How Do You Calculate Relative Formula Mass

Ever found yourself staring at a recipe, completely baffled by those little numbers next to the ingredients? You know, the ones that look like they belong in a secret spy code? Well, my friends, you're not alone! We're about to demystify the magical world of calculating relative formula mass. Think of it as figuring out the true weight of your ingredients, not just by cup or by spoon, but by the actual building blocks that make them up. It’s like knowing how much your superhero team weighs, not just by their capes, but by the individual muscles, gadgets, and sheer awesomeness of each member.
Let's be honest, the word "mass" can sound a bit… heavy. And "relative formula" sounds like something you'd only find in a dusty old chemistry textbook that smells vaguely of forgotten experiments. But trust me, it's way less intimidating than it sounds. It's basically a fancy way of saying "how much 'stuff' is in something," but in a way that’s super useful, especially when you’re trying to, say, bake the perfect loaf of bread or mix the right amount of paint for your masterpiece living room.
Imagine you’re trying to assemble some IKEA furniture. You get a big box, and it’s got a bunch of pieces. You can’t just guess how much the whole thing will weigh, right? You need to know how much each screw, each wooden plank, and each mysterious Allen key contributes to the final structure. That’s kind of what relative formula mass does for molecules. It helps us understand the individual contributions of each atom to the overall "weight" of the compound.
Must Read
So, how do we actually do this? It all starts with the periodic table. This isn't just a pretty chart with a bunch of symbols and numbers. It's like the ultimate cheat sheet for atoms. Each element on there has an atomic number (that’s the smaller number, usually on top) and an atomic mass (that’s the bigger number, usually below). The atomic mass is our key player here. Think of it as the "weight" of a single atom of that element. For simplicity, we often round these numbers to the nearest whole number. Nobody’s got time for decimals when they’re trying to figure out if they have enough flour for cookies!
Let’s take a simple example. Water. H₂O. We all know water, right? It’s the stuff that comes out of the tap, the essential ingredient for tea, coffee, and questionable late-night snack concoctions. To find the relative formula mass of water, we need to know the atomic masses of hydrogen (H) and oxygen (O).
If you peek at your trusty periodic table, you’ll find that hydrogen has an atomic mass of approximately 1. And oxygen? That’s about 16. Easy enough so far, right? It’s like knowing that one Lego brick weighs 1 gram and a bigger, fancier Lego brick weighs 16 grams.
Now, here’s where the "formula" part comes in. Look at the chemical formula for water: H₂O. That little ‘2’ next to the H tells us there are two hydrogen atoms in every molecule of water. The ‘O’ has no number, which means there’s just one oxygen atom. It’s like saying you need two small strawberries for your smoothie and one big banana.

So, to calculate the relative formula mass of water, we add up the masses of all the atoms in the formula. We have two hydrogens, each weighing about 1, and one oxygen, weighing about 16. So, it’s (2 * 1) + (1 * 16) = 2 + 16 = 18. Voila! The relative formula mass of water is approximately 18. Pretty neat, huh? It’s like the total weight of your strawberry-banana smoothie ingredients before you even blend them.
Let’s try another one. Common table salt. Sodium chloride. NaCl. No little numbers here, just one sodium (Na) and one chlorine (Cl). Your periodic table will tell you that sodium has an atomic mass of about 23, and chlorine has an atomic mass of about 35.5. Again, we can often round these for easier calculations, so let’s say sodium is 23 and chlorine is 35.5 (some might round to 36, it depends on the level of precision you need, just like how some recipes are super precise and others are "a pinch of this").
So, for NaCl, it’s (1 * 23) + (1 * 35.5) = 23 + 35.5 = 58.5. The relative formula mass of sodium chloride is about 58.5. This means that, in terms of its fundamental building blocks, a unit of salt is roughly 58.5 times "heavier" than a single unit of hydrogen. It's like comparing the weight of a small pebble to a slightly bigger rock.
What happens when there are more atoms or different types of atoms? No sweat! We just keep adding. Let’s take carbon dioxide, the stuff we breathe out and that makes fizzy drinks so… fizzy. CO₂. We have one carbon (C) and two oxygens (O).

