Ground State Electron Configuration Of Pb

Hey there, ever find yourself staring at a humble pencil, or maybe that shiny piece of jewelry, and wonder what's really going on inside? It's easy to just see the object, right? But deep down, at the tiniest, most fundamental level, everything is made of atoms. And those atoms have electrons, zipping around the nucleus like tiny, energetic bees. Today, we're going to have a little chat about the ground state electron configuration of lead – sounds fancy, doesn't it? But stick with me, because it's actually pretty cool, and surprisingly relevant to our everyday lives!
Think of electrons like little kids in a playground. They love to hang out in specific spots, or "energy levels," around the atom's center (the nucleus). The "ground state" just means they're in their most chill, lowest-energy positions. They're not all hyped up and bouncing off the walls; they're just settled in, like you after a big Sunday dinner, perfectly content. No need for extra energy to get them going!
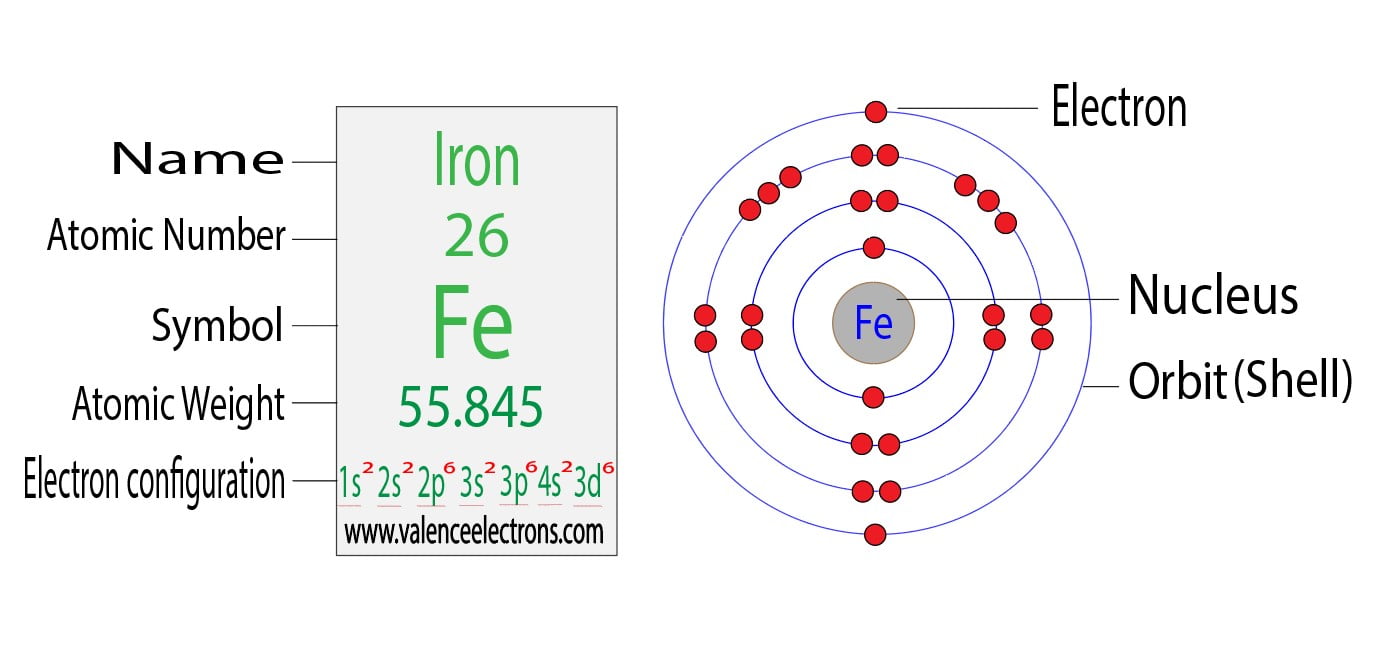
Now, lead – that's symbol Pb on the periodic table, if you ever spy it. You might know it from old plumbing (which we thankfully don't do anymore!) or as a component in some batteries. It's a pretty substantial element, a bit of a heavyweight in the atomic world. And like anything substantial, it has a good number of these electron-kids to sort into their playground spots.
Must Read
Imagine you're organizing a party. You have different rooms for different activities. Some rooms are super popular and fill up first (like the "snack table" room), while others are a bit quieter or require more effort to get to (like the "quiet reading" room). Electrons are a bit like that. They fill up the lower energy levels first, the ones closest to the nucleus, because that's where they're most comfortable and have the least energy.
For lead, this means its electrons are arranged in a very specific pattern. It's like a well-defined seating chart for a very important meeting. We use little shorthand notations to describe these arrangements, like saying someone is in "Room 1, Seat 3." The key thing to remember is that each "room" (or orbital) can only hold a certain number of electrons, kind of like how a small car can only fit so many people.
So, why should you care about lead's electron configuration? Well, it's the secret sauce that dictates how lead behaves! It determines if lead likes to bond with other elements, if it's a good conductor of electricity, and even its physical properties, like how dense or malleable it is. Think of it like a person's personality. Their DNA (their "electron configuration") shapes who they are, how they interact with others, and what they're good at.
Let's break it down a tiny bit, without getting too bogged down in jargon. The electrons in lead fill up several "shells" or energy levels. The innermost shells are like the cozy, well-worn parts of your house – they're filled first. As you move outwards, the shells get a bit bigger and can hold more electrons. Lead has quite a few electrons, so it has a lot of these shells and sub-shells to fill.

