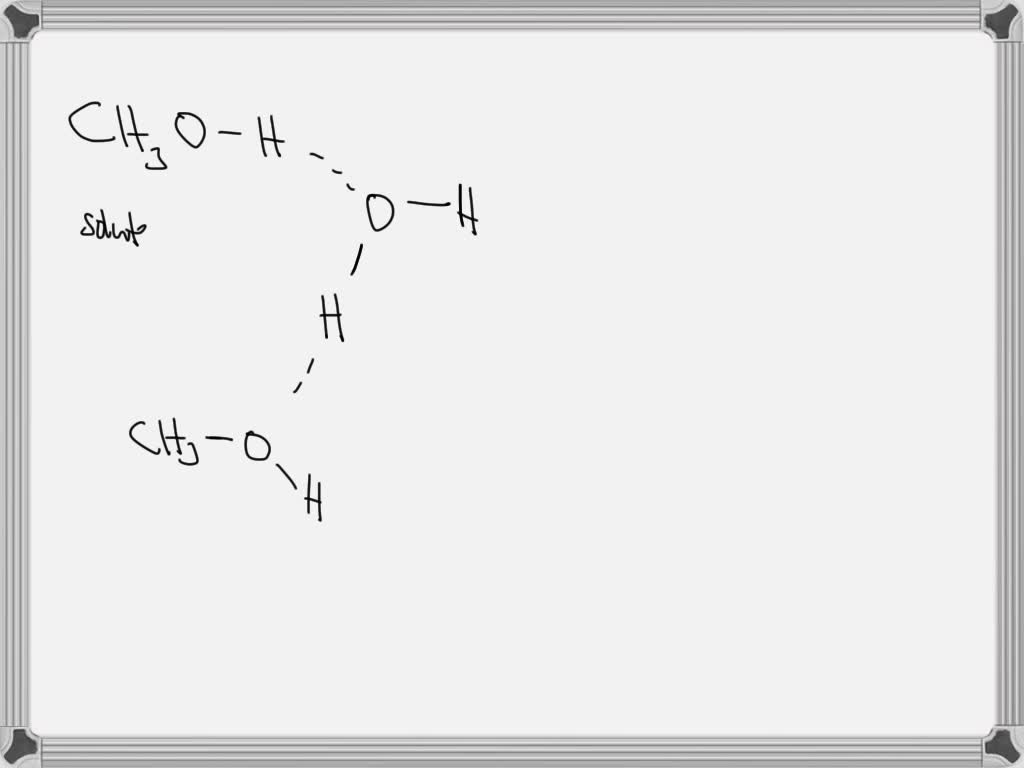How Many Hydrogen Bonds Can Methanol Form

