going, "Yep, that's the number!" Oh no, Niobium likes to keep us on our toes. It's like trying to guess how many cookies your roommate really ate last night. You think you know, but there's always a possibility of a sneaky extra one.
Must Read
So, let's break it down. When we're talking about valence electrons, we're generally thinking about the electrons in the outermost shell. The ones that are involved in chemical bonding. For most elements, it's pretty straightforward. Sodium? One. Chlorine? Seven. Easy peasy, lemon squeezy. But Niobium, being the diva it is, plays by slightly different rules. It’s like it decided the standard operating procedure was just too… common.
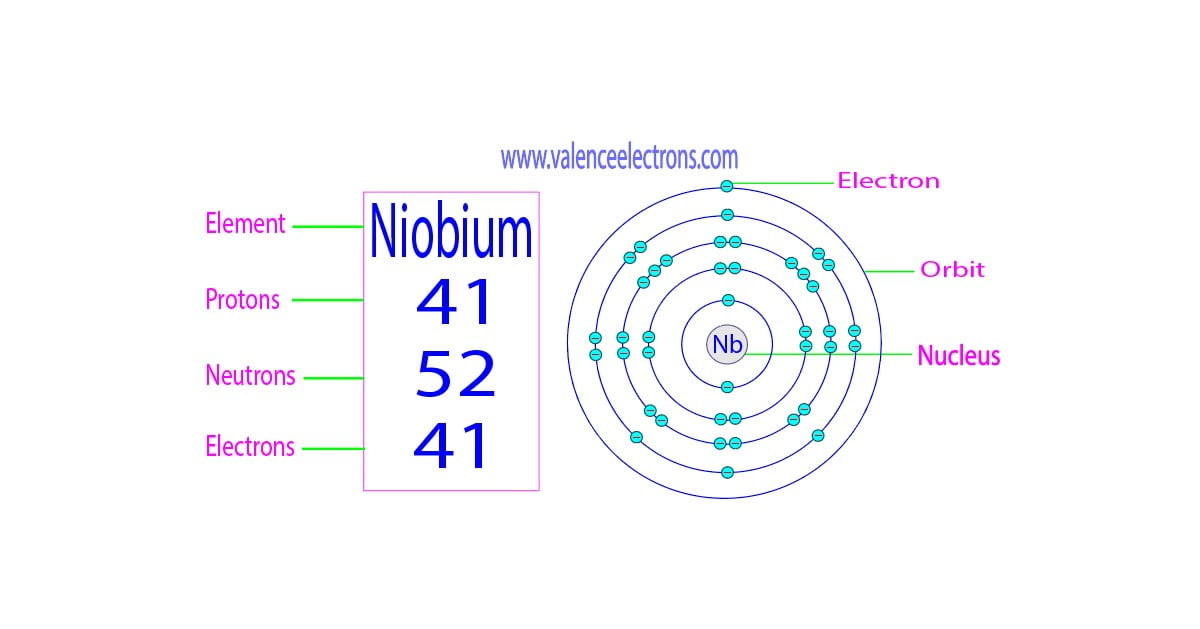
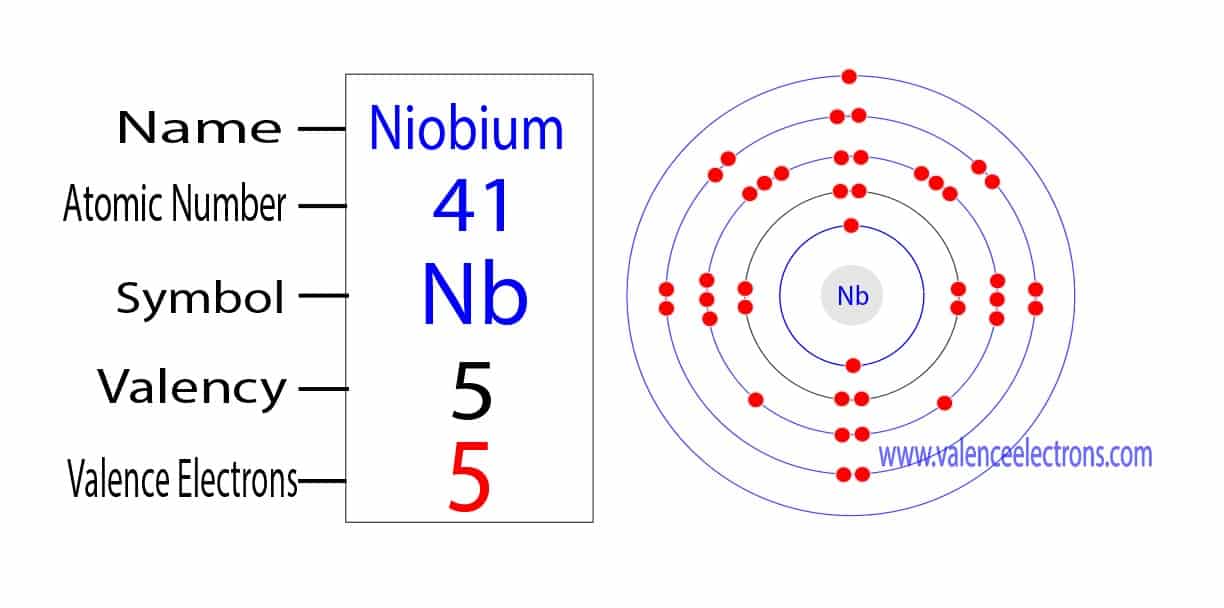
If you were to just glance at Niobium (atomic number 41, by the way, in case you wanted to impress your friends at your next trivia night… or just yourself), you might be tempted to say it has five valence electrons. And you wouldn't be entirely wrong! But you also wouldn't be entirely right. It's a bit of a Schrödinger's cat situation. It can behave like it has five, but it also has a sneaky little habit of dipping into its inner shells for a bit of bonding action. Talk about sharing is caring, right? Except in this case, it's sharing electrons it’s not supposed to be sharing!
The Periodic Table's Little Quirk
Okay, so let’s talk about that trusty periodic table. It’s our cheat sheet, our Rosetta Stone for understanding atoms. Niobium sits in Group 5. And for many elements in Group 5, like Vanadium or Tantalum (Niobium's close cousins, you could say), they do have five valence electrons. So, naturally, we'd assume Niobium is just one of the gang. And in many ways, it is! It's got that same fundamental electron setup in its outermost s and p orbitals.
But here's where Niobium decides to be a rebel. It has a very stable electron configuration where it has a full 4d subshell. Think of subshells like little apartments within the electron shell. And when an apartment is full, well, the electrons in there are pretty happy and don't really want to be disturbed. They're like, "Leave me alone, I'm comfortable here, I've got my snacks and my Netflix."

