How Many Valence Electrons Are In Rubidium

Ever wondered what makes things tick, or why some elements are super eager to bond and others are a bit more reserved? It all comes down to the tiny, but mighty, valence electrons! Think of them as the social butterflies of the atomic world, the ones that get to mingle and make connections. And today, we're going to zoom in on a particularly interesting element and uncover just how many of these social butterflies it has. Get ready to have some fun with chemistry, because we're diving into the world of Rubidium!
Understanding valence electrons is like having a secret decoder ring for the entire periodic table. It tells us so much about how an element will behave, what kind of reactions it will get involved in, and why it has the properties it does. It’s the key to unlocking why water is so refreshing, why salt is salty, and why metals conduct electricity so well. Knowing about valence electrons isn't just for super-scientists in lab coats; it’s a fundamental piece of the puzzle that explains the world around us. And for Rubidium, its valence electron count reveals some fascinating traits that make it a star player in certain areas of science and technology. So, let's get to the bottom of it!
The Intriguing World of Rubidium
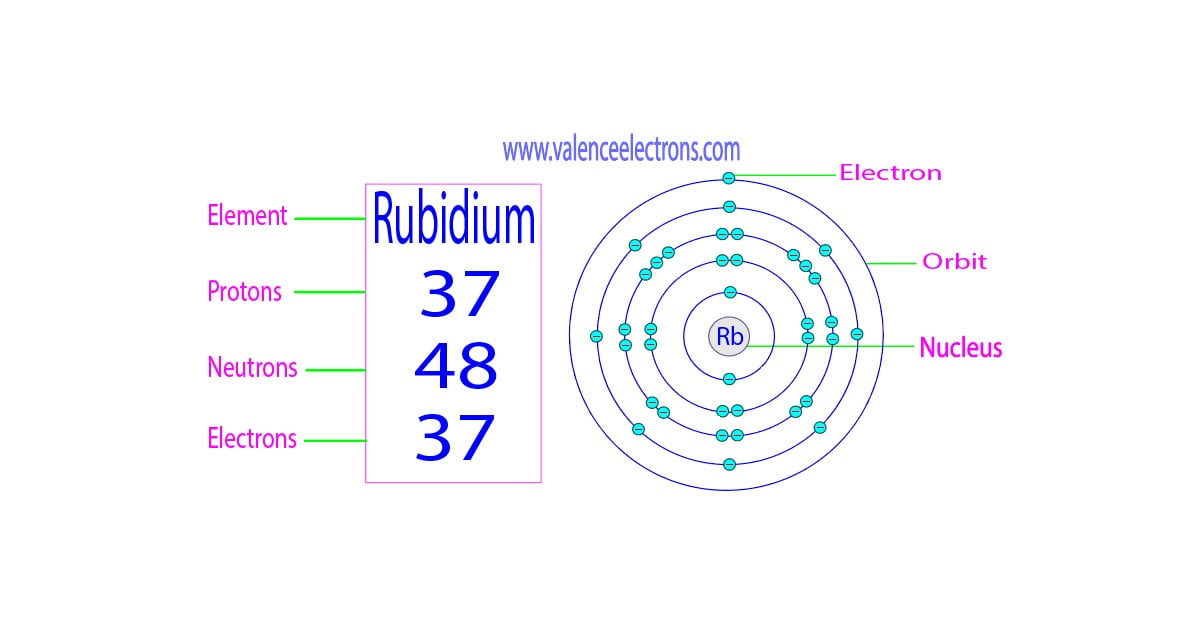
Let’s talk about Rubidium, shall we? This element, symbolized by Rb, is a fascinating character on the periodic table. It belongs to the alkali metal group, a family known for being highly reactive and having a distinct set of behaviors. You won't find pure Rubidium just hanging out on your kitchen counter; it's too eager to react with things like air and water to be handled casually. In fact, it’s often stored under oil or in sealed containers to prevent these lively interactions.
Must Read
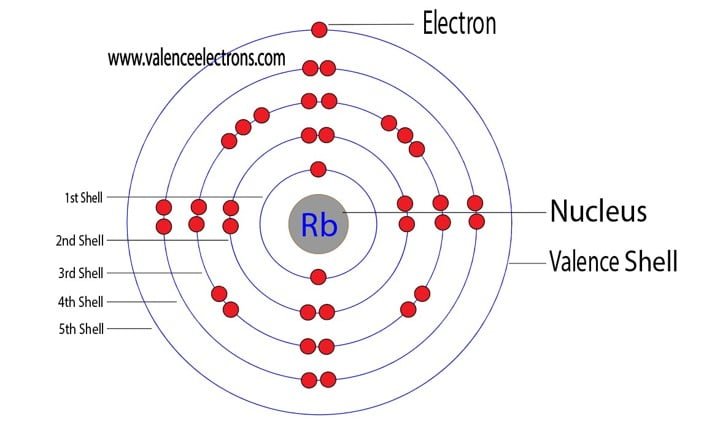
When you look at the periodic table, you'll find Rubidium sitting pretty in the first column, also known as Group 1. This prime real estate is a big clue about its electron configuration. Elements in Group 1 share a common characteristic: they all have a specific number of electrons in their outermost shell. This outermost shell is where the magic happens when elements start interacting, and the electrons residing there are our all-important valence electrons.
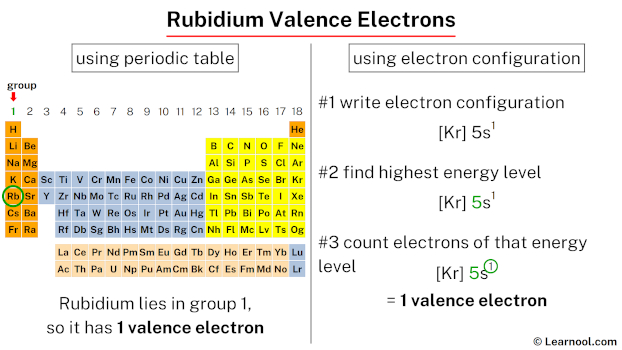
Now, how do we figure out how many valence electrons Rubidium has? It's actually quite straightforward if you know where to look on the periodic table. The number of valence electrons for most main-group elements (that's the tall columns on the left and right sides of the periodic table) directly corresponds to their group number. For elements in Group 1, this number is a neat and tidy one.

