Draw 10 Water Molecules To Create A Cluster
Hey there, science curious folks! Ever think about water? Like, really think about it? We drink it, we swim in it, we use it to make coffee. But have you ever considered what happens when you get just a handful of water molecules together? We’re talking about the teeny-tiny building blocks of all that wet stuff. And get this: you can actually draw 10 of them to create a cluster. Mind. Blown.
Seriously! It sounds like a game, right? Like, “Draw 10 monsters to win!” But nope. This is real science. And it’s way cooler than any game. We’re diving into the microscopic world of water. And trust me, it’s a wild ride.
The Secret Lives of Water Molecules
So, what exactly is a water molecule? Imagine a little Mickey Mouse head. That's kinda it. You've got one big oxygen atom, and then two smaller hydrogen atoms hanging off its sides. Like ears, but wiggly. And these little guys are super important. They're the reason water does all the cool things it does.
Must Read
But here's where it gets really fun. These water molecules aren't just floating around alone, all shy and independent. Nope! They're social butterflies. They love to stick together. And when they do, they form something called a cluster.
Enter the Cluster: Ten Tiny Buddies
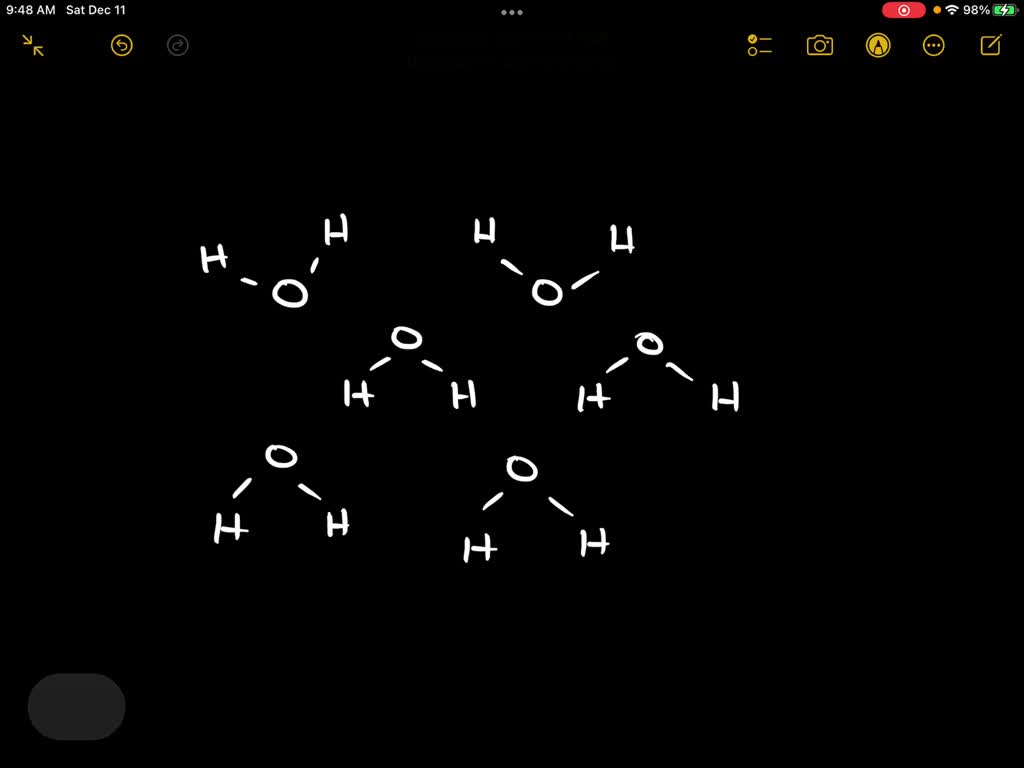
Now, when we say “draw 10 water molecules to create a cluster,” we’re not talking about grabbing a pencil and scribbling. We’re talking about thinking about them interacting. Imagine you’ve got 10 of these little Mickey Mouse water molecules hanging out. What do they do?
They start to connect! They form these amazing, ever-changing little groups. Think of it like a tiny, invisible dance party. The hydrogen atoms from one molecule get a little attracted to the oxygen atom of another. It's called a hydrogen bond. And it’s the secret sauce of water!

