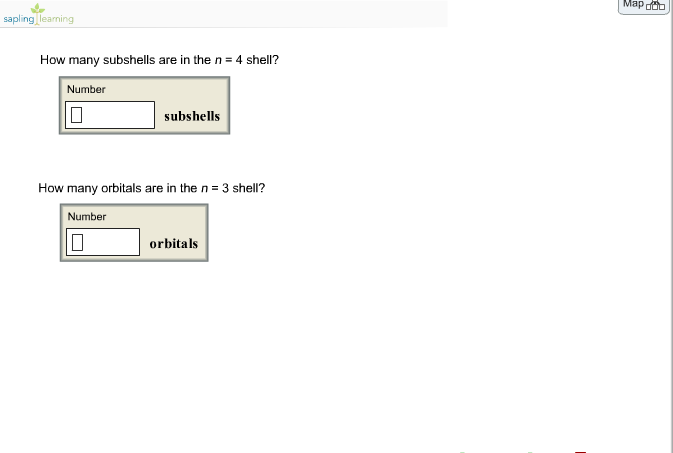
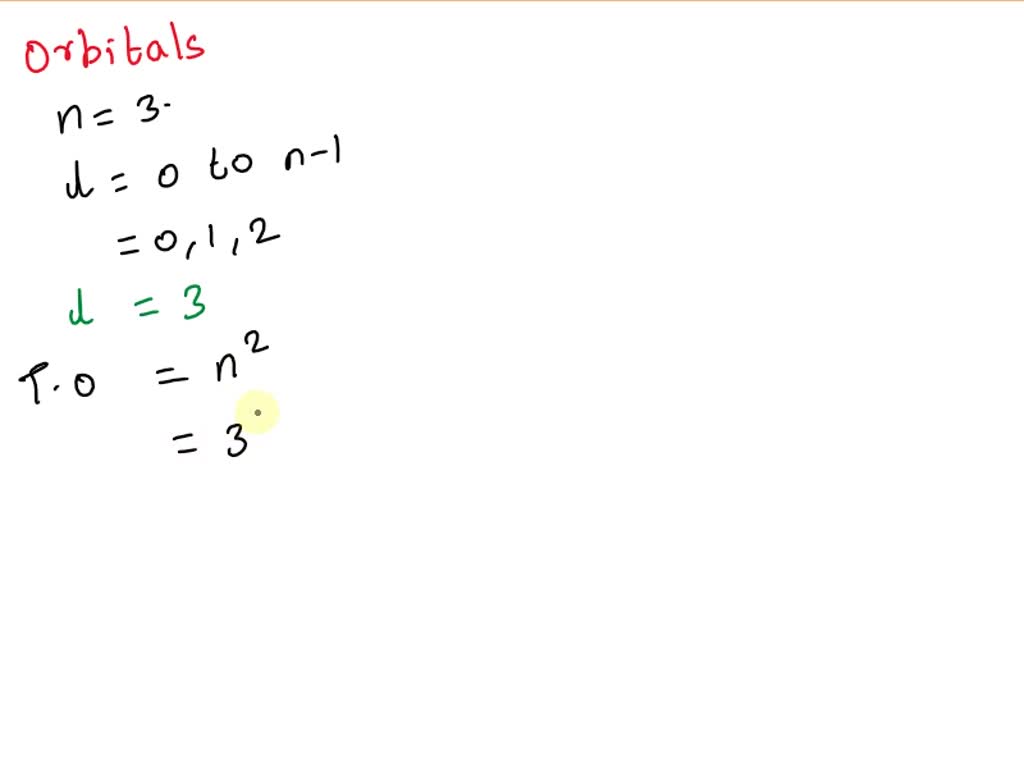How Many Subshells Are Associated With N 4

Alright folks, gather 'round, pull up a chair. Let's talk about something that sounds a bit like a secret code from a sci-fi movie, but is actually, dare I say, kinda cool. We're diving into the mysterious world of atoms, specifically, the n 4 energy level. Now, before you start picturing tiny little atoms doing the tango, let's break it down. Think of it like this: an atom is basically a miniature solar system, but instead of planets orbiting a sun, you've got electrons zipping around a nucleus. And just like our solar system has different "zones" or "orbits" for planets, atoms have these things called energy levels.
These energy levels are denoted by a number, and our focus today is on the big cheese, the main event: n 4. Imagine it as the fourth floor of a ridiculously tall apartment building for electrons. Higher floors mean more energy, more space, and… well, more stuff going on. And when we talk about "stuff going on" in these electron apartments, we're talking about subshells. These are like the different types of apartments on each floor. You could have a studio, a one-bedroom, a penthouse, a downright weird geodesic dome – you get the idea.
So, the million-dollar question (or, you know, the zero-dollar question since I’m giving this to you for free, you're welcome) is: how many subshells are associated with n 4?
Must Read
Now, for the seasoned atom-whisperers out there, this might be a no-brainer. But for the rest of us who are still trying to remember if an electron is a particle or a tiny, angry badger, let's illuminate the path. The number of subshells that can exist at a particular energy level (our 'n' value) is surprisingly straightforward. It's not some cosmic riddle; it’s actually equal to the value of 'n' itself! Mind. Blown. Right?
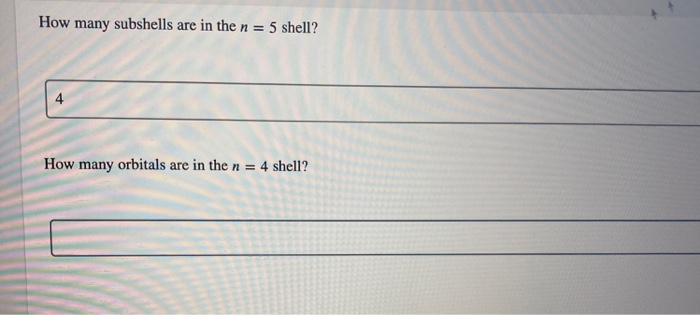
So, if n is 1, you get 1 subshell. If n is 2, you get 2 subshells. If n is 3, you get 3 subshells. And if n is, you guessed it, 4, then we're looking at… four subshells!

