Draw The Correct Structure For Benzaldehyde.
Ever wondered what gives almonds that delicious, characteristic scent? Or perhaps you've noticed how some fancy soaps and perfumes have a distinctly sweet, floral aroma? Well, a big part of that often comes down to a fascinating molecule called benzaldehyde. Learning to draw its structure might sound a bit scientific, but trust me, it’s surprisingly satisfying and opens up a world of understanding about the scents and flavors around us!
For those just dipping their toes into the world of chemistry, drawing the structure of benzaldehyde is like learning your first few chords on a guitar – it’s a foundational skill that unlocks a lot more. It’s a simple yet elegant molecule, and understanding its form helps you appreciate how it interacts with our senses. If you're a parent looking for a fun, educational activity to do with your kids, this can be a fantastic way to introduce them to the building blocks of everyday things. It’s a hands-on, visual way to make abstract concepts tangible. Hobbyists, especially those interested in perfumery, aromatherapy, or even baking with natural flavorings, will find that knowing this structure gives them a deeper insight into the ingredients they use.
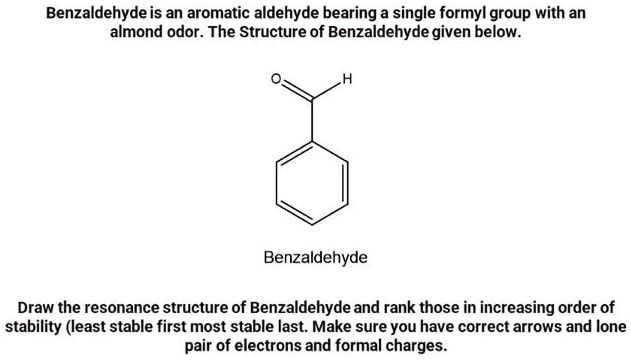
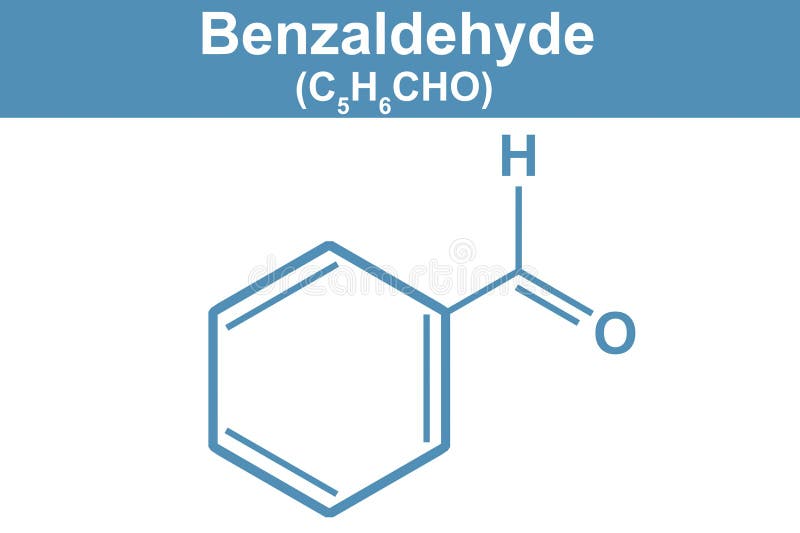
Think of benzaldehyde as a basic LEGO brick for many other aromatic compounds. Its structure is quite distinctive: it's essentially a benzene ring (a hexagon of six carbon atoms with alternating double bonds) with a carbonyl group (one carbon atom double-bonded to an oxygen atom) attached to it. This carbonyl group is then attached to a hydrogen atom, making it an aldehyde group. So, you have a stable, flat ring with a slightly reactive, scent-producing tail. Variations can occur when you add or change groups on the benzene ring, leading to a whole family of related compounds with slightly different smells and properties – like vanillin (which gives vanilla its flavor) or cinnamaldehyde (the key component of cinnamon).
Must Read
Getting started is easier than you might think. All you need is a pen and paper. First, draw a hexagon, representing the benzene ring. You can add little circles or alternating double lines inside to show the delocalized electrons that make the ring so stable. Next, pick one of the carbon atoms on the ring and attach an "CHO" group to it. This "CHO" represents the aldehyde group: a carbon atom (C) double-bonded to an oxygen atom (O) and single-bonded to a hydrogen atom (H). Easy peasy! You’ll see it represented in different ways – sometimes as a skeletal structure where carbons and hydrogens are implied, and sometimes with all atoms explicitly drawn. For beginners, drawing out all the atoms is a great way to solidify your understanding.
Drawing the structure of benzaldehyde is a small step, but it’s a gateway to understanding a vast array of chemical compounds that enrich our lives. It’s a moment of clarity where a complex-sounding name transforms into a simple, understandable shape, giving you a new appreciation for the world of molecules and the delightful scents they create. So, grab a pen and give it a try – you might just find it surprisingly rewarding!