How Many Valence Electrons Does Mg2+ Have
Ever wondered what makes different substances behave the way they do? It's all thanks to the tiny, energetic particles called electrons that buzz around the center of every atom. And when we talk about how atoms connect and form the world around us, one concept really shines: valence electrons! They're like the handshake of an atom, determining how it interacts with others. Today, we're going to dive into a specific, and actually quite fun, question: how many valence electrons does Mg2+ have?
Understanding valence electrons is super useful for so many people. For beginners just dipping their toes into chemistry, it's a foundational concept that unlocks how elements form bonds. Think of it as learning your ABCs before you can write a story! For families exploring science together, it's a fantastic way to make everyday observations more meaningful. Why does water stick to itself? Why is salt brittle? Valence electrons hold the clues! And for hobbyists, whether you're into gardening (soil chemistry!), brewing (fermentation!), or even crafting (understanding material properties!), this knowledge can add a whole new layer of appreciation.
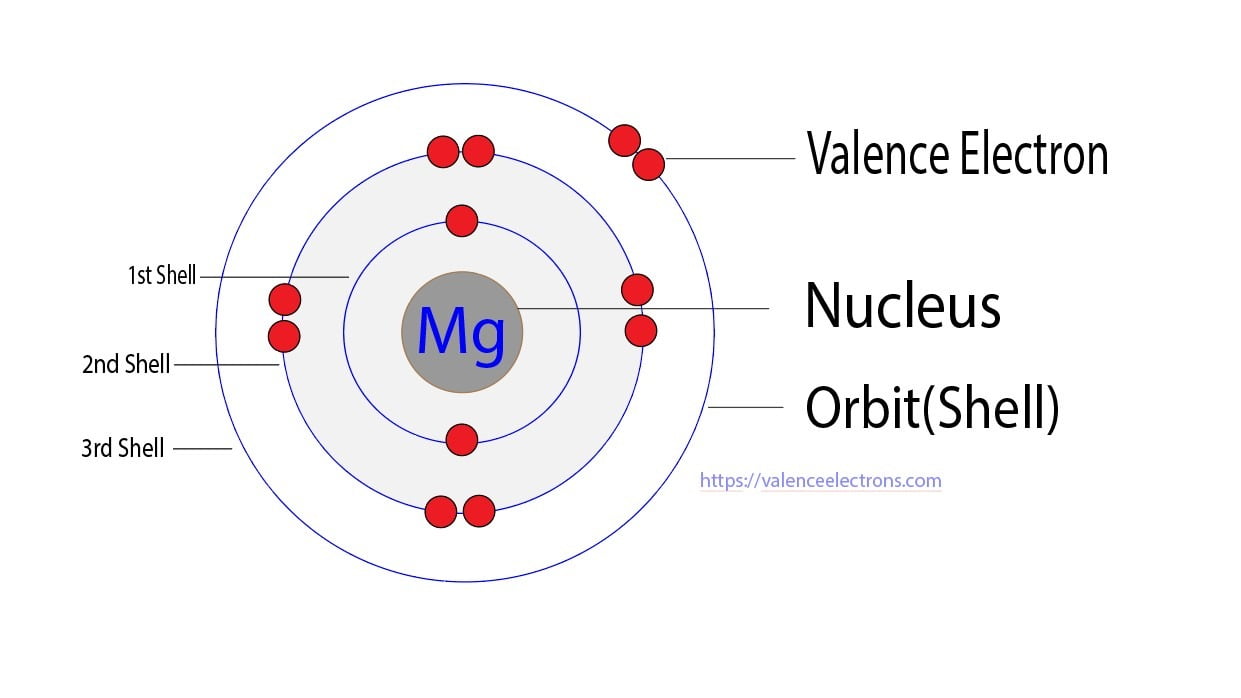
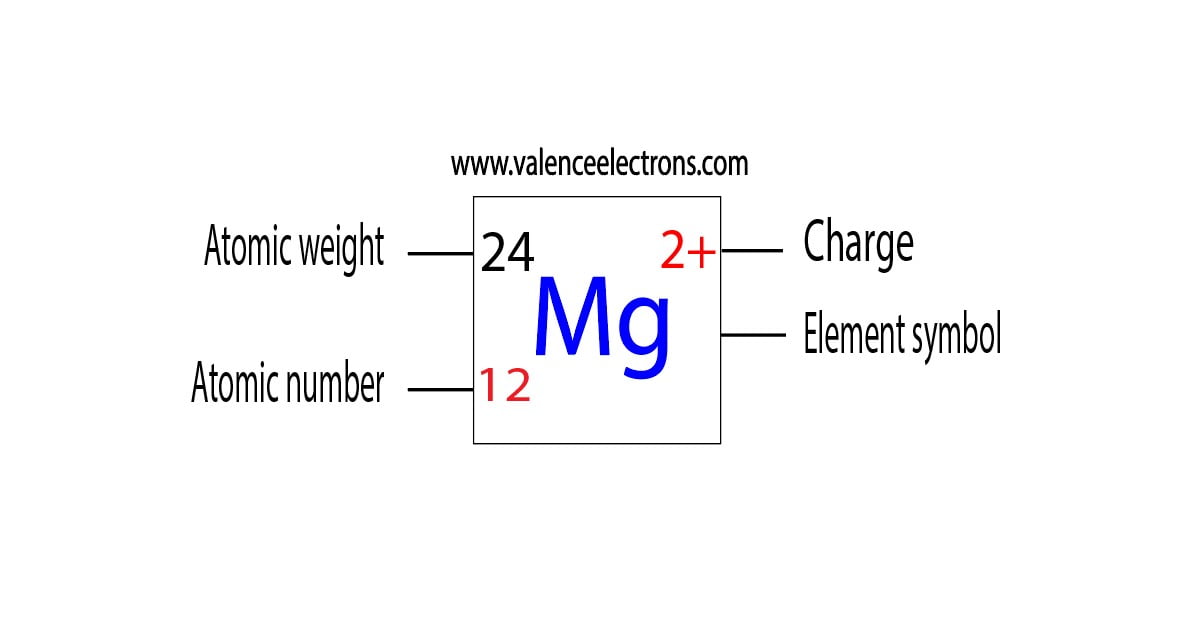
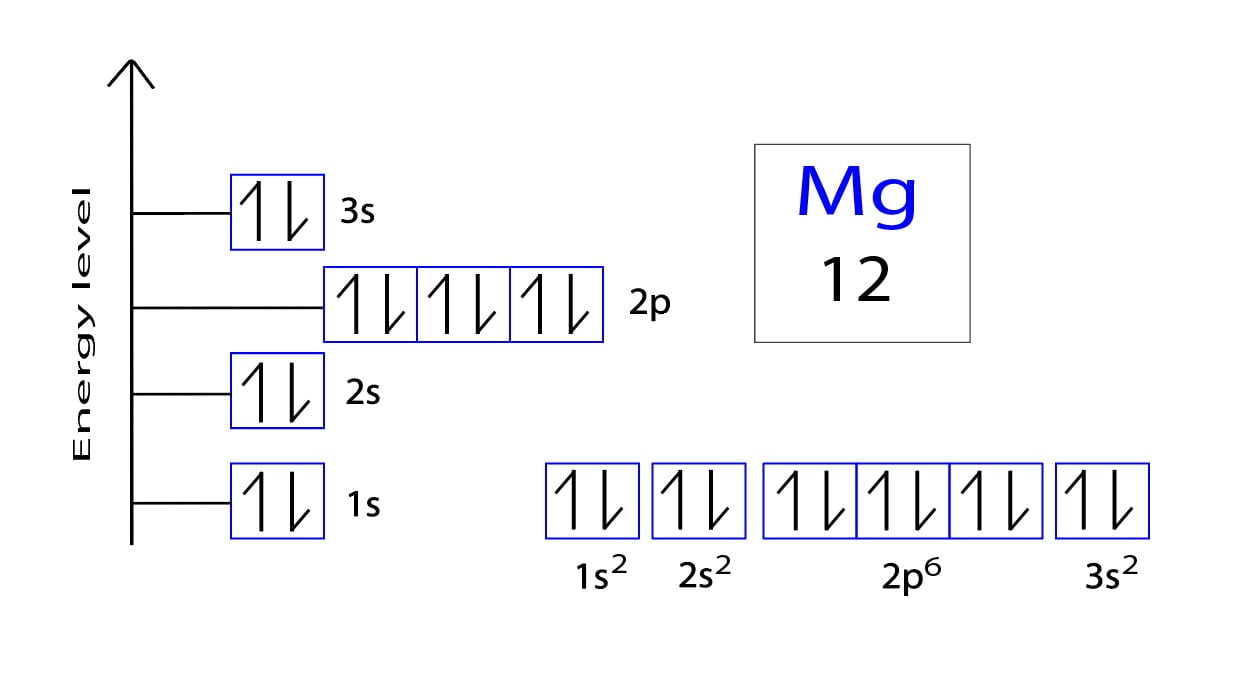
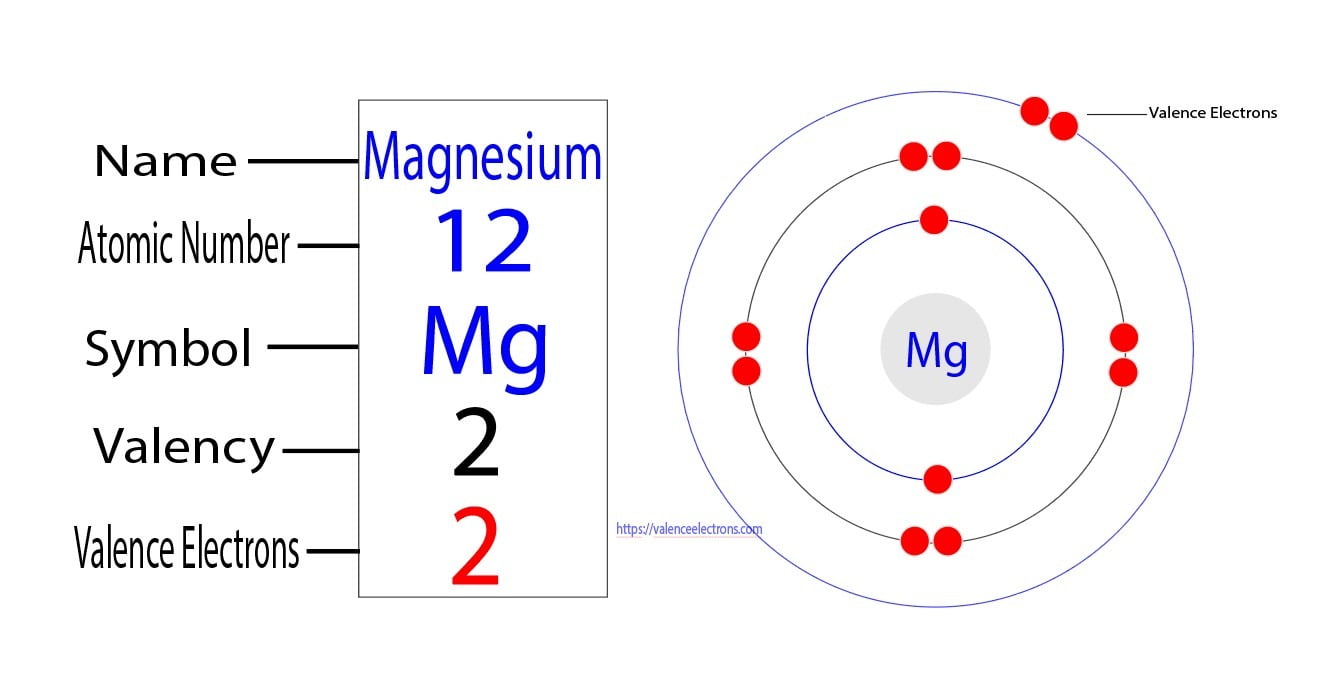
So, let's tackle our specific question: Mg2+. First, we need to think about its parent atom, Magnesium (Mg). A neutral Magnesium atom has 12 electrons in total. These electrons are arranged in shells, and the outermost shell is where the magic happens – those are the valence electrons! For Magnesium, it has 2 valence electrons in its outer shell. Now, here's where the '2+' comes in. That little '+' sign tells us that this Magnesium atom has lost 2 electrons. Why would it do that? To become more stable, like many atoms like to do! Since Magnesium started with 2 valence electrons, and it lost those 2 electrons, what's left in its outermost shell? You guessed it! Zero valence electrons.
Must Read
This isn't the only interesting variation. Consider Chlorine (Cl). A neutral Chlorine atom has 7 valence electrons. If it gains one electron to become a Cl- ion, it now has 8 valence electrons, which is a very stable configuration. Or think about Sodium (Na). A neutral Sodium atom has 1 valence electron. When it loses that electron to become a Na+ ion, it also ends up with 0 valence electrons in its outermost shell, making it very stable and ready to interact with things like Chlorine!

Getting started with understanding valence electrons is easier than you might think! You can start by looking at a periodic table. Most periodic tables will give you clues about the number of valence electrons based on the element's group (its column). For example, elements in Group 1 (like Sodium) usually have 1 valence electron, and elements in Group 2 (like Magnesium) usually have 2. You can then practice figuring out how many electrons ions might have lost or gained by looking at their charge.
In the end, exploring how many valence electrons an ion like Mg2+ has isn't just about memorizing facts. It's about understanding the fundamental forces that shape our universe, from the tiniest atoms to the grandest galaxies. It's a journey that's both intellectually stimulating and incredibly practical, making the world around you just a little bit more understandable and a lot more fascinating!