Describe How Crude Oil Is Separated Into Fractions

Ever looked at a puddle of something sludgy and dark on the side of the road after a rain shower and thought, "Wow, that looks suspiciously like...stuff"? Well, that's kind of where we're heading today, but instead of road gunk, we're talking about something a whole lot more exciting: crude oil. Yep, the stuff that makes your car go zoom and your lights shine bright.
Now, this crude oil, this thick, syrupy goo, isn't exactly ready for prime time. It's like a mystery box of chemicals all jumbled up. And our mission, should we choose to accept it (and we totally should, because it’s fascinating!), is to figure out how we sort all this marvelous muck into useful bits. Think of it like a super-duper, industrial-sized sorting hat, but for oil.
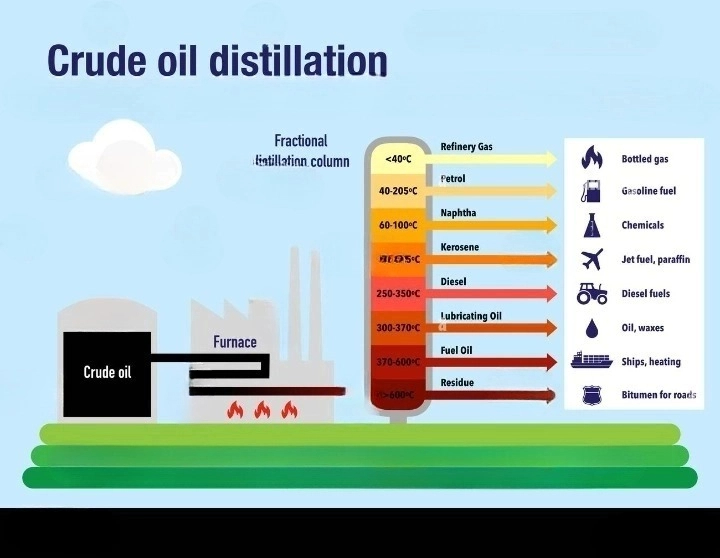
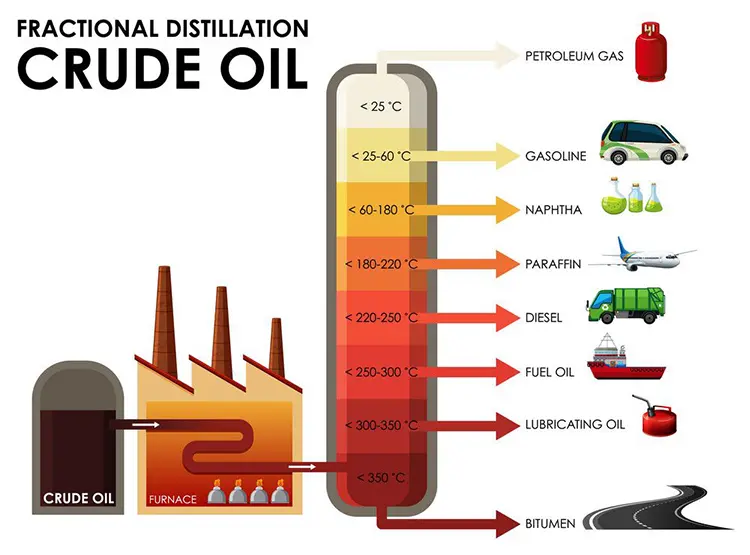
The magic happens in a place called a fractionating column. Don't let the fancy name scare you. Imagine a really, really tall building. Taller than your house. Taller than most buildings you've ever seen. And inside this skyscraper, it's all about heat. Lots and lots of heat. We're talking hotter than a dragon's sneeze on a summer day.
Must Read
First, we take our crude oil, this lovely, unrefined treasure, and we heat it up. We crank up the thermostat until it's practically boiling. Not quite boiling like your kettle, but certainly hot enough to make things… interesting. When it gets this hot, all the different bits in the oil start to get a bit antsy. They get so excited, they decide to turn into a gas. Yep, they go from a liquid to a gas, like magic, but with more industrial machinery involved.
Then, this hot, gassy mixture gets pumped into the bottom of our magnificent fractionating column. And here’s where the real fun begins. The bottom of this column is the hottest part. Naturally, right? Like the bottom of a cozy fireplace. As the gas rises up the column, it starts to cool down. Think of it like ascending a very tall, very warm mountain. The air gets cooler the higher you go.

And as these oily gases rise and get cooler, something quite wonderful happens. Different bits of the oil, the different fractions, start to decide it's time to change back into a liquid. But they don't all do it at the same temperature. It's like a group of friends at a party, and everyone has their own personal "I'm ready to sit down now" threshold.
The heavier, chunkier bits, the ones that are more stubborn and like to stick together, they cool down and turn back into liquid at the lower, hotter levels of the column. They're the first ones to say, "Okay, I'm done with being a gas, I'm going back to being a liquid." They collect in trays at the bottom. These are the things that end up being used for things like bitumen, which is what they use to pave our roads. So, the next time you're driving on smooth asphalt, you can thank these heavy-duty oil fractions for their early exit from the gaseous party.
As the remaining gas continues its journey upwards, it gets progressively cooler. And at each level, different fractions decide to condense back into liquid. It's like a series of pit stops on a very tall, very important race track. These fractions are lighter than the bitumen, and they have different boiling points. Remember those boiling points? They're like the secret handshake for each oil fraction.

Higher up the column, where it's cooler, you'll find the fractions that are lighter and more eager to become liquids. These might be things like diesel fuel, the stuff that powers trucks and buses. They condense at a slightly cooler temperature than the bitumen. They're a bit more sophisticated, you see.
Keep going up, and you’ll find even lighter stuff. Think about kerosene. This is the fuel that makes airplanes fly! Imagine that. A part of crude oil, once a gas floating up a giant metal tube, eventually helps you get to your vacation destination. It's pretty mind-boggling when you stop to think about it. Kerosene condenses at an even cooler temperature.

And if you climb all the way to the very top of the fractionating column, where it's the coolest, what do you find? You find the lightest, most nimble fractions. These are the ones that are happiest as gases for the longest. They only condense when it's really, really chilly. These are the precious gasoline, the fuel that keeps your car humming along. And even lighter than that, you have things like liquefied petroleum gas (LPG), which is used for camping stoves and heating. These are the real sprinters of the oil world.
So, in essence, this giant, hot tower is just cleverly using temperature to separate the oil into different groups, or fractions, based on how easily they turn into a gas and then back into a liquid. It’s a brilliant, if slightly steamy, way to turn a messy blob of crude oil into a whole pantry of useful products. Who knew that such a humble, albeit messy, origin could lead to such a diverse range of everyday essentials?
It's like a natural sorting machine, working tirelessly to give us the fuels we need. And all thanks to a tall tower and some very precise temperature control. So next time you fill up your car, or see a plane in the sky, or even just boil water on your gas stove, give a little nod to the humble fractionating column. It’s doing some seriously unglamorous, yet utterly essential, work.