Draw The Structure Of 3 7 Dimethyl 4 Nonyne

Alright, let's talk about something that might sound a little fancy but is actually quite fun. We're going to "draw" the structure of 3,7-dimethyl-4-nonyne. Don't worry if that sounds like a mouthful of alphabet soup. We'll break it down, like a delicious molecular puzzle.
Imagine you have a bunch of LEGO bricks, but instead of colorful plastic, they're atoms. And instead of snapping together, they're held by invisible "bonds." Chemistry is basically a super-powered LEGO builder's paradise. So, let's get our tiny atom bricks ready.
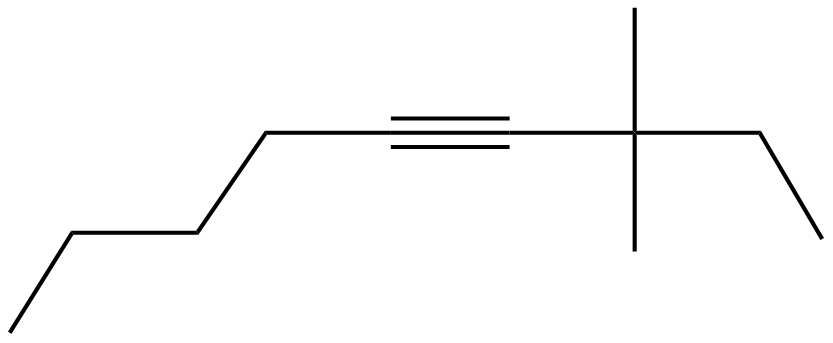
First up, the "non" in nonyne. This tells us we have a chain of nine carbon atoms. Think of it as a straight line of nine little atom buddies holding hands. This is our backbone, the main road for our molecular party.
Must Read
Now, let's add the "yne" part. This is the exciting bit! The "yne" means we have a triple bond. It's like two of those carbon buddies are holding hands extra, extra tightly, forming a super-strong connection. This triple bond is at position number four on our nine-carbon chain.
So, picture our line of nine carbons. We count them one by one. The triple bond is happening between the fourth and fifth carbon. It's a bit like a very enthusiastic handshake that’s so strong, it fuses their arms together for a moment.
Next, we have the "dimethyl." This is where things get a little more social. "Di" means two. And "methyl" refers to a little group made of one carbon and three hydrogens (we call this a CH3 group). So, we have two of these little methyl groups to place.
The numbers "3" and "7" tell us where these party guests are going. On our nine-carbon chain, we put one methyl group on the third carbon. And the other methyl group goes on the seventh carbon. They're like little side streets branching off our main molecular highway.
So, let's visualize this. We have our main nine-carbon road. We've got that super-tight triple bond happening in the middle, between carbons four and five. Then, we attach a little CH3 branch to carbon three.

And, just to make sure it's perfectly balanced, we attach another CH3 branch to carbon seven. It's like adding little decorative flags to our molecular structure. Makes it look all official and important, doesn't it?
Sometimes, when I'm drawing these things, I like to imagine them as little stick figures. The carbon atoms are the bodies, and the bonds are the arms and legs. The triple bond is like a super-flexed arm that's incredibly strong.
And those methyl groups? They're like little arms sticking out sideways. One is on the third "body," and the other is on the seventh "body." It’s a funny thought, but it helps make it less intimidating.
Now, let's talk about the hydrogens. Remember those CH3 groups? The "3" means there are three hydrogen atoms attached to that single carbon. They're like tiny little smiley faces adding to the fun.
On our main chain, the carbons that aren't involved in the triple bond, or the branches, need to be holding hands with enough hydrogens to make them happy. Carbon likes to make four connections in total.

So, the carbons at the ends of the main chain, carbons one and nine, will be attached to three hydrogens each. They're like the polite folks at the end of the line, just happily holding hands with their single carbon buddy and some hydrogens.
The carbons involved in the triple bond (carbons four and five) are already super busy. Carbon four is connected to carbon three by a single bond and to carbon five by the triple bond. That’s three connections already! So, it only needs one more connection, and that will be to a hydrogen atom.
Carbon five is also connected to carbon four by the triple bond and to carbon six by a single bond. Again, three connections. So, it also gets one hydrogen friend. They're like the really popular kids, connected to everyone.
The carbons that have a methyl group attached (carbons three and seven) are also busy. Carbon three is connected to carbon two, carbon four, and the methyl group. That's three connections. So, it also needs one hydrogen friend.
Carbon seven is connected to carbon six, carbon eight, and its methyl group. Three connections again, so it gets one hydrogen friend. See? Everyone is accounted for and has just the right number of friends.

It's like a very organized, slightly peculiar family reunion. We've got the nine-person core family, the ultra-close twins (the triple bond), and then a couple of cousins (the methyl groups) who've come to visit and brought their own little entourages (the hydrogens).
If you were to actually draw it, you’d probably use lines to represent the carbon-carbon bonds. The triple bond would be three parallel lines between two carbons. The single bonds would be single lines.
And those methyl groups? They'd be little branches with a C at the end, and then you'd imagine three H's attached to that C. Or, in some fancy drawings, you might even draw the H's as little circles.
It's funny, sometimes I feel like the molecular world is playing a game of "Simon Says" with us. "Simon says draw a nine-carbon chain." "Simon says put a triple bond on carbon four." "Simon says add methyl groups to three and seven." And we're all just trying to follow the instructions.
And the beautiful thing is, there's only one way to get this particular structure right. If you put the triple bond in the wrong place, or a methyl group on carbon two instead of three, it's a whole different molecule. It's like a secret code that only makes sense when all the parts are in their exact positions.

So, when you see 3,7-dimethyl-4-nonyne written down, don't be scared. It's just a blueprint. A very specific instruction manual for building a tiny, invisible structure. It’s a molecular recipe, if you will.
Think of it as an invitation to a molecular party. You've got your main guests (the nine carbons), your very energetic dancers (the triple bond), and your friendly socialites (the methyl groups) all mingling together. And the hydrogens? They're the enthusiastic party helpers, making sure everyone is happy and stable.
And here’s my unpopular opinion: drawing these structures can be strangely satisfying. It’s like solving a Sudoku puzzle, but with atoms. There’s a neatness to it, a logical flow that just feels right once you get the hang of it.
It’s a tiny world, but it’s full of fascinating structures. And 3,7-dimethyl-4-nonyne is just one of millions. Each one with its own unique shape and personality. It makes you wonder what other amazing molecular designs are out there, just waiting to be discovered or, in our case, drawn.
So, the next time you hear a chemical name that sounds like a magical incantation, remember this. It’s just a set of instructions, a blueprint. And with a little imagination, you can draw it, visualize it, and maybe even appreciate its quirky charm. It’s not so scary when you break it down into simple steps, right?