Do Noble Gases Have High Ionization Energy

Let's talk about something that might sound a bit scientific, but stick with me, because it's surprisingly relevant to our everyday lives and frankly, kind of fascinating! We're diving into the world of noble gases, and specifically, we're going to tackle a question that might have you scratching your head: do these elements have a high ionization energy? Now, I know what you might be thinking – "Chemistry? For fun?" But bear with me! Think of it like this: understanding these fundamental building blocks of the universe helps us appreciate the amazing technologies we use every single day, from the lights that brighten our homes to the displays on our phones.
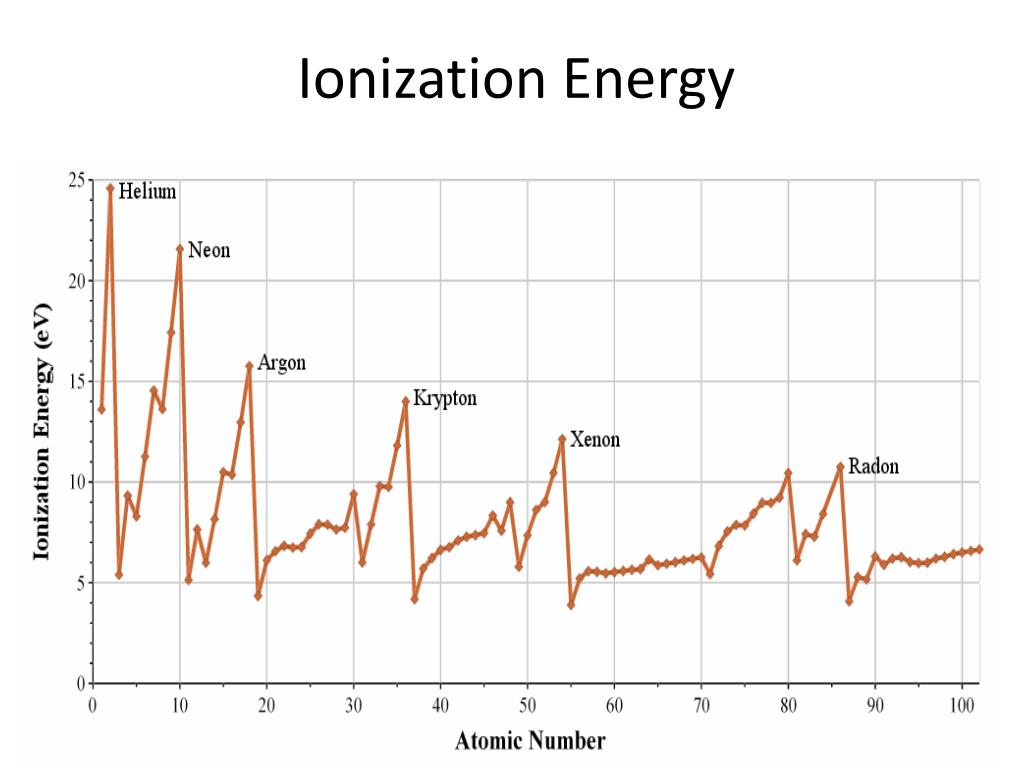
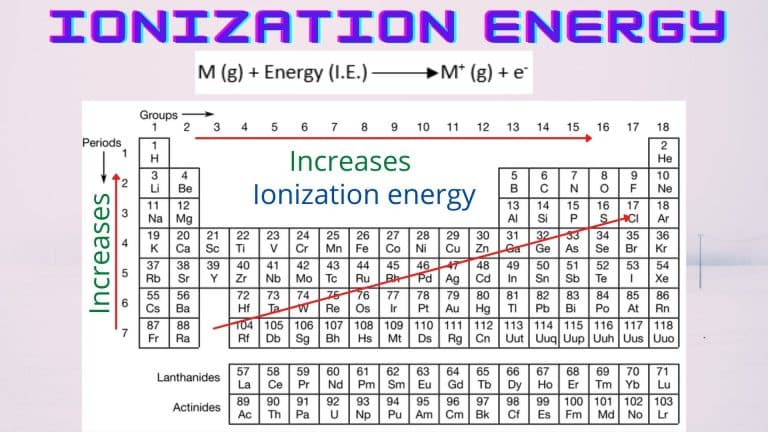
So, what exactly is ionization energy, and why should we care? In simple terms, ionization energy is the energy required to remove an electron from an atom. Imagine an atom as a tiny solar system, with electrons orbiting the nucleus. Ionization energy is like the "pull" you need to exert to get one of those orbiting electrons to break free. And the higher the ionization energy, the harder it is to snatch that electron away!
