Does Decreasing The Temperature Increase The Rate Of A Reaction
Alright, settle in, grab your latte, and let's chat about something that sounds like it belongs in a super-serious science lab, but is actually as common as your morning toast: how temperature messes with how fast stuff happens. You know, like when you’re trying to bake cookies and the recipe screams at you to use a preheated oven? There’s a reason for that, and it’s all about the temperature tango of chemical reactions.
So, the big question, the one that keeps scientists up at night (or at least gives them something to doodle on napkins during boring meetings): Does decreasing the temperature increase the rate of a reaction? Drumroll, please…
The answer is a resounding, earth-shattering, cookie-crumbling… NO!
Seriously, it’s the exact opposite. Think of it like this: imagine you’re at a party. When the music is pumping, everyone’s dancing like a maniac, bumping into each other, spilling drinks – pure chaos and energy, right? That's kind of like a reaction at a high temperature. Molecules are zipping around like caffeinated toddlers, smashing into each other with gusto. These energetic collisions are what make chemical reactions happen. They need a good shove, a little ‘oomph,’ to break old bonds and form new ones. The hotter it is, the more ‘oomph’ they’ve got.
Must Read
Now, what happens when you turn down the thermostat? The party starts winding down. People are huddled in corners, nursing their lukewarm drinks, maybe doing a polite sway. The energy levels drop dramatically. Molecules, our party guests, start chilling out. They slow their roll, moving at a glacial pace. Their collisions become less frequent and, crucially, less forceful. It's like trying to get a group of sleepy sloths to build a LEGO castle – it's just not gonna happen with any speed.
So, if you're hoping to whip up a batch of super-fast pickles by chucking them in the freezer, you're going to be disappointed. In fact, you might just end up with a sad, frozen, and un-pickled cucumber. Lower temperatures mean slower reactions. It's a fundamental rule, as reliable as gravity making your dropped toast land butter-side down. And trust me, science has confirmed this more times than you've confirmed that you definitely need another slice of pizza.

Why the Chill Kills the Chemistry
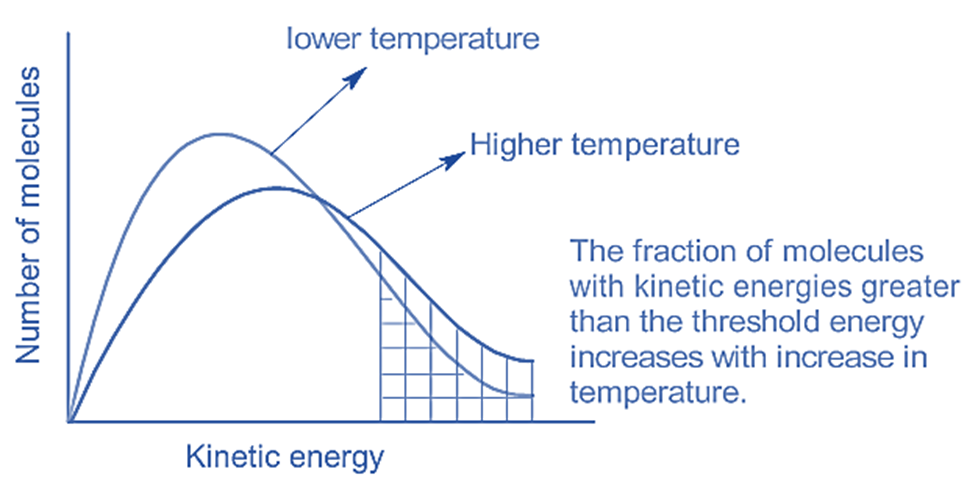
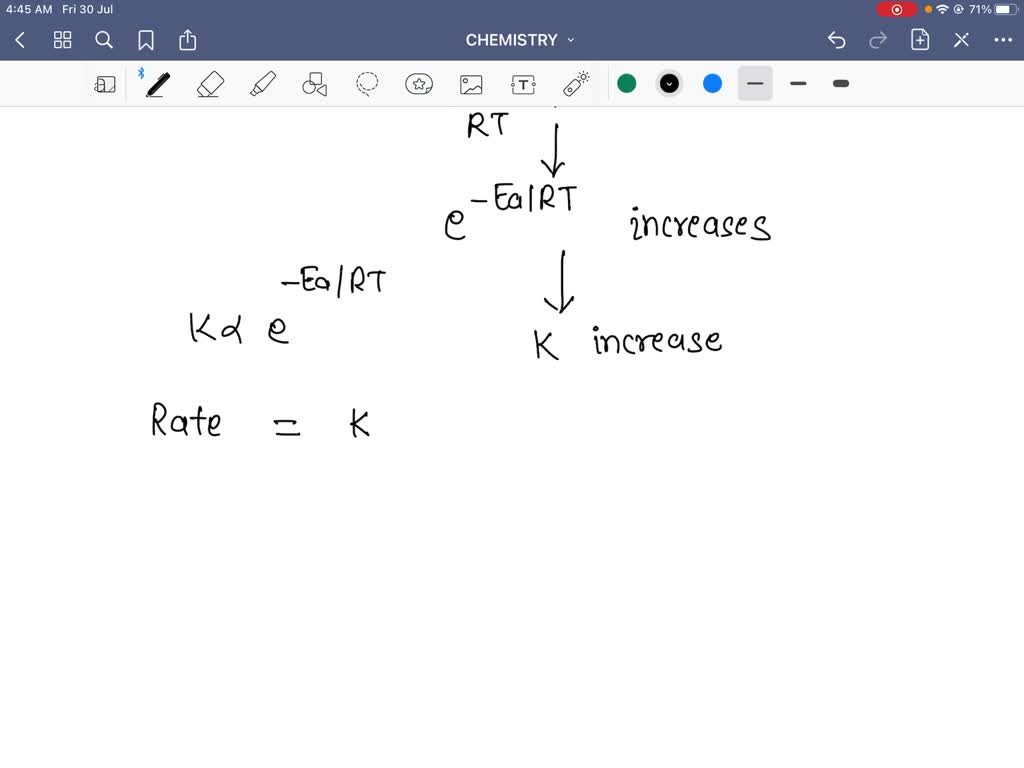
Let's get a tiny bit sciency, but I promise, no complex equations that will make your eyes water. We're talking about activation energy. Imagine this as a little hill that molecules have to climb over to react. For a reaction to happen, the colliding molecules need to have enough energy to get over this hill. Think of it like trying to get a stubborn mule up a small incline. If the mule has a good carrot (energy), it might just saunter up. If it’s already half asleep and there are no carrots in sight, it’s going to be a very long, slow process, if it happens at all.
