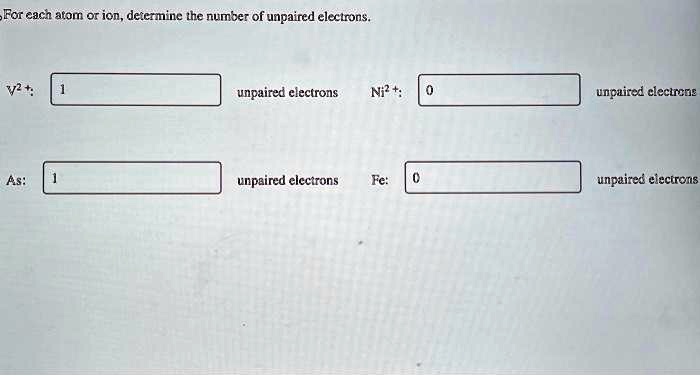
How Many Total Electrons Are In A Ni2+ Ion
You know, I remember the first time I really wrestled with the concept of ions. I was in high school chemistry, and our teacher, Mr. Henderson (bless his patient soul), was trying to explain how atoms could gain or lose electrons. I was staring at a periodic table that looked like a Jackson Pollock painting to me, and he pointed to Nickel, like, "Okay, so Nickel can become Ni2+." My brain did this little whirring sound, followed by a distinct thud. Ni2+? What does that even mean? Was it just a fancy way of saying Nickel was having a bad day? Little did I know, that seemingly simple "plus two" was going to unlock a whole new understanding of how atoms behave. It felt like trying to decipher a secret code, and I was determined to crack it.
And that’s exactly what we’re going to do today! We’re going to crack the code on the total number of electrons in a Ni2+ ion. It sounds a bit like detective work, doesn't it? Except instead of a shadowy figure, we're looking for a missing or extra particle. And trust me, it’s way more fascinating than any fictional mystery.
So, let’s start with the basics, shall we? Because you can’t really understand the Ni2+ without getting to know its parent, the humble Nickel atom. Think of the atom as the original, the "before" picture. When we talk about atoms, we're usually talking about them in their most neutral state. And in that neutral state, they’re perfectly balanced. Like a seesaw with an equal number of people on both sides.
Must Read
What makes an atom neutral? It’s all about the protons and electrons. Protons hang out in the nucleus, positively charged little guys. Electrons zip around the outside, negatively charged. In a neutral atom, the number of positive protons exactly equals the number of negative electrons. It’s a perfect harmony, a chemical equilibrium. And this number, the number of protons (which also dictates the number of electrons in a neutral atom), is super important. It’s like the atom’s fingerprint, its unique identifier. This is what we call the atomic number.