So, to answer the big question: Rubidium has one valence electron.
This single, solitary electron in its outermost shell is the driving force behind much of Rubidium's personality. It’s like having one extra toy you’re just itching to share or trade. This electron is not held very tightly by the nucleus, making Rubidium very willing to lose it to achieve a more stable electron configuration. When an atom easily gives up an electron, it becomes a positively charged ion, and this tendency is what makes Rubidium so reactive.
The benefits of understanding this are huge! Knowing that Rubidium has just one valence electron helps scientists predict how it will react with other elements. For instance, it's highly likely to react with elements that need to gain an electron to become stable, like halogens (elements in Group 17 such as chlorine or bromine). This leads to the formation of ionic compounds, where one atom gives an electron and another atom takes it, creating a strong electrostatic attraction.

This reactivity also makes Rubidium useful in various applications. Its ability to emit electrons easily when heated or exposed to light (a phenomenon called photoemission) is why it's used in vacuum phototubes and other light-sensitive devices. Furthermore, Rubidium-87, a specific isotope of Rubidium, is crucial in atomic clocks, which are the most accurate timekeeping devices known. The precise transitions of electrons within Rubidium atoms allow for incredibly accurate measurements of time, essential for everything from GPS systems to scientific research.
The fact that Rubidium has just one valence electron isn't just a dry fact; it's a gateway to understanding its role in the universe and its practical applications here on Earth. It’s a beautiful example of how the seemingly simple arrangement of electrons dictates the complex and fascinating behavior of matter. So next time you hear about Rubidium, you'll know that its single valence electron is the key to its energetic and useful life!


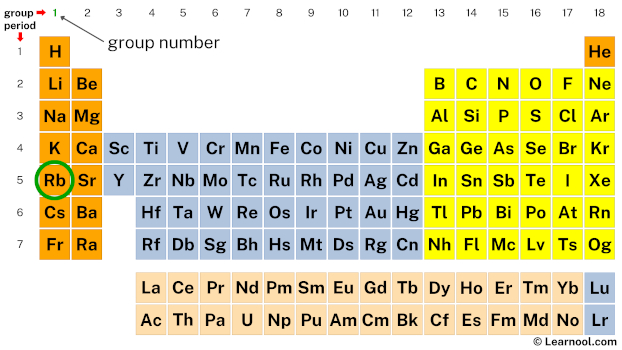
![Rubidium Electron Configuration: [Kr] 5s¹ and Full Notation](https://valenceelectrons.com/wp-content/uploads/2021/11/RubidiumRb-electron-configuration-1024x461.jpg)