These clusters aren't static, either. They’re constantly forming and breaking apart. It's like a never-ending game of molecular musical chairs. Super dynamic. Super cool. And completely invisible to our eyes. Which, let’s be honest, just makes it more intriguing, right?
Why 10 is Kind of a Magic Number
So, why 10? Is there something special about that number? Well, not in a rigid, “you MUST have 10 or it’s not a cluster!” kind of way. It’s more about giving you a manageable number to visualize. A small, understandable group that shows the basic principles of how water molecules interact.
When you have just a few molecules, they might not form super organized structures. But as you get to around 10, you start to see the beginnings of those interesting patterns. You can imagine them linking up in a ring, or a chain, or all sorts of weird and wonderful shapes.
It’s like building with LEGOs. One brick is just a brick. Ten bricks? Now you can start to build a little house, or a car. You get the idea. 10 molecules are enough to show the potential for structure, for organization, for that signature water-y behavior.

The Gooey, Wiggly Truth About Water
One of the most fascinating things about these clusters is how they contribute to water’s unique properties. Think about it. Water is a liquid at room temperature. It flows. It can take the shape of its container. That’s thanks to those constantly forming and breaking hydrogen bonds!
If water molecules were just… well, alone, they’d probably be a gas, like steam. Or maybe a solid, like ice, but in a much more rigid way. But the ability of these clusters to shift and slide past each other is what gives water its famous fluidity. It’s the dance of the water molecules!
And get this: scientists have spent ages studying these clusters. They use fancy computers and complex math to figure out how they behave. But even a simple drawing, a simple visualization of 10 molecules, can give you a glimpse into this incredible world.
Quirky Water Facts You Didn't Know You Needed
Let’s sprinkle in some fun facts, shall we? Did you know that water is the only substance on Earth that exists naturally in all three states of matter – solid, liquid, and gas – at temperatures commonly found on the planet? Pretty neat, huh? And it’s all thanks to those little Mickey Mouse heads and their social habits.

Also, that feeling when you’re trying to get that last bit of ketchup out of the bottle? That’s related to water’s cohesion (water molecules sticking to each other) and adhesion (water molecules sticking to other things). It’s science, but it feels like pure magic sometimes.
And don’t even get me started on surface tension! That’s why some bugs can walk on water. They’re basically surfing on a trampoline made of water molecules holding hands. How cool is that?
So, What's the Big Deal About Drawing Them?
The “draw 10 water molecules” idea is a fantastic way to get your brain thinking about the invisible. It’s an invitation to explore. To imagine the tiny, bustling world that makes up everything we see and touch.
It’s not about perfect accuracy. It’s about understanding the concept. Picture those 10 little guys. See them nudging each other, linking up with those hydrogen bonds. Feel the energy. Feel the movement.
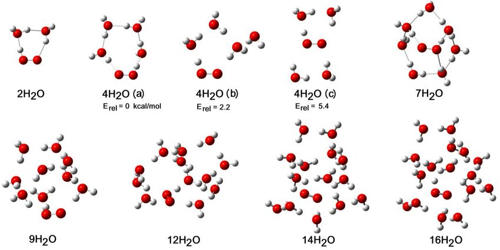
It’s a starting point. A gateway drug to the wonders of chemistry and physics. You start with 10 water molecules, and before you know it, you’re wondering about ice crystals, or how plants suck up water, or even the formation of clouds!
It's All About the Connections
Ultimately, this whole concept is about connections. The way molecules interact. The way simple components can build complex and fascinating systems. Water is just the most obvious, most accessible example of this on our planet.
So next time you see a puddle, or take a sip of water, pause for a second. Imagine those 10 (or 100, or 10 trillion!) water molecules. See them dancing, linking, and forming those essential clusters. It’s a tiny universe in every drop.
And the best part? You can draw them. You can visualize them. You can start to understand the fundamental forces that shape our world, all by simply imagining 10 water molecules creating a cluster. How’s that for a fun science project?