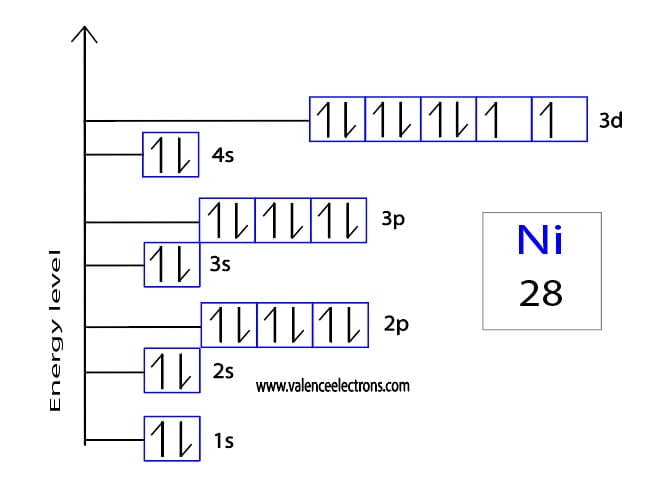
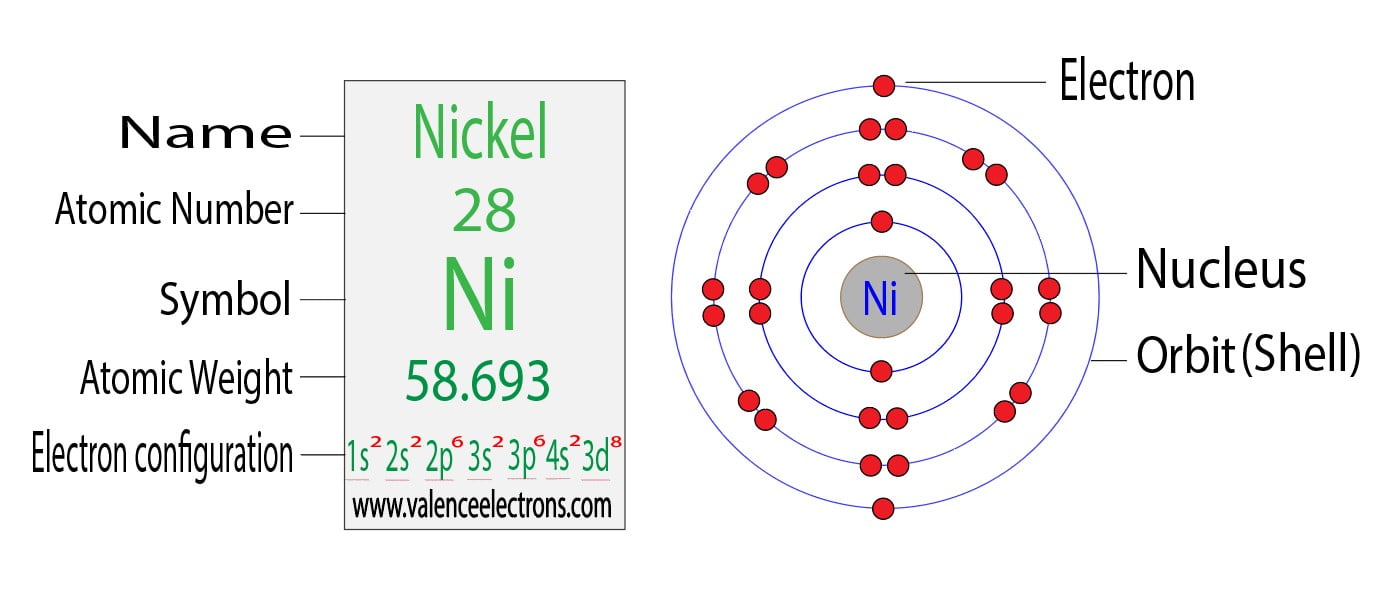
Now, where do we find Nickel on this colorful periodic table? If you’ve got one handy, take a peek. Nickel, with the symbol Ni, has an atomic number of 28. That’s a pretty significant number, right? So, a neutral Nickel atom has 28 protons in its nucleus.
And because it’s a neutral atom, it also has 28 electrons zipping around its electron shells. See? Perfect balance. 28 positive charges from the protons, and 28 negative charges from the electrons, cancelling each other out. Easy peasy so far, right? You’re probably thinking, “Okay, I’ve got this. Just tell me the answer already!” But stick with me, because the real fun starts when things get a little unbalanced.
The "Plus Two" Mystery Unveiled
So, what happens when Nickel decides it’s not content with being perfectly neutral anymore? This is where the "Ni2+" comes in. That little "2+" is the key. It's not just a random addition; it's a declaration of intent. It tells us that our Nickel atom has undergone a transformation. It’s no longer just "Ni"; it's now an ion.
What exactly is an ion? In simple terms, an ion is an atom (or a molecule) that has gained or lost one or more electrons. Because electrons carry a negative charge, losing electrons makes the atom more positive, and gaining electrons makes it more negative. It’s like adding or removing weights from that seesaw we talked about earlier. If you take off a weight (an electron), the other side (protons) becomes heavier, making the whole thing tilt positively.
Now, look back at that "Ni2+". What do you think that "2+" signifies? Yep, you guessed it! It means that the Nickel atom has become two units more positive. How does an atom become more positive? By losing those negatively charged electrons. It’s the only way to shift the balance towards a positive charge. Protons, bless their stable little hearts, don't just go around leaving the nucleus. Electrons, however, are a bit more… footloose and fancy-free.
