Does Hydrogen Iodide Have Hydrogen Bonding
Ever wondered about the tiny, invisible forces that hold things together? It's a bit like a secret handshake for molecules! Today, we're diving into a fun, and surprisingly useful, question: Does hydrogen iodide have hydrogen bonding? This might sound like a mouthful, but understanding these molecular interactions can unlock a whole new appreciation for the world around us, from the water we drink to the chemical reactions that make our lives possible. It’s a peek into the fascinating world of chemistry that anyone can enjoy!
So, who is this article for? If you're a beginner curious about the building blocks of matter, this will demystify a common chemistry concept. For families exploring science together, it’s a great way to spark curiosity and have some fun discussions about how things work. And for hobbyists who enjoy tinkering or learning new things, grasping hydrogen bonding can add a deeper layer to your understanding of various phenomena, whether you're into cooking, gardening, or even making your own cleaning solutions.
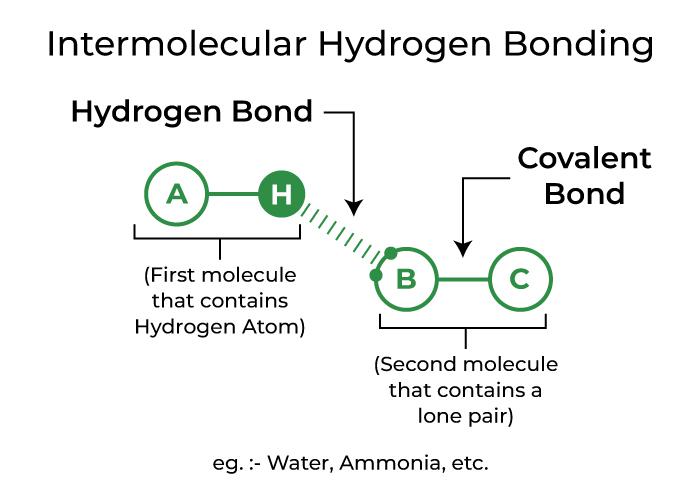
Let's get to the heart of it. Hydrogen bonding is a special type of attraction between molecules. It happens when a hydrogen atom is bonded to a highly electronegative atom (like oxygen, nitrogen, or fluorine) and is then attracted to another electronegative atom on a different molecule. Think of it as a gentle, positive pull. For example, water (H₂O) is famous for its strong hydrogen bonding. The hydrogen atoms in one water molecule are attracted to the oxygen atoms of other water molecules. This is why water has a relatively high boiling point and why ice floats – it's all thanks to these little molecular hugs!
Must Read
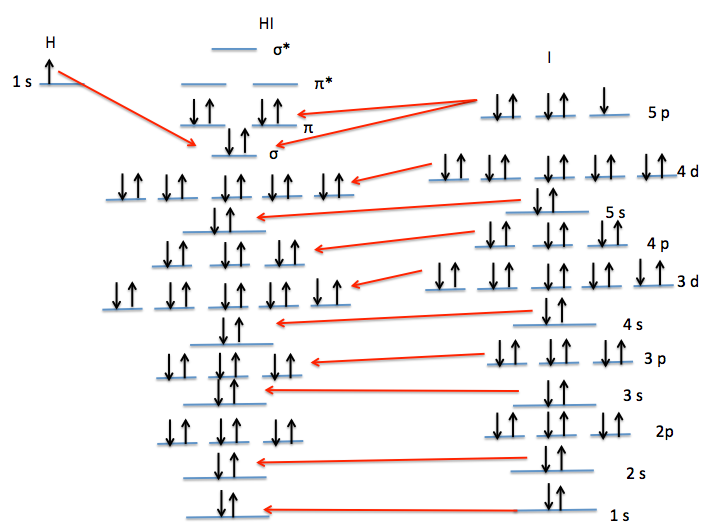
Now, what about hydrogen iodide (HI)? This molecule consists of one hydrogen atom bonded to one iodine atom. Iodine is an electronegative atom, but it's not as electronegative as oxygen, nitrogen, or fluorine. For strong hydrogen bonding to occur, there needs to be a significant difference in electronegativity between the bonded atoms. In HI, the difference isn't quite large enough to create the strong, directional attraction we see in, say, water. So, the short answer is: hydrogen iodide does NOT exhibit significant hydrogen bonding. While there might be very weak dipole-dipole interactions, they aren't classified as true hydrogen bonds. You'll find stronger attractions between HI molecules, but they aren't the characteristic hydrogen bond.
Thinking about variations, compare HI to its cousins like hydrogen fluoride (HF), which has very strong hydrogen bonding, or hydrogen chloride (HCl), which has some but much weaker than HF. This difference in bonding strength explains many of their different physical properties.

