Give The Hybridization For The C In C2f2.
Hey there, science adventurers and curious minds! Today, we're diving headfirst into a super cool, albeit slightly wiggly, molecule called C2F2. Don't let the numbers and letters scare you; it's like a tiny LEGO set that's just begging to be understood. We're going to uncover the secret handshake of the carbon atoms in this little guy.
Imagine you've got two best buddies, let's call them Carbon Charlie and Carbon Carl. They're always looking for new ways to hang out and share their toys. In C2F2, these two carbon pals are the stars of the show, and they've got some fluorine friends, F for fantastic, tagging along.
Now, when atoms get together to form molecules, they don't just stand there awkwardly. Oh no, they get all excited and start rearranging their little electron buddies to form bonds. This is where the magic, or rather, the hybridization, happens!
Must Read
Think of hybridization as atoms deciding to put on different hats before a party. They take their original, perfectly normal electron "orbitals" (which are basically just fancy word for where their electrons hang out) and mix them up to create brand new, super-duper orbitals. It's like taking a plain vanilla ice cream and swirling in some chocolate and strawberry to make a delightful Neapolitan!
So, for our dynamic duo, Carbon Charlie and Carbon Carl, they need to figure out how to best connect with each other and also with those energetic fluorines. They've got a few options for their "hats," and the universe, being the ultimate party planner, picks the perfect combination for the most stable arrangement.
In the case of C2F2, our carbon atoms are doing something truly spectacular. They're not just wearing one type of hat; they're rocking a blend! This leads to a beautiful dance of electrons and a shape that's both elegant and strong.
Let's zoom in on our carbon pals. Each carbon atom, in its original state, has a certain number of electron "rooms" or orbitals. It's like having a few different-sized boxes for its electrons.
But when they decide to bond up in C2F2, they have to make some compromises. They can't just use their original boxes as is. They need to create new, perfectly shaped boxes that are just right for sharing electrons with their neighbors.
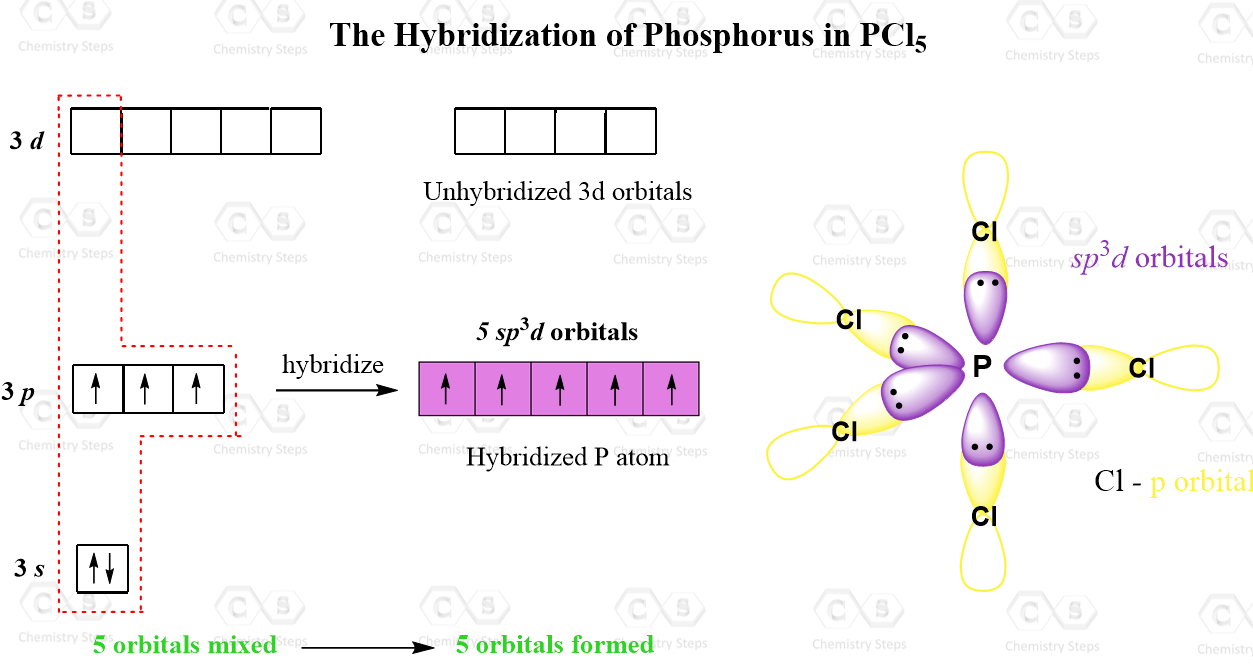
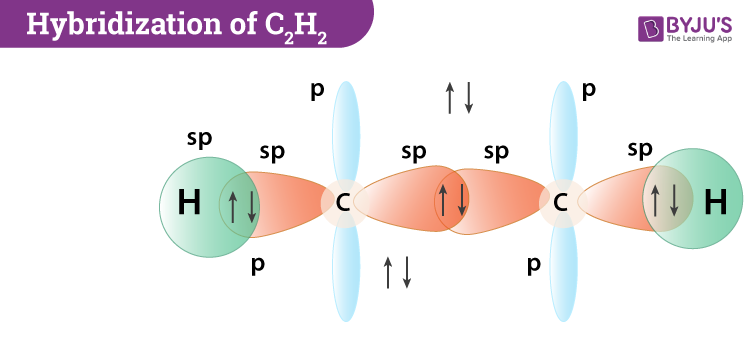
Here's the juicy bit: the carbon atoms in C2F2 undergo sp hybridization. Say it with me: sp! It sounds like a little secret code, doesn't it?