So, a Ni2+ ion means that a neutral Nickel atom has lost 2 electrons. It’s shed two of its negative charges, leaving the positive charges (from the protons) in the majority. Think of it like this: you had 28 apples (electrons) and 28 oranges (protons). Everything was balanced. Then, oops, you dropped 2 apples in the river. Now you have 26 apples and 28 oranges. The oranges are now more numerous than the apples, making the fruit bowl have a net "orange-ness" or, in chemistry terms, a net positive charge!
This is why it's called a cation. Positive ions are called cations. The "cat" in cation is often thought to be related to the word "catastrophe" (if you're a negative ion, that is!), or more accurately, it’s named after the positive electrode, the anode, where positive ions migrate. Pretty neat, huh? (Or maybe just a bit of etymology trivia for you.)
The Grand Total: Calculating the Electrons
Okay, drumroll please! We're ready for the grand finale. We know that a neutral Nickel atom has 28 electrons. We also know that a Ni2+ ion is formed when a Nickel atom loses 2 electrons.
So, to find the total number of electrons in a Ni2+ ion, we simply take the original number of electrons in a neutral Nickel atom and subtract the number of electrons that were lost.
Total electrons in Ni2+ = (Electrons in neutral Ni) - (Electrons lost)
Total electrons in Ni2+ = 28 - 2
And what does that equal? You got it!

Total electrons in Ni2+ = 26 electrons.
So, there you have it! A Ni2+ ion has a total of 26 electrons. It's not 28, and it's definitely not 30. It's 26. It still has all 28 of its protons, but it's now rocking 26 electrons. That’s what gives it that 2+ charge.
It’s a bit like having a really popular band. The original band (neutral atom) has all its members (protons and electrons). Then, two of the rockstars (electrons) decide to go off and start a solo career. The core members (protons) are still there, but the entourage (electrons) is a bit smaller, which changes the overall vibe, or in this case, the charge.
This concept of gaining or losing electrons is fundamental to so much of chemistry. It's why metals form positive ions and often participate in ionic bonding, attracting negatively charged non-metals. It's how batteries work, how we get electricity flowing, and how life itself happens on a molecular level. Pretty mind-blowing when you think about it, considering it all starts with a simple atom deciding to shed a couple of electrons.

Sometimes, I think we humans are a bit like ions too. We gain experiences, we lose old habits, and we transform. We’re constantly changing our "electron configuration" of life, so to speak. Maybe that’s why these abstract chemical concepts can sometimes feel so relatable.
So, the next time you see a chemical formula with a little plus or minus sign on it, don’t just dismiss it as a typo or some obscure notation. It’s a story. It’s the story of an atom that has been through something, that has changed its electron count. And understanding those small numbers is the first step to understanding the entire incredible world of chemistry.
It’s fascinating to think about the journey of these electrons. They’re not just sitting there; they’re in constant motion, orbiting the nucleus, participating in chemical reactions. When Nickel becomes Ni2+, those two lost electrons don’t just vanish into thin air. They go somewhere, usually to another atom or molecule, and that interaction is what makes chemistry so dynamic and, frankly, so interesting. They're like little messengers of charge, carrying the story of transformation from one atom to another.
And remember, it’s not just Nickel. This principle applies to all ions. Whether it’s Sodium losing one electron to become Na+ (10 electrons!) or Chlorine gaining one electron to become Cl- (18 electrons!), the logic is the same. You start with the neutral atom’s electron count and then you add or subtract based on the charge. It’s a universal language of atomic interactions.
So, go forth and impress your friends with your newfound knowledge of Ni2+ electron counts! Or at least, be able to explain why that little "2+" is so darn important. It’s a testament to the fact that even the smallest particles have their own little dramas and transformations. And we, as curious observers, get to decode them. Pretty cool gig, if you ask me. Keep exploring, keep asking questions, and never underestimate the power of a simple number.