Factors That Affect The Activity Of An Enzyme
You know those amazing little helpers inside your body, the ones that make everything from digesting your pizza to building your muscles happen? They're called enzymes, and they're like tiny, super-efficient workers on an assembly line. Without them, life as we know it would grind to a messy, slow halt. But even these hardworking little guys can have their off days, and a bunch of things can mess with their groove.
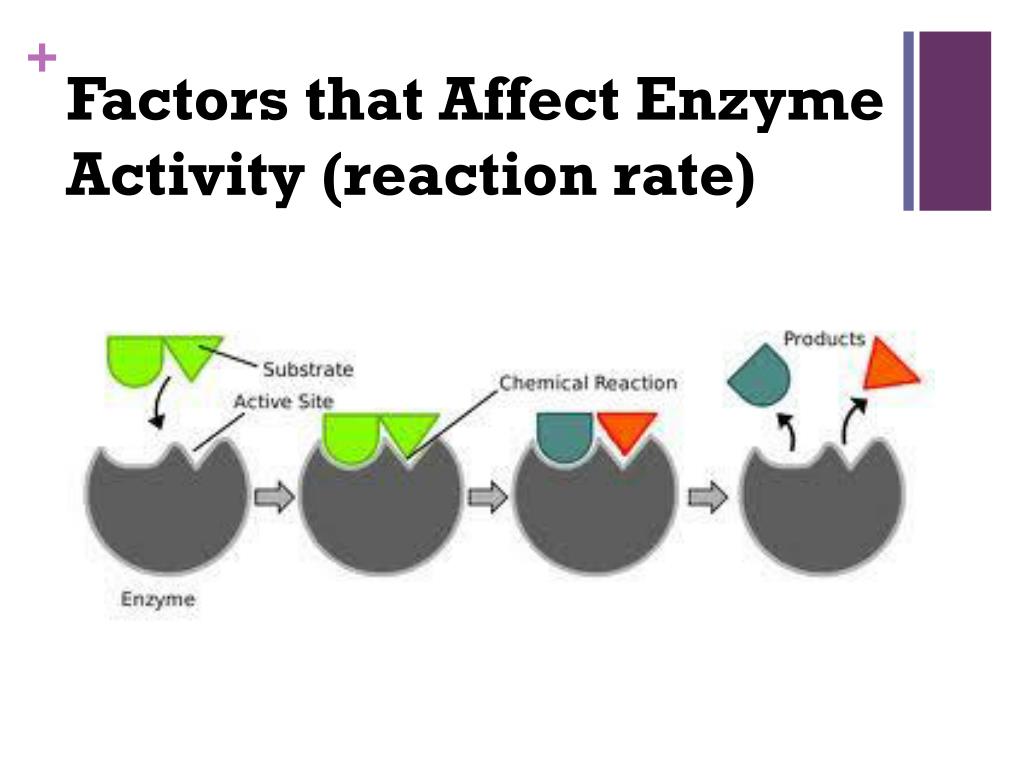
Think of an enzyme like a special key that only fits one lock. This lock is called the substrate. When the enzyme finds its matching substrate, it’s like a perfect handshake, and bam! a reaction happens. This reaction might be breaking something down or building something up. It’s a beautiful dance of molecular matchmaking!
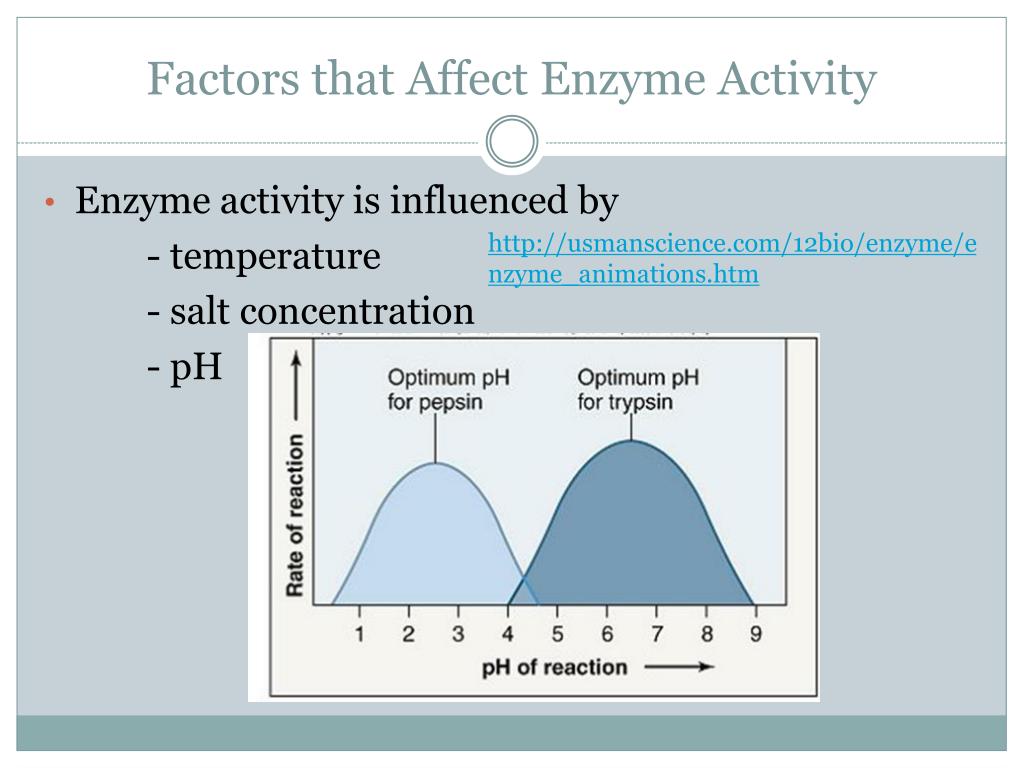
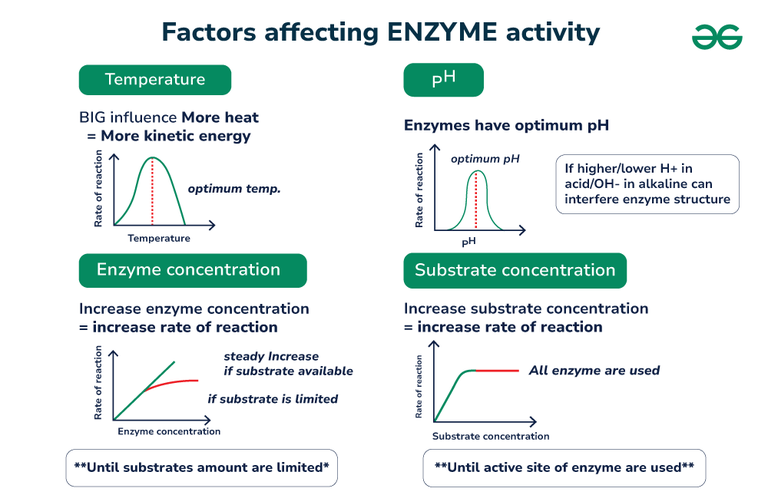
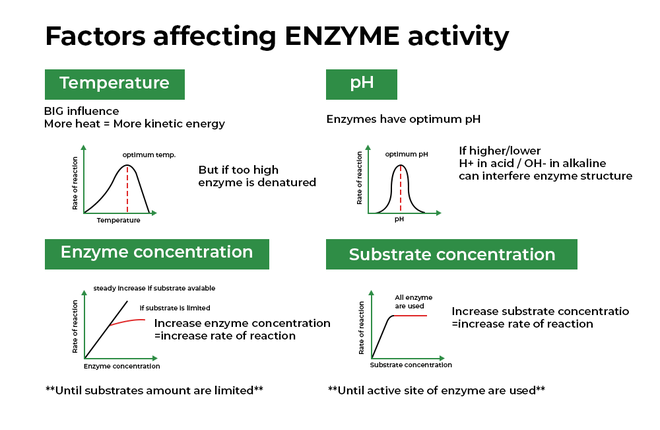
One of the biggest influences on our enzyme buddies is temperature. Just like us, enzymes have a sweet spot for temperature. Too cold, and they get sluggish, moving around like they’re wading through molasses. Too hot, and they can get stressed, even start to fall apart like a flimsy sandcastle in a tidal wave.
Must Read
Imagine a chef trying to bake a cake. If the oven is too cold, the cake won’t rise properly, right? It’ll be a sad, dense lump. Enzymes are similar! They work best within a certain temperature range. For us humans, that range is our normal body temperature, around 37 degrees Celsius (or 98.6 Fahrenheit). That’s why fevers can make you feel so miserable – it’s your enzymes struggling a bit!
On the flip side, if you accidentally overheat an enzyme, it’s like a balloon popping. Its shape gets all messed up, and it can’t do its job anymore. This is called denaturation. It’s a bit of a bummer because once denatured, most enzymes can’t go back to their original, helpful form.
Now, let’s talk about another big player: pH. This is basically how acidic or basic something is. Think of it like a scale, from super sour lemon juice to slippery soap. Enzymes, just like us, have a preferred pH environment. If they’re in an environment that’s too far from their ideal, they get grumpy and their activity takes a nosedive.
