Example Of Vaporization

Hey there, ever stopped to think about how that morning coffee just… disappears into the air? Or how a puddle on a hot day shrinks away to nothing? That’s basically vaporization in action, and honestly, it’s one of those super cool, everyday science things that’s way more interesting than it seems at first glance.
So, what exactly is this ‘vaporization’ we’re talking about? Think of it like a substance deciding to ditch its liquid or solid form and become a gas. It's like your favorite cozy sweater suddenly deciding it wants to be a breezy cloud. Pretty neat, right?
It's All About the Energy!
The whole magic behind vaporization really boils down to energy. For a liquid or a solid to turn into a gas, its tiny little particles (atoms or molecules, depending on what it is) need to get a serious pep in their step. They need to absorb enough energy to break free from their neighbors and start zipping around independently as a gas.
Must Read
Imagine you're at a party. The liquid phase is like everyone hanging out, chatting, maybe a bit close together. Solid is like everyone in a big group hug, super tightly packed. But then, the music gets really good, and people start dancing! They’re moving more, spreading out, and some might even jump up and float around – that’s your gas phase!
Where does this energy come from? Usually, it's from the surroundings. Heat is the most common culprit. When you heat water, you're essentially giving those water molecules more energy to do their thing.
Evaporation: The Slow and Steady Change
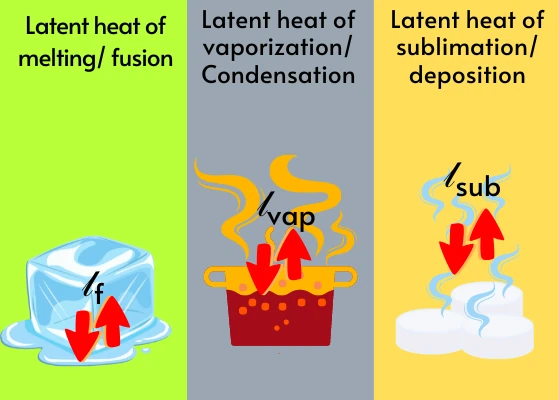
Now, vaporization isn't a one-size-fits-all deal. We have different ways this transformation can happen. The most common one we experience is probably evaporation. This is when a liquid turns into a gas, but it can happen at temperatures below the liquid's boiling point.
Think about that puddle again. The sun isn't necessarily boiling the water, but it's giving it enough energy for the surface molecules to escape into the air. It’s like a gentle nudge, rather than a big shove.

This is why clothes dry on a clothesline, even on a not-so-hot day. The water in the fabric is evaporating into the surrounding air. The air can only hold so much moisture, so as the water evaporates, it makes space for more. Pretty clever system, nature's got going on, huh?
Even when things feel dry, there's still a bit of evaporation happening. It’s a continuous process! It’s like a constant, subtle breeze carrying tiny bits of liquid away.
Boiling: The Dramatic Transformation
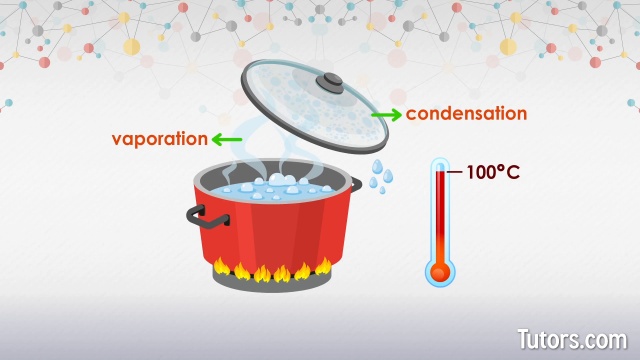
Then there’s boiling. This is the more dramatic, energetic cousin of evaporation. Boiling happens when you heat a liquid to its specific boiling point. At this temperature, the liquid has enough energy for the particles throughout the entire liquid to turn into a gas, not just at the surface.
When you see water boiling in a pot, those bubbles you’re seeing? Those are actually pockets of water vapor forming inside the liquid. They rise to the surface and pop, releasing the gas into the air. It's like the liquid is having a fizzy, bubbly party!

The boiling point is a pretty special temperature for each substance. For water at sea level, it's 100 degrees Celsius (or 212 degrees Fahrenheit). But if you’re on a mountain, where the air pressure is lower, water boils at a slightly cooler temperature. Isn't that wild? The environment can actually change when something boils!
Boiling is super important in lots of things we do. Cooking, of course, is a big one. Think about pasta, rice, or making soup – all rely on boiling water. It’s also used in industrial processes and even in some power plants to generate electricity.
Sublimation: The Sneaky One
But wait, there’s more! Vaporization isn't just limited to liquids. There’s also a phenomenon called sublimation. This is when a solid directly turns into a gas, skipping the liquid phase entirely. Pretty sneaky, right?
The classic example of sublimation is dry ice. Dry ice is frozen carbon dioxide. If you leave a piece of dry ice out on a table, it doesn't melt into a puddle of liquid. Instead, it turns directly into carbon dioxide gas, which you can often see as a foggy mist. It’s like a magician’s trick, making something disappear without a trace of its liquid form.

Another everyday example? When your freezer gets that frosty coating. Those ice crystals on the walls can actually sublimate directly into water vapor, even though it's cold in there. It’s a slow process, but it happens!
Why does sublimation happen? It’s all about the pressure and temperature. Under certain conditions, the solid can absorb enough energy to skip the intermediate liquid stage and become a gas.
Why Is Vaporization So Cool?
So, why should we care about vaporization? Well, it's pretty fundamental to how our planet works and how we live. Think about the water cycle! Evaporation from oceans, lakes, and rivers is what puts water vapor into the atmosphere, which eventually forms clouds and then rain or snow. Without evaporation, we wouldn't have weather as we know it!
It's also responsible for cooling things down. When we sweat, the evaporation of that sweat from our skin takes heat away, which helps our bodies regulate temperature. It’s our built-in air conditioning system!

On a more fun note, think about ice cream on a hot day. The melting ice cream is a liquid, and then it drips. But the cold evaporating from it actually helps to keep it from melting too fast. It’s a delicate balance!
And let's not forget about cooking. From steaming vegetables to brewing coffee, vaporization plays a role in many of our favorite culinary experiences. The aroma of freshly brewed coffee? That’s the smell of coffee compounds vaporizing and wafting into the air.
Everyday Wonders
Honestly, the more you look, the more you see vaporization everywhere. It's in the way perfumes and colognes release their scents. It's in the drying of paint. It's in the humidifiers and dehumidifiers we use in our homes.
It’s just a reminder that the world around us is constantly in motion, undergoing subtle transformations driven by energy. It’s a beautiful dance of molecules, turning the ordinary into the extraordinary. So next time you see steam rising from a hot drink or notice a puddle disappearing, give a little nod to the amazing process of vaporization. It’s a little bit of everyday science that’s truly out of this world!

:max_bytes(150000):strip_icc()/boiling-kettle-in-kitchen-135879673-5c00874e46e0fb00019fec34.jpg)
