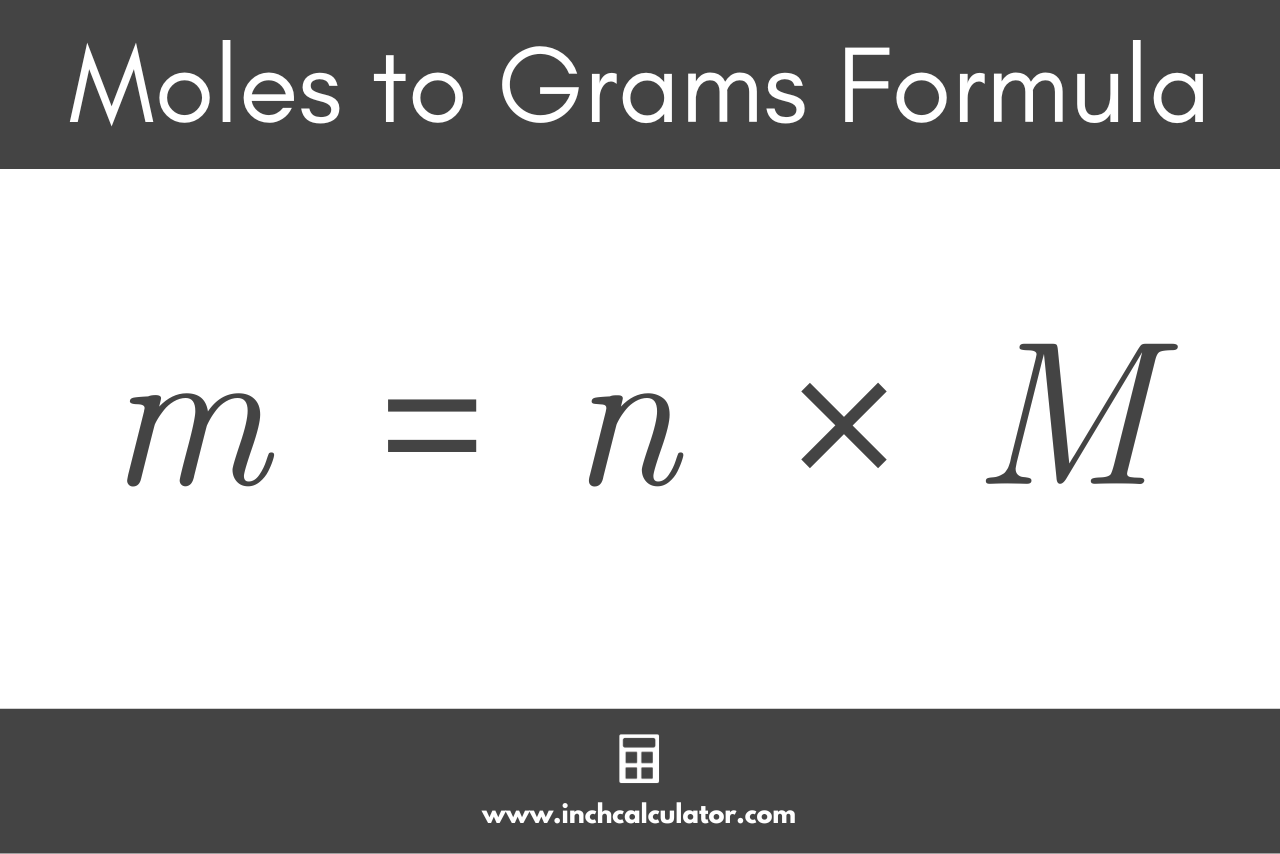How Do You Convert Moles Into Grams

Ever felt like you were staring at a science problem that looked like a secret code? You know, all those 'moles' and 'grams' floating around? Well, buckle up, because converting between these two is less of a brain-buster and more of a delightful little puzzle. Think of it like switching between two different languages, but with a super handy translator.
And what's the magic translator, you ask? It's a concept so elegant, so simple, it’s almost mischievous. It’s called the molar mass. This isn't just some stuffy chemistry term; it's your golden ticket.