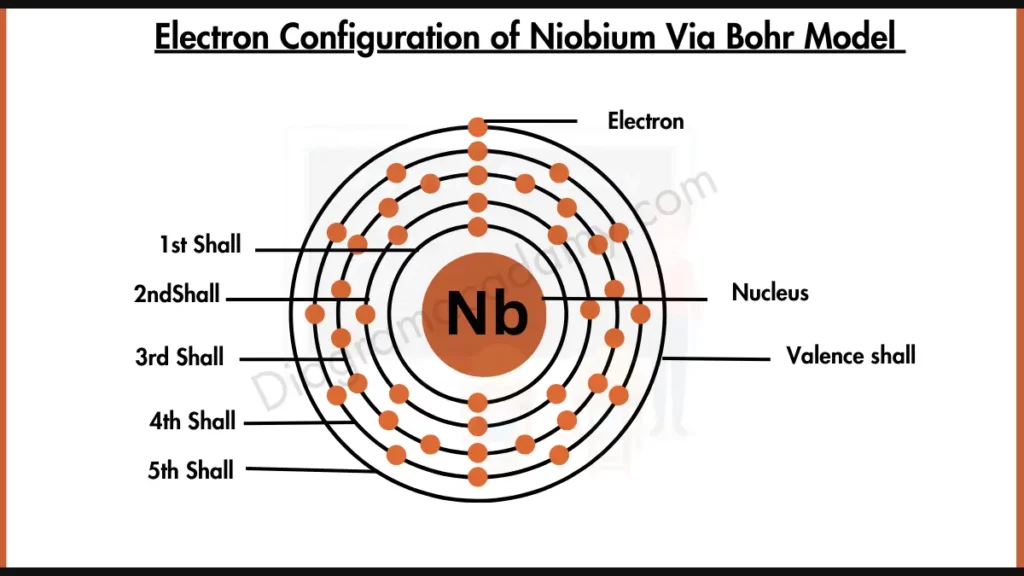
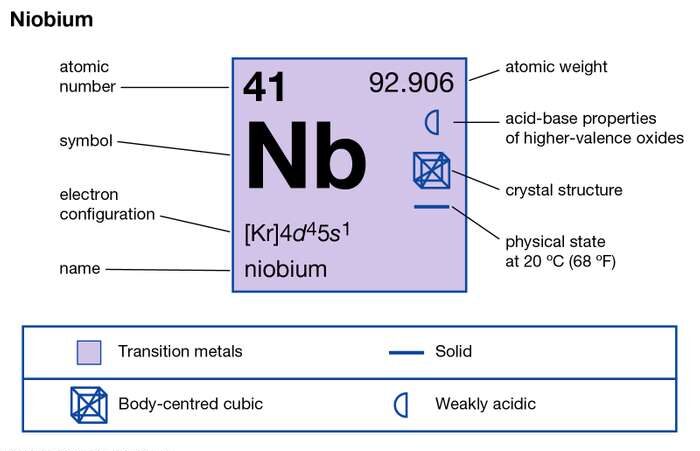
So, Niobium’s electron configuration looks something like [Kr] 4d⁴ 5s¹. Now, that might look like a typo, right? Four d electrons and only one s electron in the outermost shell? That’s not the typical 5s² 4d³ or something more "normal" for Group 5. And that's the magic! That 5s¹ electron is definitely a valence electron. It's out there, ready to mingle. But what about those 4d electrons? They’re so close, and they’re in a subshell that’s almost full.
Because that 4d subshell is so close to being completely filled with 10 electrons, Niobium can, and does, often involve those 4d electrons in its chemical reactions. So, while it technically has one electron in its outermost s orbital, it often behaves as if it has more. It can be a bit of a show-off, pulling in those d electrons to make bonds. It’s like it's saying, "Why have one trick when you can have… well, more than one trick?"
So, What's the Official Answer?
Alright, let's get down to brass tacks, or in this case, brass electrons. The most common way Niobium participates in chemical bonding is by using its single 5s electron and also some of its 4d electrons. This means it often exhibits oxidation states that suggest it’s contributing more than just that one lone electron. For instance, you'll often see Niobium in a +5 oxidation state. How does it get to +5 if it only has one 5s electron? You guessed it! It's using those 4d electrons too!

So, if someone asks you, "How many valence electrons does Niobium have?" the most accurate and nuanced answer is: Niobium typically behaves as if it has five valence electrons, even though its outermost electron configuration is a bit unusual. It’s got that one 5s electron that's clearly a valence electron, but it also readily utilizes electrons from its nearly filled 4d subshell in bonding. It’s that unique electron arrangement that makes Niobium so interesting and versatile.