Getting started with understanding this is easy! You don't need a lab. Start by looking at the molecules you interact with daily. Think about water – why is it liquid at room temperature? That's hydrogen bonding! Then, consider other common substances and research their chemical formulas. See if they contain hydrogen bonded to oxygen, nitrogen, or fluorine. Websites like Wikipedia or educational chemistry sites are great resources. Imagine the molecules as little Lego bricks, and hydrogen bonding is like a special, strong type of connector that holds certain bricks together in a unique way.
Exploring the world of molecular interactions, like whether hydrogen iodide has hydrogen bonding, is a journey of discovery. It’s about understanding the invisible forces that shape our visible world. So next time you see water boiling or ice melting, remember those tiny molecular handshakes. It’s a simple concept with profound implications, making chemistry accessible and enjoyable for everyone!

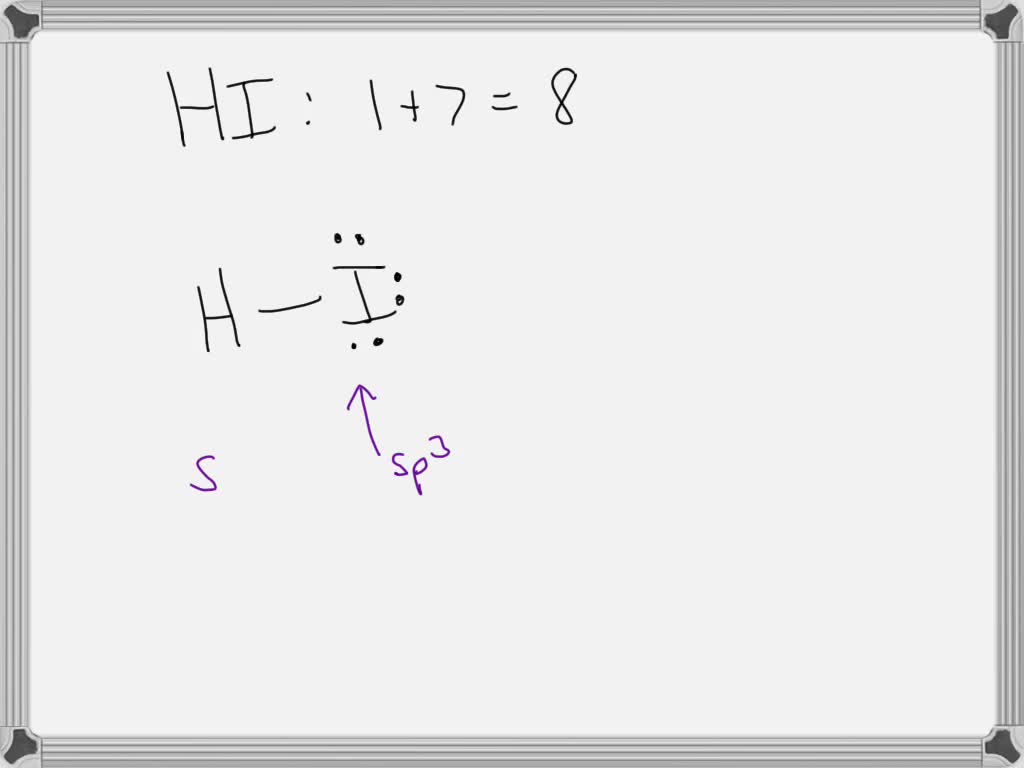


![[Solved]: Consider the synthesis of hydrogen iodid](https://media.cheggcdn.com/study/f79/f79d8866-0ecf-43f0-887e-a3e11d1d964a/image.jpg)