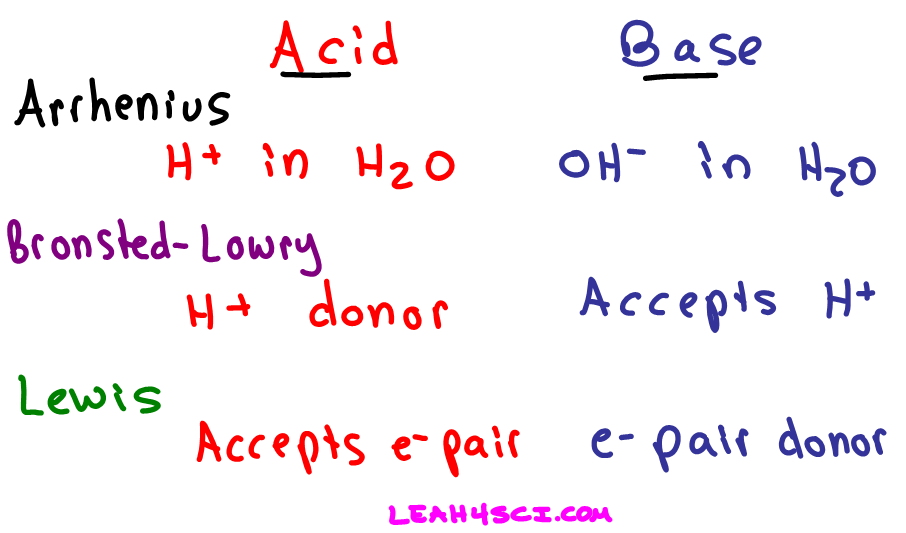Difference Between Bronsted Lowry And Lewis

Alright, so you’re at a café, right? The barista just botched your latte with about as much foam as a baby’s burp, and you’re staring into the abyss of lukewarm mediocrity. To take your mind off it, let’s dive into something… well, let’s just say it’s going to make your brain do a little jig: the difference between Bronsted-Lowry acids and bases, and Lewis acids and bases. Think of it like this: we’re upgrading our chemistry superhero costumes!
So, imagine you’re at a party. A really, really nerdy chemistry party. There are people wearing lab coats and talking about electron configurations like they’re the latest gossip. Now, our first two characters on the scene are Bronsted and Lowry. These guys are like the friendly neighborhood watch of the acid-base world. They’re all about sharing. Specifically, they’re all about sharing protons. You know, those little positively charged guys hiding in the nucleus of a hydrogen atom? They’re the hydrogen ions, H+.
According to Bronsted and Lowry, an acid is basically a proton donor. It’s that generous soul at the party who’s always offering to buy everyone a round. It’s like, “Here, have a proton! Take it, it’s yours!” They’ve got protons to spare, and they’re not afraid to hand them out like free samples at a Costco.
Must Read
And then you have the base, in the Bronsted-Lowry universe. This guy is the opposite. He’s the one who takes the proton. He’s like, “Ooh, a proton! Yes, please! I’ll take that!” He’s a proton acceptor. Think of them as collectors, but with a very specific, positively charged commodity.
So, the whole interaction is a proton exchange. It’s like a chemical handshake where a proton changes hands. For example, if you have hydrochloric acid (HCl) chilling with some water (H2O), the HCl is the acid. It’s like, “Here, water, have my proton!” The water, being a base, is like, “Yay, thanks!” and happily accepts it, forming hydronium ions (H3O+). See? Simple, clean, proton-y fun.
But wait, there’s more! Because the universe, as we know, is never content with just one way of doing things, along comes Lewis. Now, Lewis is a bit more… abstract. He’s not just looking at who’s giving away protons. Lewis is thinking about the deeper, darker, more electron-y secrets of the universe. He’s looking at electron pairs. Yes, those tiny, negatively charged particles that zip around atoms like hyperactive squirrels.