Imagine you have a bunch of LEGO bricks. A mole is like saying you have a specific number of those LEGO bricks – a really, really, really big number, to be exact. Grams, on the other hand, are just how much those LEGO bricks weigh. So, converting between moles and grams is like figuring out the weight of a giant pile of LEGOs when you know how many you have.
Must Read
The real fun begins when you realize this isn't just about numbers. It’s about understanding the hidden weight of tiny, invisible things. It’s like having a superpower to know how much a specific amount of anything chemical weighs. Pretty neat, right?
Let's dive into this adventure! Your first stop on this exciting journey is finding your trusty sidekick, the periodic table. This chart, often seen as a bit intimidating, is actually your treasure map. It holds all the secrets to the universe's building blocks.
Each element on that periodic table has a little number associated with it. This number is incredibly important. It’s not just a random digit; it tells you something profound about that element.
This number is called the atomic mass. It represents the average mass of a single atom of that element. Think of it as the weight of one single, perfect LEGO brick of that type.
Now, here's where the magic really starts to sparkle. A mole of any substance is Avogadro's number of particles – that's about 6.022 x 10^23. Don't let that big number scare you; it just means a HUGE amount.

When we talk about a mole of a compound (that’s when two or more elements team up), we’re talking about Avogadro’s number of those molecules. And to find the weight of that giant collection, we use those atomic masses we found on the periodic table. It’s like building a LEGO castle and knowing the weight of each type of brick.
So, how do you get the molar mass of a compound? You simply add up the atomic masses of all the atoms in that compound. If your compound is water (H₂O), you take the atomic mass of hydrogen, double it (because there are two hydrogens), and then add the atomic mass of oxygen. Voilà! You have the molar mass.
This molar mass is your key. It’s the conversion factor, the bridge that connects moles and grams. It tells you how many grams are in one mole of that specific substance. It's like knowing that one bag of red LEGOs weighs 50 grams.
Now, let’s get to the fun part: the actual conversion. Suppose you have a certain number of moles of a substance, and you want to know how many grams that is. It’s like having 3 bags of those red LEGOs and wanting to know the total weight.
You take the number of moles you have. Then, you multiply it by the molar mass of that substance. It’s a straightforward multiplication, like multiplying 3 bags by 50 grams per bag.

The units themselves are your guide here. If you have moles and you multiply by (grams/mole), the 'moles' cancel out, leaving you with 'grams'. It’s like the numbers guiding you through the process.
And what if you have grams and want to find out how many moles you have? It’s just the reverse operation! You take the number of grams you have and divide it by the molar mass. It's like knowing the total weight of your LEGOs and wanting to figure out how many bags you have.
This is where the "aha!" moments happen. You start to see how these abstract concepts have real-world applications. It’s not just for lab coats and beakers; it’s a way to quantify and understand the physical world around us.
Think about baking. Recipes often call for ingredients in grams, but chemists often work in moles because it represents a specific number of molecules. Being able to switch between these is like having a universal recipe translator for the chemical kitchen.
The beauty of this conversion is its universality. It applies to everything in chemistry. Whether you're dealing with a simple element like iron or a complex molecule like DNA, the principle remains the same. It’s a fundamental law of chemical measurement.