When you increase the temperature, you're essentially giving more molecules a running start. A larger percentage of them will have enough energy to clear that activation energy hurdle. It's like giving all the party-goers a shot of espresso – suddenly, everyone’s got the energy to dance like nobody’s watching (even if your Aunt Mildred is). More collisions with sufficient energy = faster reaction.
Conversely, when you decrease the temperature, you're like a party pooper who confiscates all the energy drinks. Fewer molecules have the necessary pep to get over that activation energy hill. The collisions that do happen are more like polite nods than enthusiastic handshakes. They just don't have the oomph to break those chemical bonds and create something new. So, the reaction just… meanders. Slowly. Painfully slowly.
A Few Fun Facts (Because Science Should Be Fun-ish)
Did you know that some incredibly slow chemical reactions are actually happening right under our noses? Take the rusting of iron. That beautiful, earthy orange we see? That’s a chemical reaction. But it takes ages! If you put a brand new shiny bike in a hot, humid place, it’ll start to rust much faster than if you leave it in a cool, dry garage. Mother Nature’s speed control, courtesy of temperature!
And what about that carton of milk in your fridge? Why is it there? To keep it from going bad! Bacteria are tiny little chefs, and they do their work through chemical reactions. When you cool down milk, you're essentially telling those bacterial chefs to put down their spatulas and take a very long nap. The reactions that cause milk to spoil slow to a crawl. It’s like putting the entire restaurant on lockdown.
![[ANSWERED] How does decreasing the temperature of the reactants affect](https://media.kunduz.com/media/sug-question-candidate/20220510210340746439-4401941.jpg?h=512)
Conversely, think about cooking! When you heat up food, you're speeding up all sorts of delicious chemical reactions. The browning of meat, the softening of vegetables, the transformation of raw dough into fluffy bread – all these are accelerated by heat. If you tried to bake a cake at room temperature, you’d probably end up with a very sad, uncooked puddle. Your oven is basically a giant chemical reaction accelerator for baked goods.
Here’s a slightly mind-bending one: the rate of many biological reactions, like the ones happening in your own body, roughly doubles for every 10-degree Celsius increase in temperature. So, if you’re feeling a bit sluggish, maybe it’s just your internal chemistry asking for a bit of a warm-up! (Disclaimer: Please don’t try to speed up your metabolism by sitting in a sauna for hours. Your body has limits, and they're not as forgiving as a lenient professor.)
So, next time you’re fumbling with a recipe or wondering why your leftovers are still good after a week in the fridge, remember the humble temperature. It’s the unseen force, the grand conductor of the chemical symphony, dictating the tempo of change. And the rule of thumb is pretty darn simple: hotter means faster, colder means slower. Unless you’re a scientist trying to make a super-cooled superconductor react, in which case, the rules get a little… weird. But for us regular folks, it’s a beautifully simple, often delicious, and sometimes pungent truth.