It’s like a chameleon, you know? It can adapt its "valence electron count" depending on the situation. Some chemists might argue about the precise definition, and that's okay! Science is all about debate and refinement. But for practical purposes, when you're thinking about how Niobium will react, thinking of it as having about five electrons available for bonding is a pretty good bet. It's got that flexibility, that adaptability, that makes it a superstar in high-performance applications.

Think about it: this element, with its slightly quirky electron setup, is used in superconducting magnets (hello, MRI machines!), jet engines (super strong and heat resistant!), and even fancy jewelry (yes, really!). It's not just about having a certain number of electrons; it's about how those electrons are arranged and how they can be used. Niobium's arrangement is a bit of a masterclass in chemical efficiency.
It's kind of like when you're packing for a trip. You might have a general idea of what you need, but you also know you can probably borrow your friend's charger or use that hotel hairdryer. Niobium is like that friend. It’s got its own core set of "belongings" (the 5s electron), but it's also willing to "borrow" from its "neighboring drawers" (the 4d subshell) when the situation calls for it. This makes it incredibly useful in all sorts of chemical reactions and material science applications.
So, in conclusion, don't be fooled by the simple "group number equals valence electron count" rule when it comes to Niobium. It's a bit of an outlier, a bit of a rebel. But that's what makes it so special! It's the element that proves sometimes, the rules are meant to be bent, or at least interpreted with a little bit of… artistic license. And that's what makes understanding elements like Niobium so darn fascinating. It's not just about memorizing numbers; it's about understanding the why behind them. And the "why" for Niobium is a beautifully complex dance of its electrons, allowing it to be incredibly useful and resilient. Pretty neat, right? Now, who needs a refill?