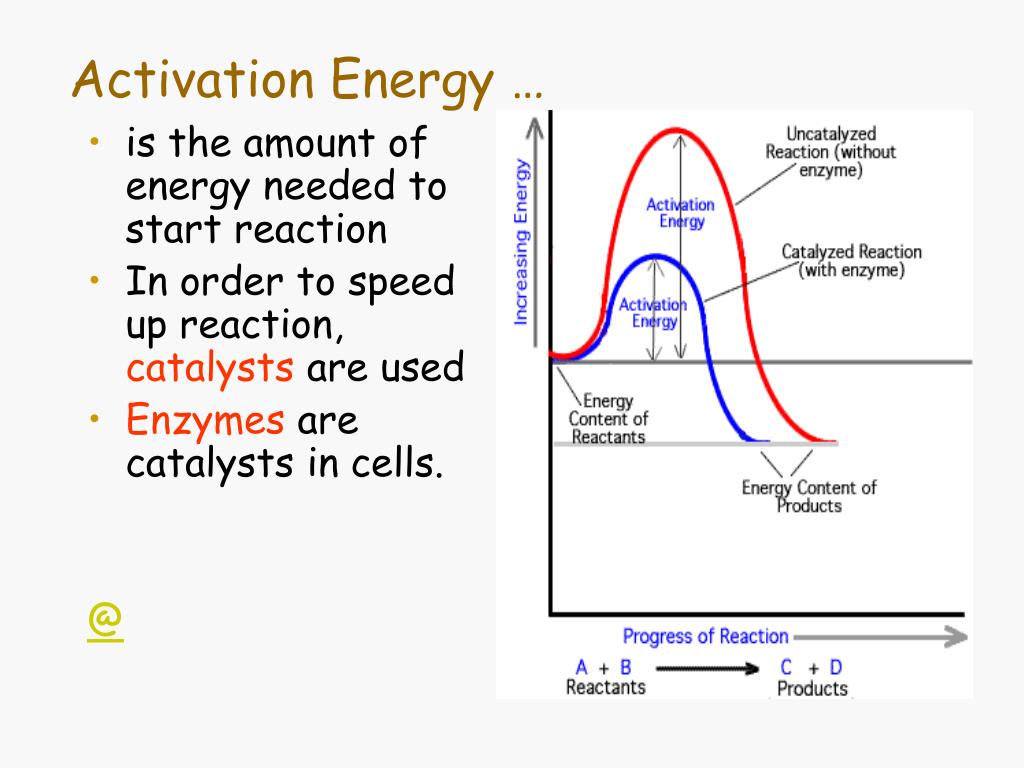
How Do Enzymes Reduce The Activation Energy

So, imagine you're trying to bake a cake. You've got all your ingredients laid out – flour, sugar, eggs, the whole shebang. Now, the recipe says you need to mix them all together. Easy enough, right? But here's the thing: if you just dumped everything in a bowl and stared at it, it's not exactly going to magically turn into a delicious cake. You need to do something. You need to apply energy, right? You've got to grab that whisk (or mixer, if you're feeling fancy) and start swirling. That initial effort, that 'get-things-going' push, is kind of like what scientists call the activation energy.
Think of it as a little hill your ingredients have to climb before they can really get down to business and become a cake. Without a good shove, they're just going to sit there, looking decidedly un-cake-like.
Now, what if I told you there's a way to make that cake-baking process go way faster, with less effort? Like, imagine you had a tiny, super-efficient baking assistant who knew exactly what to do with your ingredients, guiding them over that little hill with minimal fuss. That, my friends, is essentially what enzymes do in our bodies. Pretty neat, huh?
Must Read
The Mysterious Hill of Activation Energy
Okay, so let's get a little more science-y, but don't worry, we'll keep it light. In the world of chemistry, reactions don't just happen because you want them to. Molecules have to bump into each other, they have to collide with enough force, and they have to be in the right orientation. It's like trying to get two incredibly shy teenagers to high-five. They might be in the same room, but getting them to actually connect? That takes a bit of encouragement!
This "encouragement" is our friend, the activation energy. It's the minimum amount of energy required to get a chemical reaction started. It’s the initial spark that kicks off the transformation. Without it, most reactions would just fizzle out before they even began. And our bodies? Well, our bodies are a constant whirlwind of chemical reactions. From digesting your breakfast to building new muscle cells, it's all happening at the molecular level.
If every single one of these reactions needed a huge amount of activation energy, we’d be in a bit of a pickle. Imagine needing to build up a massive amount of energy just to, say, blink. That sounds exhausting, doesn't it? Thankfully, our bodies are far too smart (and efficient!) for that.
Enter the Enzyme: The Ultimate Reaction Facilitator
So, how do enzymes swoop in and save the day? It’s all about lowering that activation energy. Think of it like this: instead of having to hike up a steep mountain, the enzyme creates a much gentler, more accessible path. It’s like finding a secret tunnel that bypasses the most challenging part of the climb.