Your stomach, for example, is a very acidic place. This is perfect for the enzymes there, like pepsin, which helps break down proteins in your food. But if pepsin somehow ended up in your intestines, which are more on the alkaline (basic) side, it would get confused and stop working efficiently.
It’s kind of like trying to have a sophisticated tea party in a noisy, chaotic playground. The atmosphere just isn’t right for the delicate task at hand. Different enzymes are adapted to live in very specific pH neighborhoods, and they thrive when they’re in their comfort zone.
Then there are these things called inhibitors. These are like little troublemakers that get in the way of our enzyme friends. Some inhibitors are like a tiny pebble dropped into a complex gear system, slowing everything down. Others are more aggressive and can permanently jam the works!
These inhibitors can attach to the enzyme in different ways. Some might just hang around the active site, the spot where the substrate is supposed to bind, blocking it like a bouncer at a club. Others might latch onto a different part of the enzyme, causing it to change shape and no longer fit its substrate, like someone changing the lock on your front door.
Interestingly, some of these inhibitors are actually used in medicines! They can be designed to block specific enzymes that are causing problems in our bodies, like those involved in the growth of harmful bacteria or even cancer cells. So, while they sound like villains, inhibitors can sometimes be our unsung heroes in disguise.
On the other hand, we have activators. These are the encouraging cheerleaders for our enzymes! Activators are substances that help boost enzyme activity, making them work even faster and more efficiently. They might help the enzyme bind to its substrate more effectively or help the enzyme change into its most active form.
Think of an activator like giving your favorite athlete a pep talk and a well-deserved energy drink before a big game. Suddenly, they’re more focused, more energetic, and ready to perform at their peak. Activators are crucial for many biological processes, ensuring that reactions happen at just the right speed.