Carbon’s atomic mass is about 12. Oxygen, as we know, is about 16. So, for CO₂, it’s (1 * 12) + (2 * 16) = 12 + 32 = 44. The relative formula mass of carbon dioxide is about 44. This means that the collective "weight" of the atoms in a CO₂ molecule is about 44 times the "weight" of a single hydrogen atom.
Sometimes, you’ll see parentheses in chemical formulas. For example, calcium hydroxide is Ca(OH)₂. This is where things can get a little trickier, but it’s still just as straightforward once you get the hang of it. The parentheses mean that everything inside the parentheses is repeated as many times as indicated by the subscript outside. So, (OH)₂ means you have two lots of OH. In other words, two oxygen atoms and two hydrogen atoms.
Let’s break down Ca(OH)₂. Calcium (Ca) has an atomic mass of about 40. Oxygen (O) is about 16. Hydrogen (H) is about 1.
So, we have one calcium atom: 1 * 40 = 40.
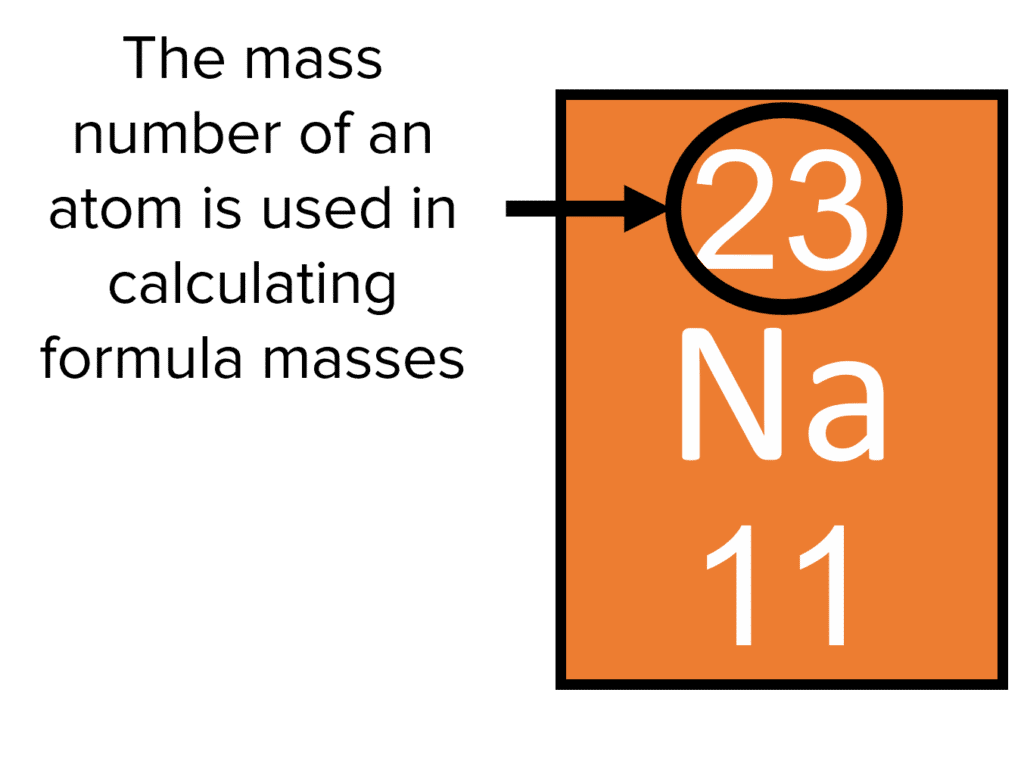
Then, we have the (OH)₂. This means we have two oxygens (2 * 16 = 32) and two hydrogens (2 * 1 = 2). The total from the hydroxide part is 32 + 2 = 34.
Now, add it all up: 40 (calcium) + 34 (two hydroxides) = 74. The relative formula mass of calcium hydroxide is approximately 74.
It’s like making a sandwich. If you have one slice of bread (calcium), and then you have two layers of your favourite filling (OH), you count all the ingredients. Two layers of filling means double the amount of filling!
Why is this even useful, you ask? Well, imagine you’re a baker. You need to make a cake. The recipe calls for 100 grams of flour and 50 grams of sugar. But what if you want to understand how many molecules of flour and sugar you’re actually using? This is where relative formula mass comes in handy. It’s the first step towards understanding the mole concept, which is like a chemist's way of counting things in bulk. Instead of counting individual grains of sugar (which would take forever!), we use moles. And to get to moles, we need relative formula mass. It’s the bridge between the microscopic world of atoms and the macroscopic world of grams and recipes.

Think of it as planning a party. You know you need 50 balloons and 20 streamers. But if you want to know the total "party supply weight" to estimate shipping costs, you need to know the weight of each individual balloon and streamer. Relative formula mass is like figuring out the weight of one complete balloon (one molecule) or one complete streamer (another molecule).
It’s also crucial in manufacturing. If a company is making, say, fertilizer, they need to know exactly how much of each ingredient to mix to get the desired chemical compound with the right "strength." They can’t just wing it. They use relative formula masses to precisely measure out their components. It's like a chef meticulously measuring spices – a little too much of one thing, and your delicious curry might turn into a fiery inferno!
The beauty of relative formula mass is that it’s a universal constant. A molecule of water will always have a relative formula mass of around 18, no matter if it’s in your teacup, in the ocean, or in outer space. This consistency is what makes chemistry so powerful and predictable.
So, the next time you see a chemical formula, don’t run for the hills! Grab your periodic table, get your calculator (or your trusty fingers for simple sums!), and start adding up those atomic masses. You'll be a relative formula mass calculating pro in no time. It’s a little bit of detective work, a dash of arithmetic, and a whole lot of understanding about the building blocks of everything around us. And who knows, maybe this new skill will even help you win a science quiz or impress your friends at your next dinner party with fascinating tidbits about molecules. Cheers to unraveling the mysteries of the universe, one atom at a time!