This sp hybridization is like taking one 's' box and one 'p' box and smashing them together to create two new, identical boxes. These new sp boxes are like super-efficient containers, perfectly shaped for bonding.
So, each carbon atom in C2F2 uses its sp hybridized orbitals to connect with its neighbors. It’s like they’re wearing these special, custom-made hats that allow them to form really strong connections.
Now, what happens to the other electron "rooms" that weren't involved in the sp mix? Well, they don't just sit there doing nothing! They remain as pure 'p' orbitals, and in the case of C2F2, they get to do something truly exciting: they form a pi bond.
Think of a pi bond as a side-hug between two atoms. It's formed when these leftover 'p' orbitals from each carbon atom overlap sideways, kind of like two dancers holding hands over their heads. This adds an extra layer of connection and strength to the molecule.
So, in C2F2, we have a sigma bond between the two carbon atoms (formed by the sp orbitals pointing directly at each other, like a firm handshake) and a pi bond (that super cool side-hug). This combination of a sigma and a pi bond is what we call a double bond!
It's like Carbon Charlie and Carbon Carl are not just shaking hands; they're also giving each other a friendly high-five while they're at it. That's a double dose of bonding power!
And the fluorines? They get their own connections too! Each fluorine atom typically forms a single sigma bond with one of the carbon atoms. They're happy to join the party and share their electrons, creating a stable molecular structure.

The overall arrangement of atoms in C2F2, thanks to this sp hybridization of the carbon atoms, results in a linear shape. Imagine a perfectly straight line, with the two carbon atoms in the middle and the two fluorines at either end. It’s incredibly efficient and elegant!
This linear geometry is a direct consequence of those sp hybridized orbitals pointing in opposite directions, 180 degrees apart. It's like the carbon atoms are saying, "Let's spread out and give everyone plenty of space!"
So, to recap our fun little journey, the carbon atoms in C2F2 are definitely rocking sp hybridization. This allows them to form a double bond with each other and a single sigma bond with each of the fluorine atoms. And all of this leads to a wonderfully straight, linear molecule!
It's pretty amazing when you think about it. These tiny, invisible particles are following these intricate rules of engagement, mixing and matching their electron hats to create stable and unique structures. It’s a testament to the incredible order and beauty that exists at the molecular level.

So next time you hear about C2F2, you can confidently say, "Ah, yes! That's the one with the sp hybridized carbon atoms forming a double bond and a lovely linear shape!" You'll sound like a molecular maestro!
It’s these little molecular interactions, these decisions about how to share electrons, that build everything around us. From the air we breathe to the materials we use, it all starts with these fundamental bonding principles.
The world of chemistry is full of these fascinating stories, and hybridization is just one chapter in the epic saga of how atoms come together. It’s a process that’s both complex and utterly captivating.
And remember, even though we're talking about atoms and electrons, it's all about connections. Just like us humans, atoms form bonds, share energy, and create something bigger and more stable than they could be alone.
So, give yourself a pat on the back for exploring the world of C2F2 and its sp hybridized carbon atoms. You’ve just unlocked a little piece of the universe’s amazing blueprint! Keep that curiosity alive, because there are countless more molecular marvels waiting to be discovered. Happy bonding!