Think about building a Lego castle. You start with the base, adding bricks layer by layer. The ground state electron configuration is like that carefully constructed base. The electrons are placed in the most stable, lowest-energy spots available. They don't just randomly float around; they follow specific rules, like not overcrowding any single "room."
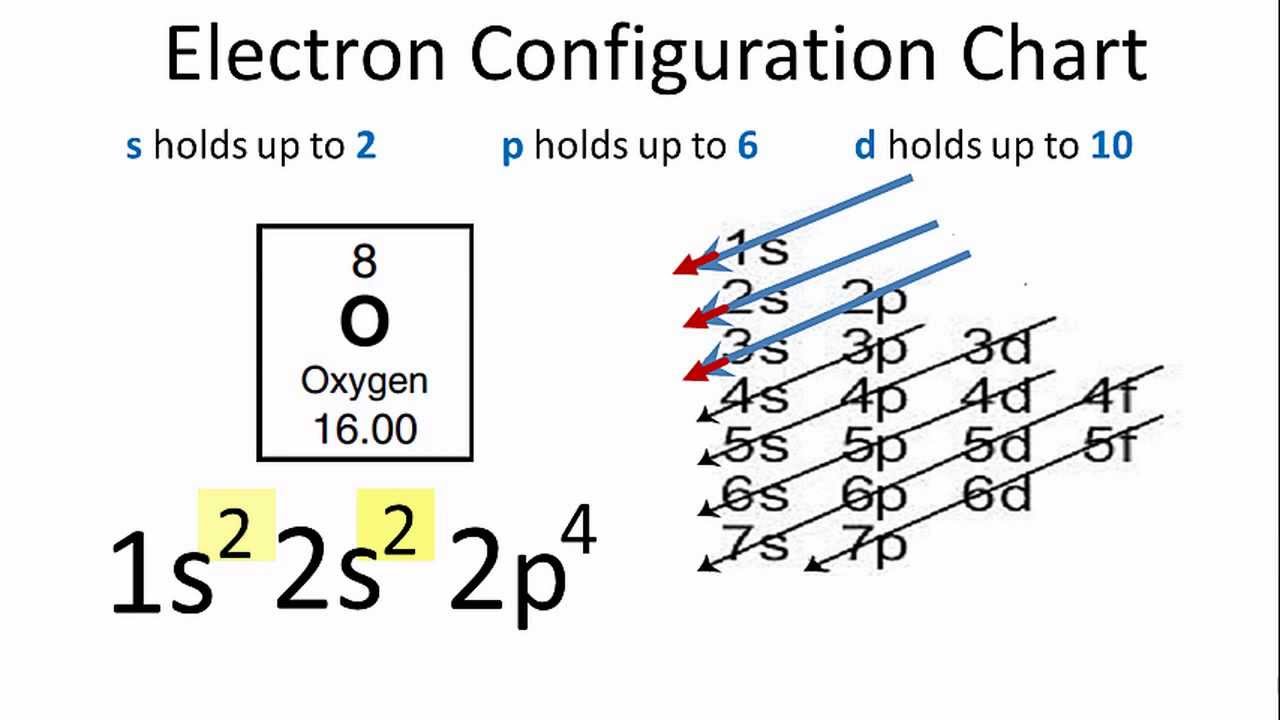
Lead's electron configuration is often written as: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶ 6s² 4f¹⁴ 5d¹⁰ 6p². Now, don't let those numbers and letters scare you! Each part tells us about a specific "room" and how many electrons are chilling in it. The 's', 'p', 'd', and 'f' are like different types of rooms on the playground – some are round, some are shaped differently, and they have different capacities.
The 6s² electrons are like the two most pampered pets in a household, always having the best spot. The 4f¹⁴ and 5d¹⁰ electrons are in deeper, more spacious rooms, holding a good number of companions. And finally, the 6p² electrons are in the outermost "rooms," ready for some action. These outer electrons are the ones that really get to interact with the outside world, like the kids on the swings or the slide, engaging with others.

So, why is this outer shell, the 6p² in lead's case, so important? Because it's the part of the atom that's involved in chemical reactions! It's like the front porch of a house. It's the first thing people see and interact with. If lead wants to make friends with other atoms (form chemical bonds), it's these 6p² electrons that will do the heavy lifting.
For example, if lead has these two electrons in its outermost 'p' orbital, it might be inclined to share them or even give them away to form certain types of compounds. This is how we get all the amazing materials and substances around us. The way these electrons are arranged dictates whether lead will be used in a sturdy alloy for building, or perhaps in some specialized electronic component.

Think of it like a chef preparing a dish. The ingredients are the atoms, and their electron configurations are like the inherent properties of those ingredients. A chef knows that sugar behaves differently than salt. This difference in behavior stems from the fundamental structure of their atoms, including their electron configurations. Lead's unique electron arrangement gives it its unique "flavor" in the chemical kitchen.
Understanding the ground state electron configuration of lead helps scientists and engineers predict and control how lead will behave. It's like knowing a person's strengths and weaknesses; you can then figure out the best jobs for them or how to best help them. This knowledge is crucial for developing new technologies, understanding environmental processes, and even creating safer materials.
So, next time you encounter lead, whether it's in a history book about old pipes or in a modern gadget, remember that its story is written in the quiet, orderly dance of its electrons. Their ground state configuration is the fundamental blueprint that makes lead, well, lead. It’s a fascinating glimpse into the microscopic world that shapes our macroscopic reality, all in a wonderfully chill, no-fuss way!