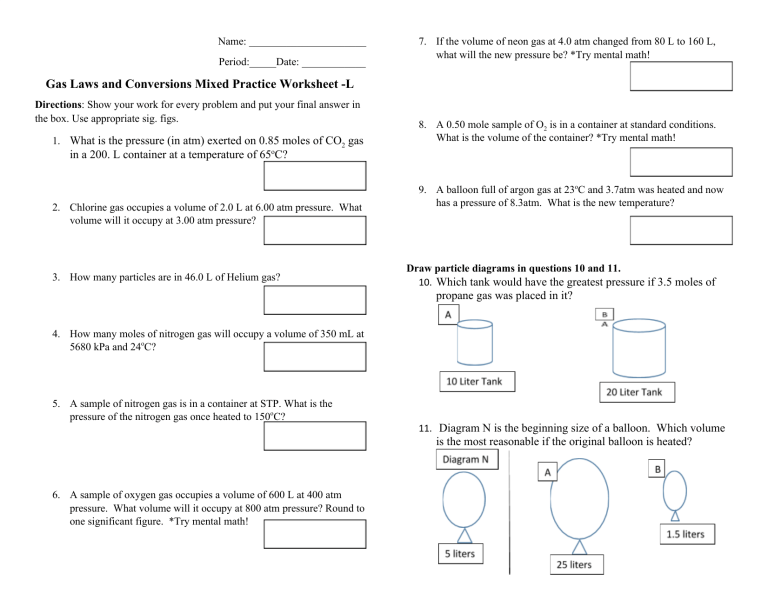
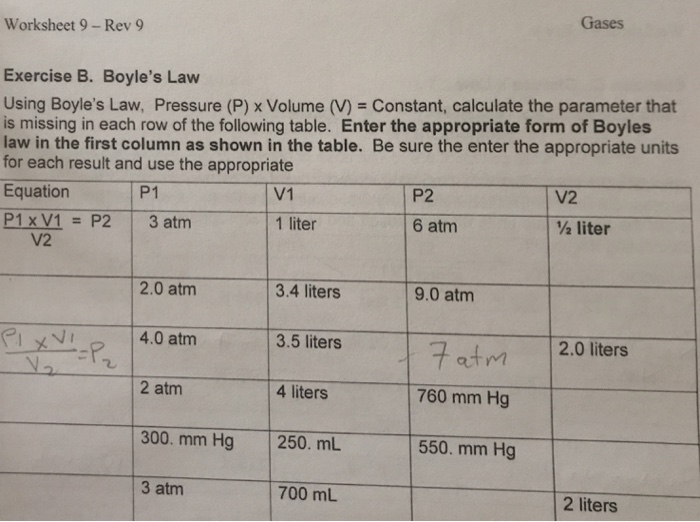
Gas Laws Mixed Practice Worksheet Answer Key

Hey there, science superstar! So, you’ve been wrestling with those tricky Gas Laws, huh? Don't worry, we've all been there. It’s like trying to herd a bunch of enthusiastic molecules – they just don't always do what you think they should! But guess what? You’ve conquered the Gas Laws Mixed Practice Worksheet, and now you're looking for that sweet, sweet relief of an answer key. Well, you’ve come to the right place!
Think of this as your personal victory lap, your little high-five from the universe of chemistry. We’re going to unpack those answers together, not with a stern lecture, but with a friendly chat, maybe a few giggles, and definitely without making your brain feel like it’s about to spontaneously combust (unless it's from sheer brilliance, of course!).
So, grab your favorite beverage, maybe a snack that isn't suspiciously resembling a gas molecule, and let’s dive into the wonderful world of why your answers match (or don't match, and that’s okay too – we’ll figure it out!).
Must Read
The Grand Unveiling: Your Gas Laws Answer Key!
Alright, let’s get down to business. You’ve put in the work, you’ve pondered pressure, you’ve juggled volume, and you’ve probably developed a newfound respect for temperature (seriously, that stuff is important!). Now, for the moment of truth:
Problem 1: The Boyle's Law Bonanza
Remember Boyle's Law? It’s the one where if you squeeze a gas, its pressure goes up, and if you let it relax, the pressure chills out. Mathematically, it’s usually something like P₁V₁ = P₂V₂. It's like a seesaw – one side goes up, the other goes down. Pretty neat, right?
If your answer for Problem 1 involved something like a pressure of 2 atm, given an initial volume and pressure, and a new volume, then you’re likely on the right track! Did you remember to keep your units consistent? That’s often the secret sauce to getting these problems right. No one likes a unit mix-up; it’s the equivalent of trying to measure your pizza in inches and then calculating its area in square feet. Chaos!
Playful Aside: Imagine trying to explain to a pizza delivery person that you want your pepperoni distributed evenly across a surface measured in ‘square giggles’. They’d probably just back away slowly, and honestly, who could blame them?
Problem 2: The Charles's Law Charm
Next up, Charles’s Law! This one’s all about temperature and volume. When you heat up a gas, it wants to expand. It’s like giving it a little warm hug, and it just gets bigger. The formula often looks like V₁/T₁ = V₂/T₂. Crucially, remember that temperature must be in Kelvin. Celsius or Fahrenheit? Nope, those won’t cut it. The gas molecules get really grumpy if you don’t give them the proper K-loving!
So, if your answer for Problem 2 resulted in a volume of, say, 5.0 L, after increasing the temperature, you’re probably doing great. Did you convert your Celsius to Kelvin by adding 273.15? If you forgot, no worries! Just pretend you accidentally put on your socks on inside out; a quick fix and you’re back in business. It happens to the best of us.