Hey there, science curious folks! Ever wondered about the tiny, invisible connections that make molecules do their thing? Today, we're diving into the super cool world of methanol and its special handshake with other molecules. Get ready, because we're talking about hydrogen bonds, and methanol is a surprisingly charming participant in this molecular dance.
So, what's the big deal about methanol? Well, it's a pretty simple molecule. Think of it as a tiny little buddy with a carbon atom, three hydrogen atoms hanging out with it, and one extra special hydrogen atom attached to an oxygen atom. That little OH group is where all the magic happens. It's like the molecule's friendly waving hand, ready to connect with others.
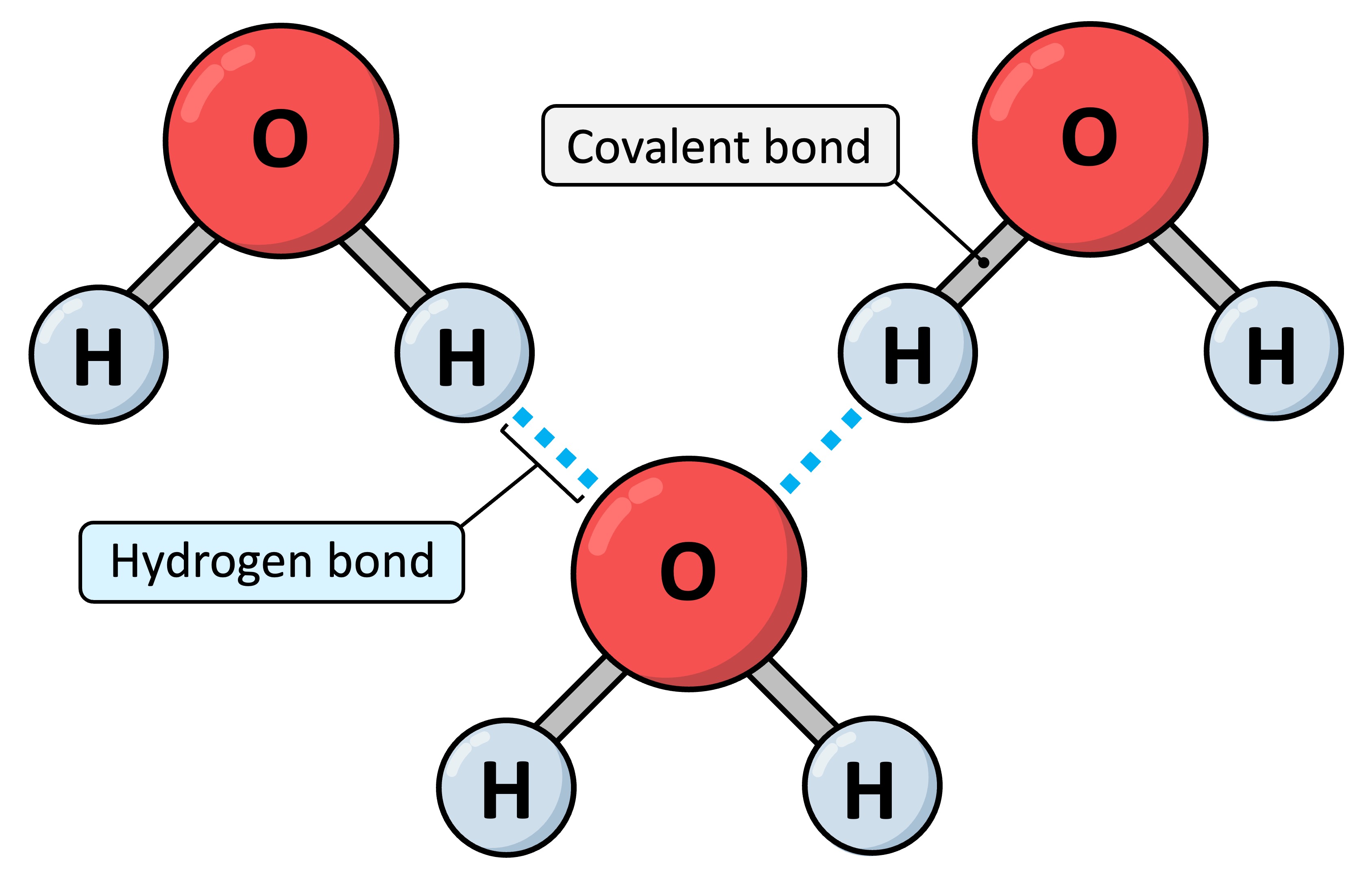
Now, these hydrogen bonds aren't like the strong, glued-together bonds that hold atoms within a molecule. Nope, they're more like a gentle, electrostatic attraction. Imagine two people holding hands – not a super tight grip, but a nice, comfy connection. That's kind of what a hydrogen bond is like. It happens when a hydrogen atom is linked to a super electronegative atom, like oxygen (which methanol has plenty of!). This makes the hydrogen a little bit positive, and the oxygen a little bit negative.
Must Read
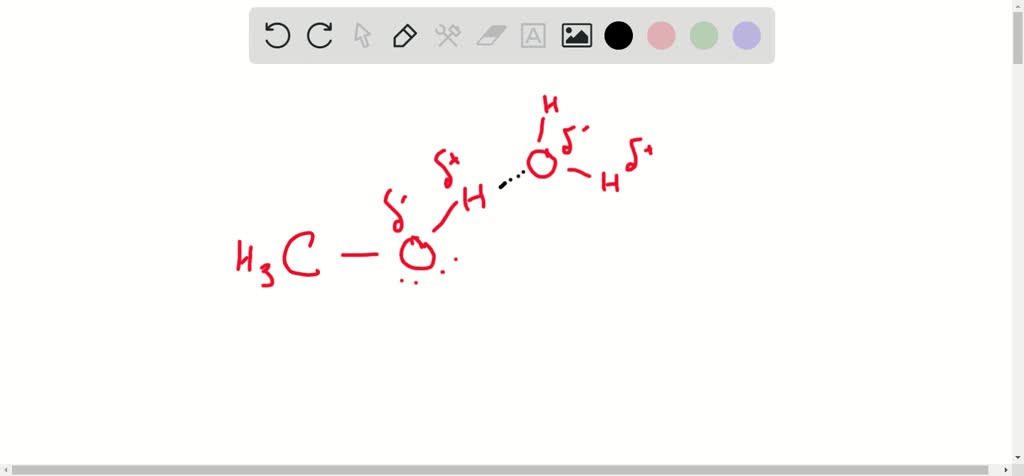
And here's where methanol really shines: it can actually form two of these awesome hydrogen bonds! Yep, you heard that right, two! It’s like methanol has two hands ready to hold on. One connection happens when the oxygen atom in methanol reaches out and grabs onto a hydrogen atom from another molecule. Think of it as methanol giving a hug.
But wait, there's more! The other hydrogen bond happens when the hydrogen atom attached to methanol's oxygen atom reaches out and is attracted to the oxygen atom of another methanol molecule. So, it’s like methanol can both give and receive these little molecular hugs. It’s a real team player!