Yep, that’s it. Four. It's not like it’s a Fibonacci sequence or a secret handshake. Just plain old four. Now, you might be thinking, "Okay, four subshells. But what do we call these things? Do they have names like 'The S-loth', 'The P-enguin', 'The D-ragonfly', and 'The F-luffy-bunny'?"
While I appreciate the creativity, the actual names are a bit more… utilitarian. They’re represented by letters: s, p, d, and f. Think of them as the "first four letters of the alphabet for electron apartments." And guess what? These letters correspond to the shapes of the regions where you're most likely to find these pesky electrons. The 's' subshell is a sphere – like a perfectly round bouncy ball. Simple, elegant, and probably what the minimalist electrons prefer.
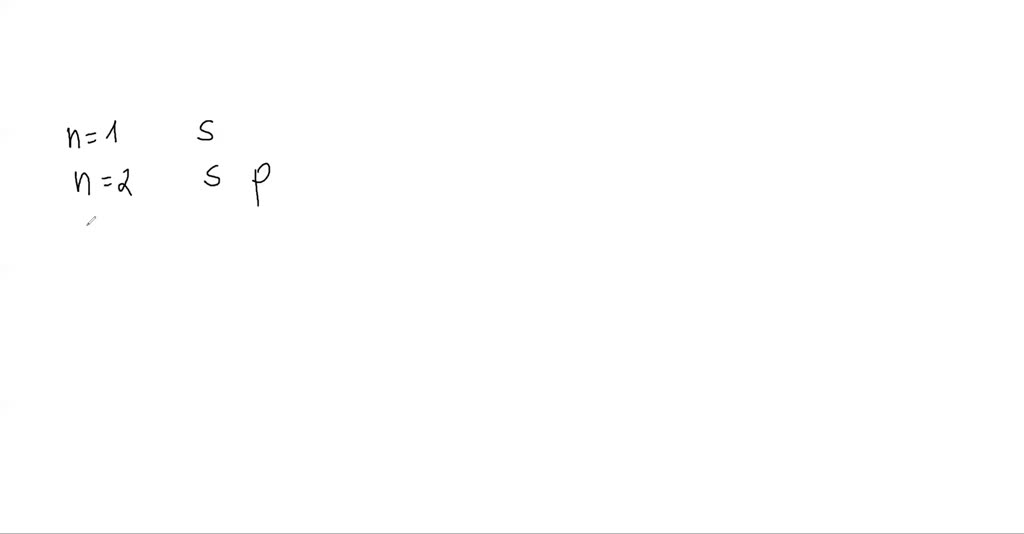
Then we have the 'p' subshell. These are shaped like dumbbells, or maybe like two balloons tied together at the knot. They’re a bit more directional, sticking out in different directions. Imagine tiny, infinitely precise party balloons. Very organized party balloons.
The 'd' subshell? Oh, these are where things get interesting. They're a bit like cloverleaves, or maybe four interconnected balloons. They're more complex, more… spacious. These are the fancy loft apartments with multiple views.
And finally, the 'f' subshell. These are the truly exotic ones, with shapes that look like they were designed by a surrealist artist who’d had a bit too much espresso. They’re even more complex and have more lobes than a particularly enthusiastic ear. Honestly, trying to visualize an 'f' subshell is like trying to fold a fitted sheet – a noble effort, but prone to confusion.
So, for our n 4 energy level, we have all four of these types of subshells available: the 4s, 4p, 4d, and 4f. It’s like the fourth floor of our electron hotel has every single room option available, from the basic studio ('s') to the ridiculously complex presidential suite ('f').
Now, here's a little nugget of atomic trivia for you: you might think that as the energy level 'n' gets bigger, you’d just keep getting new letters for subshells. Like after 'f', maybe we’d have 'g', 'h', 'i', and so on. But nope! The alphabet soup of electron subshells effectively stops at 'f' in the vast majority of cases we encounter. If an atom were to get so big that it needed an 'f' subshell, it would likely be so unstable that it would have spontaneously combusted or dissolved into pure existential dread long before we got there.
Think about it: a 'g' subshell would be the quantum equivalent of a super-duper-mega-mansion for electrons. Imagine the energy bill! The sheer complexity of just finding an electron in a 'g' subshell would give a quantum physicist a migraine that could power a small city. So, 'f' is usually the deepest dive we take into the intricate world of subshell shapes.
Therefore, when you hear "n 4," just picture that fourth floor, brimming with possibility. It’s got the reliable spheres of the 4s, the directional dumbbells of the 4p, the stylish cloverleaves of the 4d, and the wonderfully weird, almost avant-garde shapes of the 4f. All four of them are there, ready to house electrons, ready to contribute to the grand dance of chemistry. It’s a bustling neighborhood, the n 4 level!
So, to recap, without getting too bogged down in the theoretical weeds (because who needs more weeds when you've got electrons?), the number of subshells associated with any given energy level 'n' is simply 'n' itself. And for our star of the show, n 4, that means four subshells: s, p, d, and f. It’s a fundamental rule, a building block of atomic understanding. And now, you know it! Go forth and impress your friends, your cat, or perhaps just yourself with this knowledge. You’ve officially leveled up your atomic understanding!


![[ANSWERED] How many possible sublevels subshells are in n 4 32 O 2 4](https://media.kunduz.com/media/sug-question-candidate/20220621221649053696-3498554.jpg?h=512)