Some enzymes also need a little something extra to get them going, a helper molecule. These are called cofactors and coenzymes. Cofactors are often metal ions, like zinc or magnesium, which can literally help hold the enzyme in the right shape or participate directly in the chemical reaction. Coenzymes are usually vitamins or their derivatives, acting like tiny tools that assist the enzyme.
Imagine a sculptor working on a masterpiece. The sculptor is the enzyme, and the clay is the substrate. A cofactor might be like a special chisel that the sculptor needs to get the finer details just right, or a coenzyme could be like a specific brush that helps apply the finishing touches smoothly. Without these helpers, the sculpture might be unfinished or not quite as magnificent.
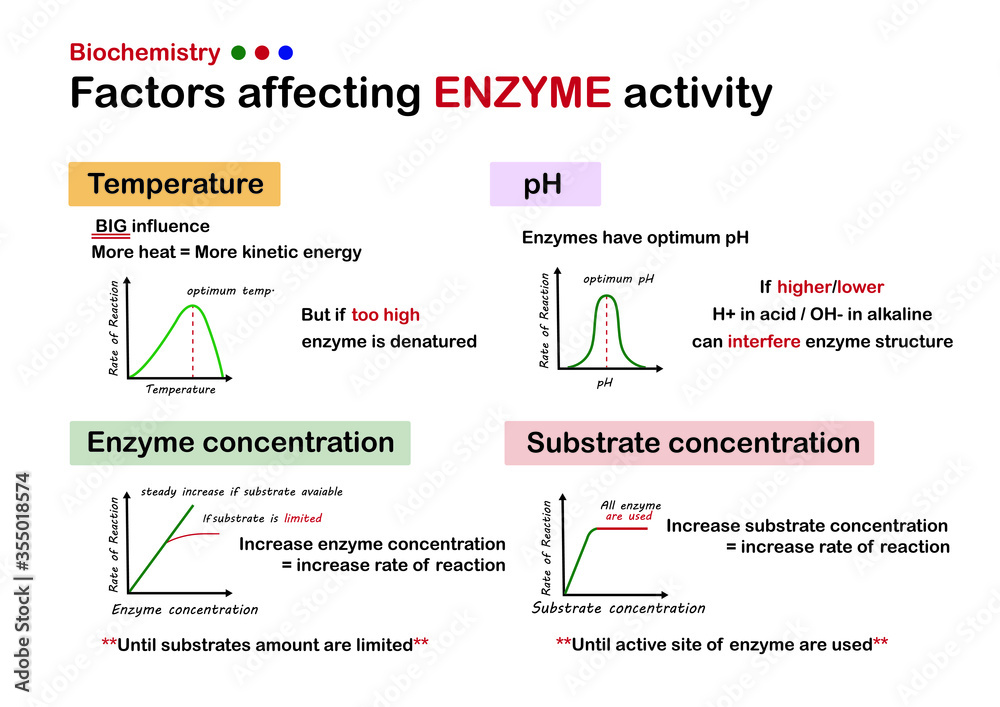
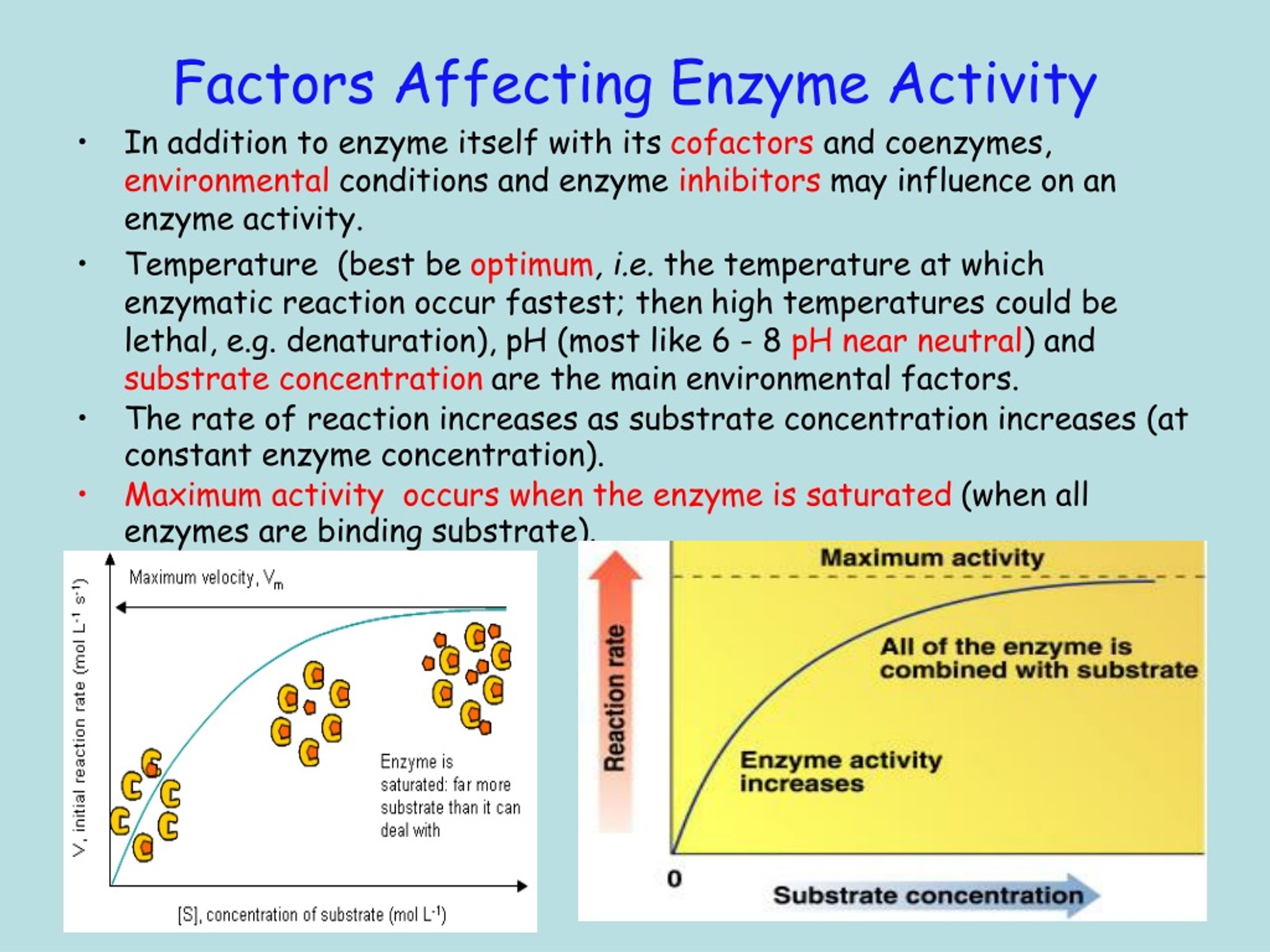
It's fascinating how interconnected everything is. Even the concentration of the enzyme and its substrate can play a role. If you have very few enzyme workers and a mountain of work (substrate), things will move slowly. But if you have plenty of workers and just a little bit of work, the job gets done in a flash!
As the amount of substrate increases, the rate of the reaction usually speeds up – up to a point. Eventually, you run out of enzyme workers to handle all the substrate. It’s like a busy restaurant; at first, more customers mean more business, but once all the tables are full and the kitchen is at capacity, adding more customers won’t make the service any faster.
So, next time you’re enjoying a delicious meal, or even just breathing, take a moment to appreciate the incredible work of your enzymes. They’re sensitive, complex, and influenced by so many things, but they’re also incredibly resilient and vital to your existence. They’re the unsung heroes of your inner world, working tirelessly to keep you running!