Joke Break: Why did the Kelvin thermometer break up with the Celsius thermometer? Because he felt he was always being taken for granted (and also, Celsius was just too hot-headed sometimes!).
Problem 3: The Gay-Lussac's Law Gala
Gay-Lussac’s Law is our friend for when volume stays the same, but pressure and temperature get to dance. As you heat a gas in a rigid container (think of a bomb, but much less explosive and way more scientific!), the pressure shoots up. The formula? You guessed it: P₁/T₁ = P₂/T₂. Again, Kelvin is your best friend here!
If your answer for Problem 3 landed you with a pressure of 3.5 atm, then you’ve likely mastered this concept. It’s all about that direct relationship: more heat, more oomph (pressure-wise!). Imagine a tightly sealed soda bottle you leave in the sun. Yep, that’s Gay-Lussac’s Law in action, and it’s a good reminder to not do that!
Problem 4: The Combined Gas Law Carousel
Now, the Combined Gas Law! This is where things get really interesting because it brings Boyle’s, Charles’s, and Gay-Lussac’s laws all together in one magnificent equation: (P₁V₁)/T₁ = (P₂V₂)/T₂. It’s like the Avengers of gas laws, assembling to solve pretty much any situation where P, V, and T are all changing.
Did your answer for Problem 4 come out to a volume of 10 L? Or perhaps a pressure of 0.8 atm? If you plugged in your initial and final conditions and solved for the unknown, you’ve just wrangled the beast! This is where you really get to show off your calculation skills. Remember to cancel out units that appear on both sides before you start crunching numbers – it saves you a headache and makes the math a lot cleaner.
Playful Aside: If you get this one right, you basically have a superpower. You can predict the behavior of gases. Just try not to use it to win bets about how many jellybeans are in a jar. Unless the stakes are really high, then maybe go for it.

Problem 5: The Ideal Gas Law Idol
Ah, the Ideal Gas Law! PV = nRT. This is the superstar, the main event, the celebrity of gas laws! It connects pressure (P), volume (V), the number of moles (n), the ideal gas constant (R), and temperature (T). This is your go-to when you need to figure out the amount of gas (n) or when you have a bunch of variables thrown at you.
The key here is knowing your R value. Depending on your units, R can be 0.0821 L·atm/(mol·K) or 8.314 J/(mol·K), or even others! Make sure your R value matches the units of your P, V, and T. It’s like wearing the wrong size shoes; it just doesn’t feel right and you can’t perform at your best.
If you calculated a number of moles (n) of, say, 0.5 mol, or a final pressure of 1.5 atm, then you’ve successfully invoked the power of the Ideal Gas Law. This law is so useful, it’s practically royalty in the chemistry kingdom.
Problem 6: Stoichiometry Meets Gas Laws!
Now we’re getting into the real fun! This is where you combine your knowledge of chemical reactions (stoichiometry) with your gas laws. You might need to calculate the moles of a reactant or product first, and then use the Ideal Gas Law (or another gas law) to find its volume, pressure, or temperature. It’s a two-for-one deal!
For example, if a reaction produces 22.4 L of gas at Standard Temperature and Pressure (STP), you’re probably on the right track. Remember, STP is usually 0°C (273.15 K) and 1 atm. If you had to calculate moles first and then find a volume, and your answer was around 30 L, give yourself a pat on the back. You’re basically a chemical detective!
Playful Aside: Stoichiometry is like following a recipe, but instead of flour and sugar, you’re dealing with atoms and molecules. And sometimes, the recipe calls for an ingredient you don’t have, so you have to figure out how to substitute it, or how much of another ingredient you’ll need. It’s culinary chemistry!
Problem 7: The Dalton's Law Dilemma
Dalton's Law of Partial Pressures is all about mixtures of gases. It states that the total pressure of a mixture of gases is simply the sum of the partial pressures of each individual gas. So, if you have gas A with a pressure of 2 atm and gas B with a pressure of 3 atm in the same container, the total pressure is 5 atm. Easy peasy!