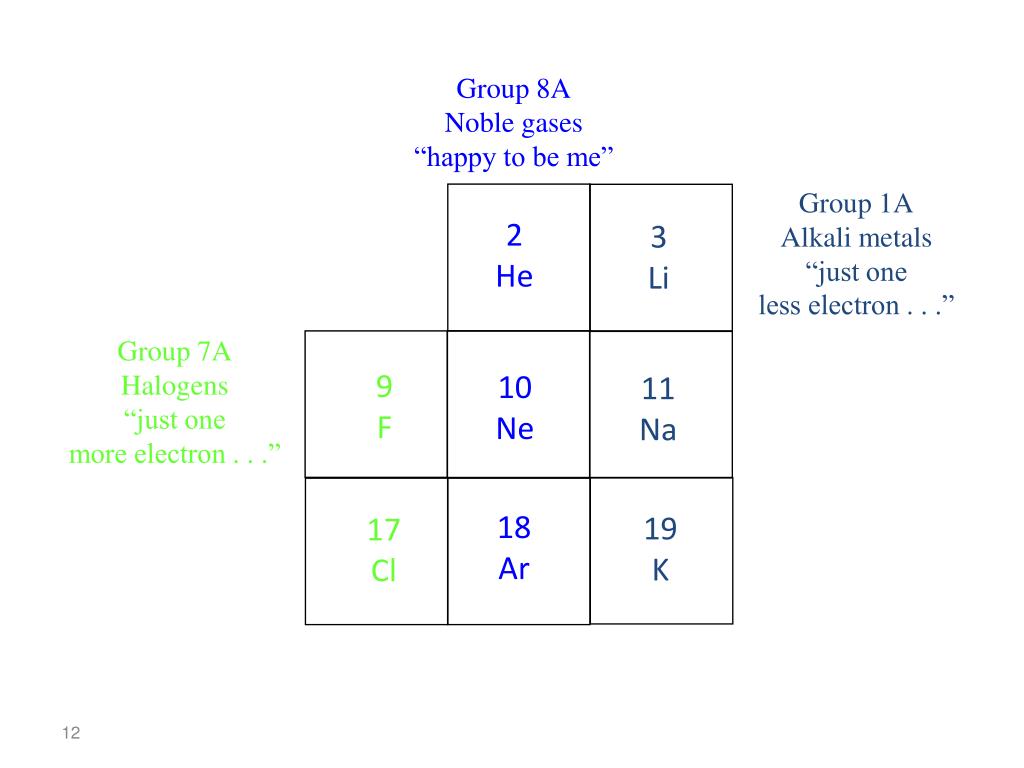
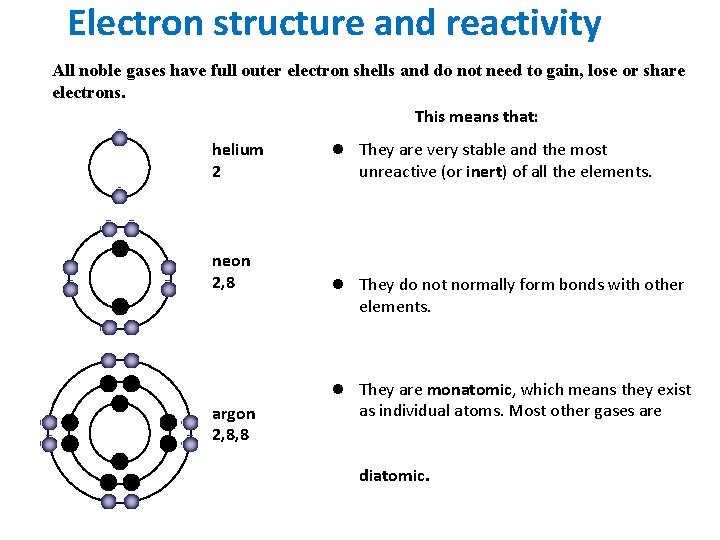
Now, back to our noble gases: helium, neon, argon, krypton, xenon, and radon. These guys are the introverts of the periodic table. They're famously unreactive, like that friend who always prefers to stay home rather than go to a party. This aloofness is directly tied to their ionization energy. Noble gases have exceptionally high ionization energies. Why? Because their outermost electron shells are completely full. This makes them incredibly stable. They don't need to gain or lose electrons to achieve stability; they already have it in spades!
Must Read
This high ionization energy is precisely why noble gases are so useful in so many applications. Take neon signs, for instance. When electricity passes through neon gas at low pressure, it excites the electrons, causing them to emit light. This is a direct consequence of neon's specific energy levels, influenced by its ionization energy. Similarly, argon is used in welding to create an inert atmosphere, preventing the hot metal from reacting with oxygen in the air. Think about the incandescent light bulbs in your home (though less common now, they are a classic example!) – they often contain argon or a mixture of noble gases to prevent the filament from burning out too quickly. Even in high-tech applications like lasers and some types of lighting in smartphones and displays, noble gases play a crucial, albeit often unseen, role.
So, how can you better "enjoy" this concept? Start by looking around you! The next time you see a glowing neon sign, appreciate the science behind it. Next time you use a light bulb, think about the invisible gas inside, contributing to its function. You can also explore online resources that explain atomic structure in a visual way. Many excellent educational websites and videos break down concepts like ionization energy with engaging animations. For a more hands-on (though carefully supervised!) approach, consider simple science kits that demonstrate gas properties. Understanding these fundamental principles gives you a deeper appreciation for the world around you, turning everyday objects into little wonders of scientific ingenuity. It’s a rewarding intellectual journey that highlights the elegance of nature's design.