The Lewis definition is *way broader. It’s like upgrading from a bicycle to a spaceship. Lewis acids and bases don’t even need protons to get involved. They’re more interested in who’s got electron pairs to lend and who’s desperate to borrow them.
So, a Lewis acid is an electron pair acceptor. Imagine this: Lewis acid is at the party, looking around, and sees everyone with their little electron pairs, holding them tight. This guy is a bit of a social climber, or maybe just really hungry. He’s like, “Hey, you! With the extra electron pair! Can I… uh… borrow that? For a bit?” He’s looking for a lonely electron pair to cozy up with.
And the Lewis base? Well, they’re the generous souls, the electron pair philanthropists. They’re the ones with a surplus of electron pairs, just *itching to share. A Lewis base is an electron pair donor. They’re the ones who see Lewis acid looking forlorn and go, “Aw, don’t worry, I’ve got plenty of electron pairs! Here, take one!” It’s like a chemical cuddle puddle of electron pairs.

Now, here’s where it gets really interesting, and possibly a little mind-bending. Every Bronsted-Lowry acid is also a Lewis acid, but not every Lewis acid is a Bronsted-Lowry acid. It’s like saying every golden retriever is a dog, but not every dog is a golden retriever. The Lewis definition is the big umbrella, the ultimate cosmic rulebook.
Think about it. A Bronsted-Lowry acid donates a proton. That proton, remember, is just a bare H+. It’s missing an electron. So, when it’s donated, something else has to be ready to grab onto it. And what’s good at grabbing things, especially positively charged things? Something with a spare electron pair! Voila! Your Bronsted-Lowry acid is essentially setting up a situation for a Lewis base to donate an electron pair to that proton.
But then you get to the Lewis-only territory. This is where things get really wild. Take something like boron trifluoride (BF3). Now, BF3 is a perfectly respectable Lewis acid. It’s got this big ol’ empty spot in its electron shell, just begging for some electron love. It’s like a tiny, shiny vacuum cleaner for electron pairs. It’ll eagerly accept an electron pair from a Lewis base.

But does BF3 have any protons to donate? Nope. Zilch. Nada. It’s not a Bronsted-Lowry acid. It’s a Lewis acid, and a bit of a loner in the proton department. It’s like the party guest who doesn’t drink but is really good at listening and accepting compliments (electron pairs!).
Similarly, all Bronsted-Lowry bases are Lewis bases. When a Bronsted-Lowry base accepts a proton, it’s using a lone pair of electrons to form a bond with that proton. So, by accepting a proton, it’s inherently donating an electron pair. They’re basically doing double duty as proton-grabbers and electron-pair-givers.
But there are Lewis bases that aren't necessarily involved in proton exchange. They might just be really good at donating electron pairs to other things, not necessarily a proton. They’re the ones who have an abundance of electron pairs and are just looking for any electron-deficient center to bond with.

So, to recap, like a well-caffeinated marathon runner: Bronsted-Lowry is all about the proton tango. Acids give ‘em, bases take ‘em. Simple and sweet. Lewis, on the other hand, is throwing an all-out electron pair rave. Acids are the electron-pair moochers, bases are the electron-pair providers. And Lewis is the ultimate party planner, because if you can accept or donate electron pairs, you’re in!
The main takeaway? Bronsted-Lowry is a subset of the Lewis definition. Lewis is the bigger, bolder, more inclusive party. It accounts for a wider range of chemical reactions that traditional proton exchange just can’t explain. It’s like moving from understanding how to make a peanut butter sandwich to understanding the entire culinary arts!
So next time you’re at that café, staring at that sad, foamy latte, you can ponder the elegance of electron pair donation. You can think about how BF3 is out there, electron-pair-hungry, while a humble water molecule is happily playing proton diplomat. And maybe, just maybe, it’ll make your latte taste a little bit better. Or at least, give you something infinitely more interesting to think about than the questionable foam art.