Difference Between Endothermic And Exothermic

Let's talk about heat. Specifically, let's dive into the wild world of chemical reactions and how they play with temperature. It's not as scary as it sounds, I promise. Think of it like a party. Some parties give you energy, and some… well, they kind of drain it out of you.
We've got two main types of heat-loving (or heat-hating) reactions. On one side, we have endothermic reactions. And on the other, the more boisterous bunch, are the exothermic reactions.
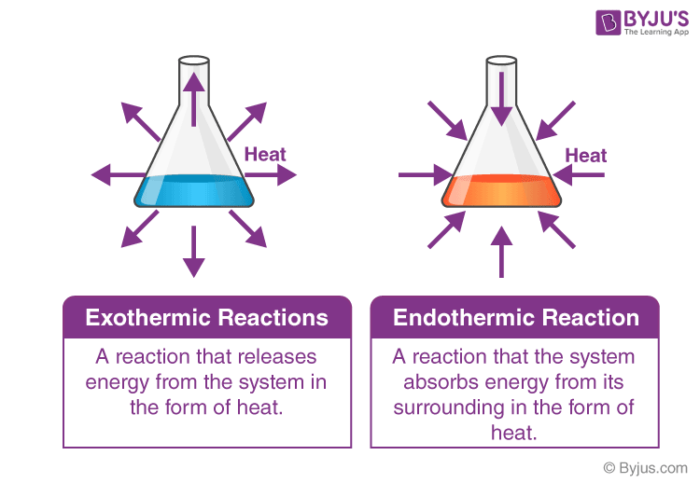
Now, I have an unpopular opinion about this. I think exothermic reactions are the life of the party. They're the ones who bring the fireworks, the loud music, and maybe even a spontaneous dance-off. They're all about giving off energy. Think of a campfire. You get close, and BAM! Warmth. That's your classic exothermic reaction. It's literally releasing heat into its surroundings. It's saying, "Here, have some warmth! You're welcome!"
Must Read
My personal favorite example of an exothermic reaction is when you strike a match. Little spark, a bit of drama, and suddenly you have a flame. That flame is pure, unadulterated energy escaping. It’s like the match is shouting, "I'm here, and I'm hot!" And honestly, who doesn't love a good, hot flame? It’s so satisfying. It’s the chemical equivalent of a perfectly timed mic drop.
Another great one? Those little hand warmers you get in the winter. You crack them open, and they start to heat up. They’re full of chemicals that are just eager to give you some cozy warmth. They’re like tiny, portable suns. They take something that’s just… existing, and they turn it into delicious, toasty heat. They’re the unsung heroes of cold fingers everywhere. I salute you, little hand warmers. You are truly exothermic champions.

Then there are reactions that feel more like… well, a Sunday afternoon nap. These are the endothermic reactions. They don't give off heat; they take it in. They're the ones that say, "Hey, can you spare some warmth? I’m feeling a bit chilly, and I need it for my grand chemical transformation." They absorb energy from their environment.
Think about an instant cold pack. You snap it, and it gets ice cold. Where did that coldness come from? It didn't magically appear. The reaction inside the pack absorbed the heat from its surroundings (including your possibly bruised knee) to make itself happen. It's like a sponge for heat. It's a little bit of a drama queen, always needing something from you to get its job done.
I find endothermic reactions fascinating, but also a little… needy. They’re like the friend who always needs a favor. "Oh, can you just turn the thermostat up? I'm trying to, like, become something new, and it requires a lot of energy." They’re not inherently bad, of course. They’re just… different. They require a purchase of energy. You have to give them something to get them going.
Imagine dissolving sugar in your tea. It’s not usually a dramatic temperature change, but some dissolutions are endothermic. The sugar molecules are getting all excited and breaking apart, and they need a little bit of heat energy from the tea to do that. The tea gets a tiny bit cooler as it fuels this sugary fiesta. It's a subtle exchange, not a fireworks show.
The big difference, in my humble, slightly biased opinion, is enthusiasm. Exothermic reactions are enthusiastic. They're bursting with energy. They’re the life of the party, the loud laugh, the one who starts the conga line. They’re putting on a show for you.
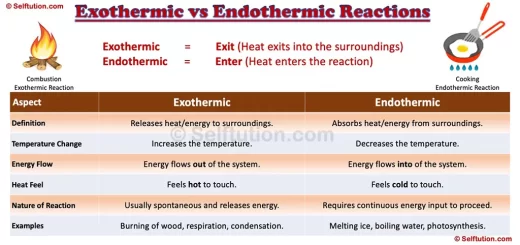
Endothermic reactions, on the other hand, are more introspective. They're the quiet observer, the one who needs a cozy blanket and a warm mug. They’re absorbing, thinking, and transforming. They require you to be the provider of the warmth, the energy source. They’re the student studying hard, needing all the brain fuel they can get.
So, next time you feel a sudden warmth or a noticeable chill from a chemical interaction, you can give a knowing nod. You're witnessing the drama of energy exchange. Whether it's a cozy campfire (exothermic!) or a surprisingly cool instant cold pack (endothermic!), these reactions are playing their part in the grand chemical play of life. And I, for one, am always rooting for the team that brings the heat.
It’s not about which is "better." It’s about their style. One is a generous giver, the other a focused receiver. And both are absolutely essential. But if I had to pick a favorite at a party? Give me the exothermic reaction every time. They just know how to liven things up!