Why is this so exciting? Because these simple little connections have a huge impact on how methanol behaves. For starters, it's why methanol is a liquid at room temperature. If it couldn't form these hydrogen bonds, it would be a gas, floating around invisibly! Thanks to these connections, methanol molecules like to stick together, making it a liquid we can actually work with and study. It’s like they’re all holding hands in a big, friendly circle.
This ability to form two hydrogen bonds is what makes methanol a bit of a superstar in the world of small molecules. It’s not too complicated, but it’s got just enough oomph to create these really important interactions. It's like a simple recipe that creates a delicious outcome. You have the basic ingredients, and then a little bit of special technique (the hydrogen bonding!) makes all the difference.

Think about it: water, another super common liquid, is famous for its hydrogen bonding. But methanol, with its simple structure and dual hydrogen bonding capability, offers a slightly different flavor of intermolecular fun. It’s like comparing two delicious desserts – both great, but each with its own unique charm. Methanol’s charm lies in its straightforward ability to engage in these connections.
So, when you hear about methanol, you can now imagine this little molecule actively participating in these invisible connections. It’s not just sitting there; it’s reaching out, holding hands, and influencing its surroundings. It’s like watching a tiny dancer gracefully twirling and interacting with its partners on the dance floor.
The fact that methanol can form two hydrogen bonds makes it a particularly good solvent for certain things. That means it can dissolve other substances really well. This is because it can effectively surround and interact with other molecules, pulling them into solution. It's like having a friendly, helpful assistant who can hold hands with many different things and help them get where they need to go.

It’s also fascinating to think about how these simple bonds contribute to methanol’s uses. In industry, it's used in everything from antifreeze to a fuel source. And in the lab, chemists rely on its properties, which are largely dictated by these very hydrogen bonds. It’s a testament to how fundamental, even tiny, molecular interactions can have big, real-world consequences.
So, the next time you encounter methanol, whether it’s in a science textbook or a product you use, remember its secret superpower: the ability to form two hydrogen bonds. It’s this simple, yet elegant, feature that makes methanol so interesting and so useful. It’s a little molecule with a big capacity for connection, and that’s pretty darn cool if you ask me!

It's a reminder that even the smallest things in the universe have complex and beautiful ways of interacting. And methanol, with its dual hydrogen bonding ability, is a perfect example of this. It’s a little piece of chemistry that’s both accessible and endlessly intriguing.
Go ahead, give it a thought. How many hydrogen bonds can methanol form? The answer is a delightful two, and that little detail unlocks a world of fascinating properties.
It’s these kinds of insights that make exploring the world of chemistry so entertaining. You start with a simple question, and you uncover a whole network of interconnected phenomena. Methanol’s hydrogen bonding is a prime example of this; it’s a gateway to understanding so much more about how molecules behave and interact. So, keep that curiosity buzzing!