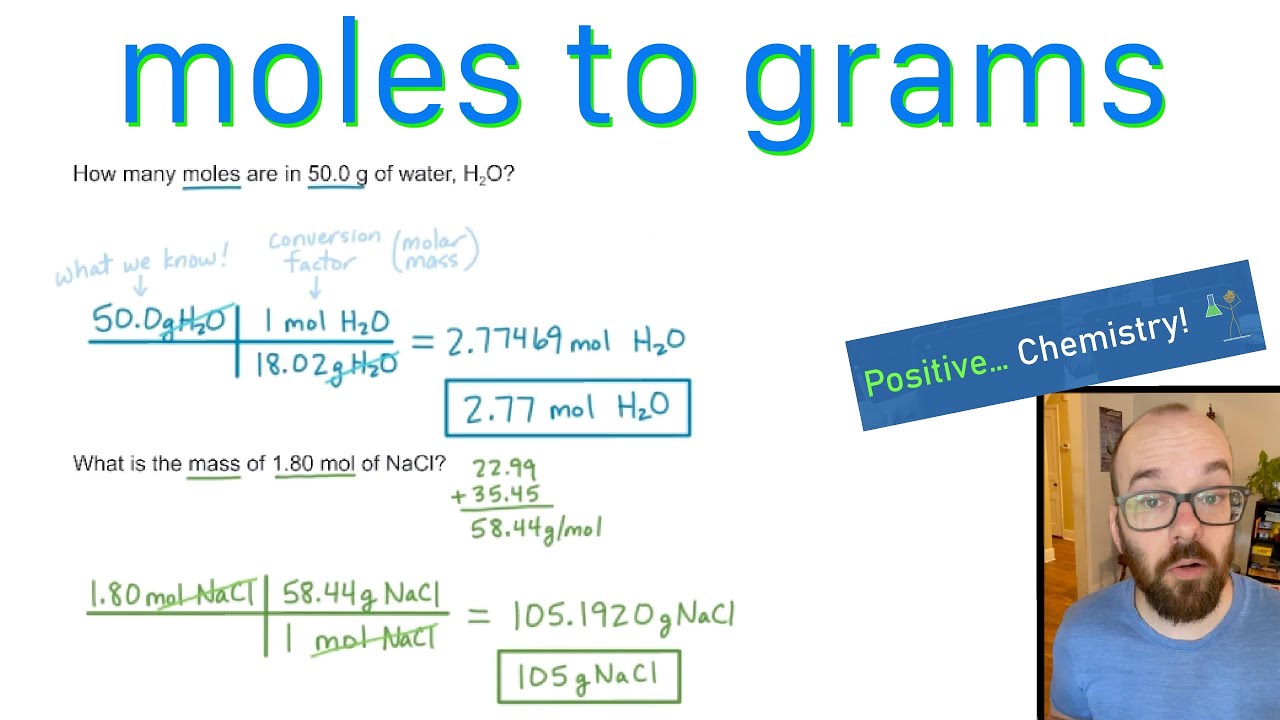
And the more you practice, the more intuitive it becomes. You start recognizing common molar masses, and the process feels less like a calculation and more like second nature. It’s like learning to ride a bike; at first, it's wobbly, but soon you're cruising.
So, why is this whole mole-to-gram conversion so entertaining? Because it demystifies a fundamental aspect of chemistry. It takes something that looks complicated and breaks it down into a logical, step-by-step process. It’s a tiny victory every time you solve one.
What makes it special? It's the power it gives you. You're no longer just looking at numbers; you're understanding the weight of matter at a molecular level. You're unlocking the secrets of how much stuff you actually have.
Imagine a chemist in a lab, carefully measuring out a precise amount of a substance. They're not just guessing; they're using the principles of molar mass and mole-to-gram conversion to ensure accuracy. It’s a quiet act of scientific precision.
And you can do it too! With a periodic table and a bit of practice, you can master this skill. It opens up a whole new way of looking at the world, from the air you breathe to the food you eat.

So, next time you see those terms, don't shy away. Embrace the challenge! Think of it as a fun game of chemical translation. The molar mass is your cheat sheet, and the periodic table is your guide.
It's a journey of understanding the relationship between the invisible world of molecules and the tangible world of weight. It’s about connecting the dots in a way that’s both logical and surprisingly delightful.
This isn't just about passing a test; it's about gaining a deeper appreciation for the fundamental building blocks of our universe. It’s about wielding the language of chemistry with confidence.
So, go ahead, grab a periodic table. Pick a compound. And start converting! You might just find yourself hooked on the elegant simplicity of turning moles into grams. It’s a small skill with a surprisingly big impact.
The journey of a thousand miles begins with a single step, and the journey of understanding chemical quantities begins with mastering the conversion between moles and grams.
It’s a fundamental skill, yes, but it’s also a gateway to understanding so much more. It’s the first step in a much larger, more fascinating chemical adventure. And who knows where that adventure might lead you? Perhaps to discovering new medicines, or understanding climate change, or even just making a perfect batch of cookies.
The world of chemistry is waiting, and the ability to convert moles to grams is your invitation. It’s a simple, yet powerful tool that unlocks a deeper understanding of the matter that surrounds us. So, step right up and give it a try!