But how does it do that? This is where it gets really interesting, and honestly, a little bit magical. Enzymes are typically proteins, and they have these really specific three-dimensional shapes. Inside that shape, there's a special little spot called the active site. This active site is like a perfectly tailored glove, designed to fit a specific molecule, or a group of molecules, called the substrate.
It's not a random meeting. The substrate finds its way to the enzyme, and when it slots into the active site, something special happens. The enzyme basically embraces the substrate, holding it in a way that makes it much easier for the reaction to occur.
The Enzyme-Substrate Dance: A Closer Look
Let’s break down the actual mechanics of this "dance." When the substrate binds to the active site, the enzyme can do a few key things to lower the activation energy:
1. It Strains the Substrate: Imagine you have a stubborn piece of LEGO that needs to be snapped together with another. If you just try to force it, it might be difficult. But what if you could slightly bend or twist the LEGO piece just a little bit? That would make it much easier to click into place. The enzyme, by binding to the substrate, can induce this kind of strain. It contorts the substrate into a shape that’s closer to the transition state – the unstable, high-energy intermediate that needs to form before the reaction can complete.
2. It Provides a Favorable Microenvironment: Sometimes, the conditions needed for a reaction to happen are a bit… harsh. Maybe it needs a specific pH, or it needs to be shielded from water. The active site of an enzyme can create just the right conditions. It's like having a cozy, climate-controlled room where the reaction can happen without any unwanted interference. This can involve adjusting the acidity, polarity, or even bringing reactive groups closer together.

3. It Orients the Reactants Correctly: Remember those shy teenagers trying to high-five? If they’re not facing each other, it’s not going to happen. Enzymes are brilliant at bringing the substrate molecules (or parts of a single substrate molecule) together in exactly the right way. They hold them in a precise orientation so that the reactive parts are perfectly aligned for a collision that leads to product formation. It’s like having a perfect handshake facilitator.
4. It Directly Participates in the Reaction (Temporarily!): In some cases, the enzyme might even get a little hands-on. It can form temporary chemical bonds with the substrate, acting as a kind of "helper molecule" that facilitates the bond-breaking and bond-forming processes. Once the reaction is complete, these temporary bonds are broken, and the enzyme is released, ready to go again. It’s like the helper quickly hands off the finished product and steps back, job done.
The "Lock and Key" (and Sometimes "Induced Fit") Analogy
You might have heard of the lock and key analogy for enzymes. The enzyme is the lock, and the substrate is the key. Only the right key fits into the right lock. This is a good starting point, but it's not the whole story. It implies a very rigid fit.
A more accurate, and I think, cooler analogy is the induced fit model. Imagine a glove. When you put your hand in it, the glove isn't just a static shape waiting for your hand. It actually molds around your hand, conforming to its specific contours. Similarly, when the substrate enters the active site, it can cause a slight change in the enzyme's shape. This subtle shift is what helps to optimize the binding and strains the substrate, setting the stage for the reaction.
So, it’s not just a pre-set keyhole; it's more like the enzyme slightly adjusts its shape to get the perfect grip on the substrate. This flexibility is crucial for its efficiency!

Why This Matters: The Speed of Life
So, why is all this lowering of activation energy so important for us? Because life, as we know it, would simply not be possible without it. Our cells are constantly performing thousands of chemical reactions every second. If each of these reactions had a high activation energy, our bodies would be incredibly slow, inefficient, and frankly, wouldn't be able to keep up with the demands of survival.
Think about digestion. When you eat, your body needs to break down complex molecules like carbohydrates, proteins, and fats into simpler ones that can be absorbed. Enzymes like amylase (for carbs), proteases (for proteins), and lipases (for fats) are the workhorses here. They massively speed up the breakdown process. Without them, digesting a single meal could take days, or even weeks!
Or consider metabolism – the entire process of how your body extracts energy from food. This involves a long, intricate series of reactions, each catalyzed by specific enzymes. If these enzymes weren't doing their job of lowering activation energy, you'd basically have no energy. Not exactly conducive to running, jumping, or even just thinking.
Even things like muscle contraction, nerve signaling, and DNA replication rely heavily on enzyme activity. They are the unsung heroes, working tirelessly behind the scenes to keep everything running smoothly.
Enzyme Specificity: Not Just Any Old Enzyme Will Do
One of the other amazing things about enzymes is their specificity. An enzyme that breaks down carbohydrates won't usually be found messing around with proteins. This is because the shape of the active site is so specific to its substrate. It's like having a specialized tool for every single job. You wouldn't use a hammer to screw in a screw, right? Same idea!