If your answer for Problem 7 involved adding up individual pressures to get a total pressure, you've nailed it. This law is super intuitive, almost like common sense for gases. It’s like saying, “The total amount of people in a room is just the sum of all the individuals.” Mind-blowing, I know!
Problem 8: Effusion and Diffusion Fun
Graham's Law deals with how gases escape from a container (effusion) or spread out (diffusion). The key idea is that lighter gases move faster than heavier gases. The rate of effusion or diffusion is inversely proportional to the square root of its molar mass. Think of it this way: a tiny, lightweight molecule can zoom around much faster than a big, heavy one.
If you had to compare the rates of two gases, or calculate the relative speeds, and your answer involved a ratio or a comparison of speeds, you’re on the right track! Did you remember to use molar masses? That’s the key to unlocking Graham’s Law.
Joke Break: Why did the helium atom break up with the oxygen molecule? Because he said, "You’re just too heavy for me! I need someone who can float my boat!"
Problem 9: Real Gases vs. Ideal Gases
This one often trips people up! Ideal Gas Law works great for ideal gases, but real gases can be a bit… well, real. At very high pressures and very low temperatures, real gases start to behave differently because the molecules are close together (and can interact!) and don't have as much kinetic energy. Deviations from ideal behavior become noticeable.
If your answer involved explaining why a gas might deviate from ideal behavior, or identifying conditions under which it would, you’re thinking critically! It’s like understanding that while a superhero can fly in the movies, in real life, gravity tends to keep us grounded. Different rules apply!

Problem 10: The Grand Finale Mix
This is your ultimate test! A problem that might throw a curveball, requiring you to use multiple gas laws or concepts. Perhaps it involves a chemical reaction followed by a change in conditions, or a mixture of gases undergoing a process. If you managed to solve this one, you deserve a medal. A gold medal made of pure, unadulterated scientific triumph!
Your answer might have involved a complex calculation, but the result should make sense within the context of the problem. Did you use the right R value? Did you convert to Kelvin? Did you keep track of your moles? If the answer is yes to all of those, then congratulations! You’ve officially conquered the mixed practice worksheet.
A Few Final Thoughts (and Encouragements!)
So, how did you do? Did your answers line up nicely with the expected solutions? If they did, fantastic! You’ve built a solid foundation in understanding how gases behave. If some answers were a little off, that’s perfectly normal and, dare I say, even beneficial!
Think of it this way: every wrong answer is just a stepping stone to understanding. It’s a chance to go back, re-examine the problem, and see where your thinking might have taken a little detour. Did you forget to convert to Kelvin? Did you mix up your P₁ and P₂? These are common hiccups, and identifying them is a huge part of the learning process.
Embrace the mistakes! Seriously. They’re not failures; they’re data points telling you where to focus your brilliant mind next. The fact that you’re doing these practice problems, and seeking out the answers, shows initiative and a genuine desire to learn. That’s the most important ingredient for success in science, or really, in anything you set your mind to.
Remember, gas laws aren’t just abstract equations. They explain why your tires inflate, why a hot air balloon floats, and why baking soda and vinegar create such a fizzy reaction. They are the invisible forces that shape so much of our world.
You’ve tackled a challenging worksheet, you’ve navigated the complexities of pressure, volume, and temperature, and you’re emerging on the other side with more knowledge and confidence. So, give yourself a huge, enthusiastic round of applause! You absolutely rocked this. Keep that curiosity alive, keep asking questions, and keep celebrating your victories, big and small. The universe of science is waiting for your next brilliant discovery!