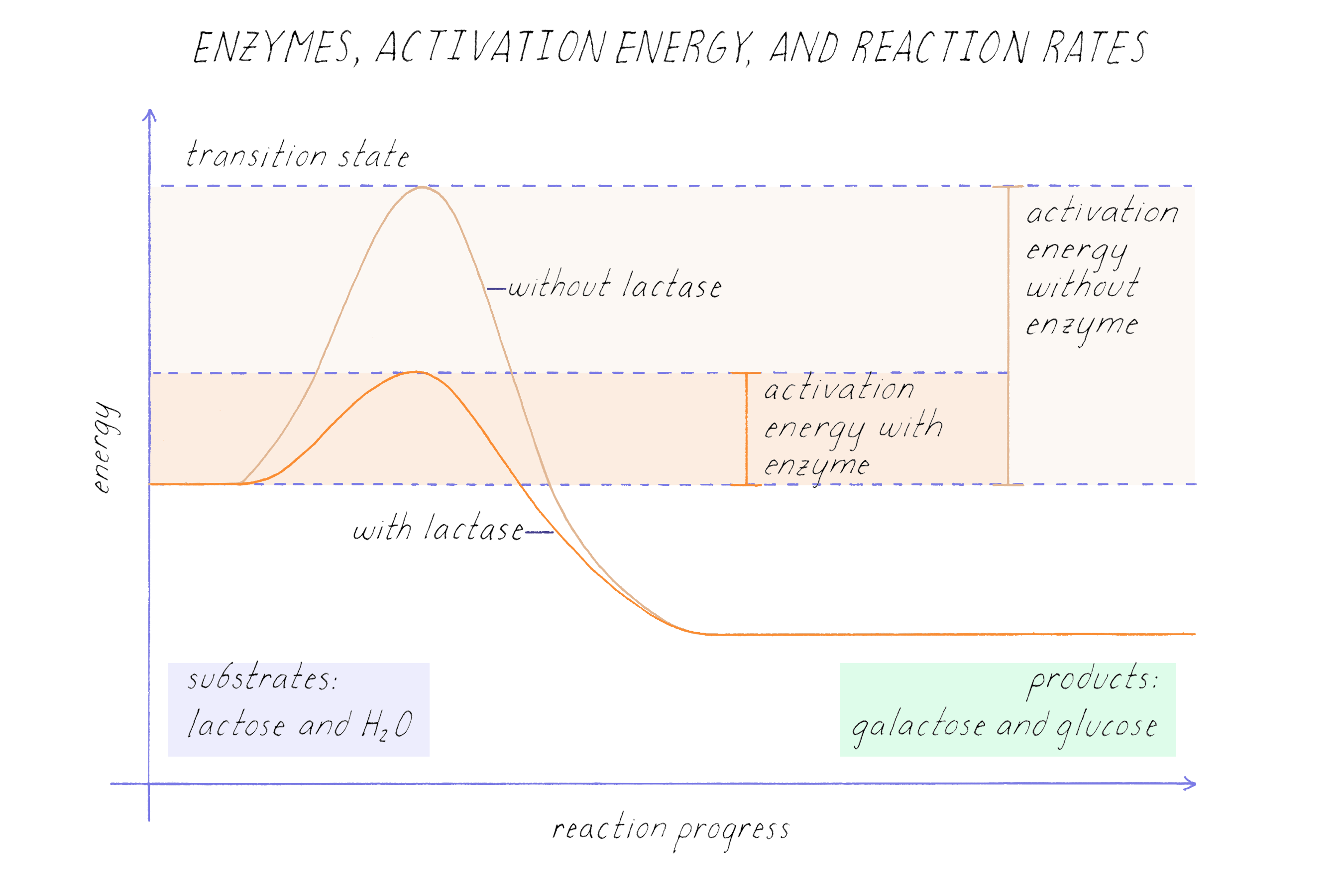
This specificity is crucial for maintaining order in our cells. Imagine the chaos if every enzyme could act on every molecule! It would be a biochemical free-for-all.
What Happens When Enzymes Go Rogue (or Don't Show Up)?
Sometimes, things go wrong with enzymes. Genetic mutations can lead to enzymes that are malformed and can't bind to their substrates properly, or they might be completely inactive. This can have serious consequences. Diseases like phenylketonuria (PKU) are caused by a deficiency in an enzyme needed to break down an amino acid. Without this enzyme, the amino acid builds up to toxic levels.
Conversely, sometimes enzymes are too active, or their regulation is off, which can also lead to problems. So, the precise control and functioning of enzymes are absolutely vital for health.
In Summary: The Unsung Catalysts of Life
So, the next time you marvel at how quickly your body processes food, how your muscles move, or how your brain thinks, spare a thought for the incredible world of enzymes. These remarkable molecules are the masters of lowering activation energy, creating efficient pathways for the countless chemical reactions that underpin life itself. They are the unsung catalysts, the tiny biological machines that make it all happen, without which, well, we wouldn't be here!
It’s a beautiful illustration of how even the most complex processes can be orchestrated by nature's genius for efficiency. They take a difficult, energy-intensive task and make it… well, doable. And for that, I think we can all be pretty grateful!