Copper Has Two Naturally Occurring Isotopes

Copper. Just the word itself conjures images of ancient artifacts, gleaming cookware, and perhaps even the faint scent of old pennies. This versatile metal has been a staple in human endeavors for millennia, not just for its practical applications but also for its undeniable artistic charm. From intricate jewelry to grand architectural features, copper possesses a warmth and a unique character that has captivated creators and enthusiasts alike.
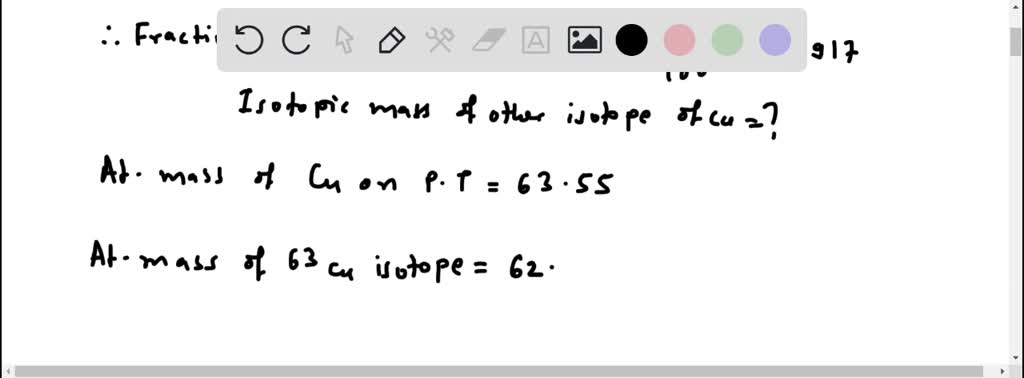
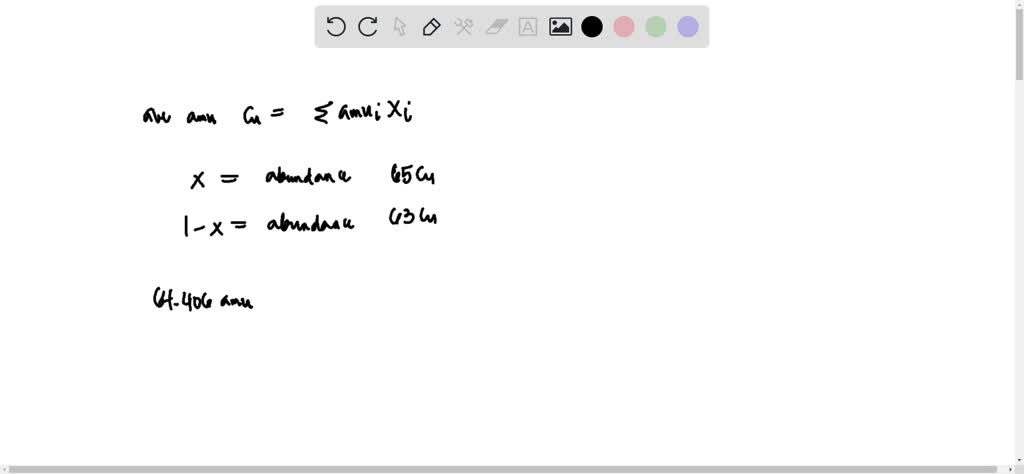
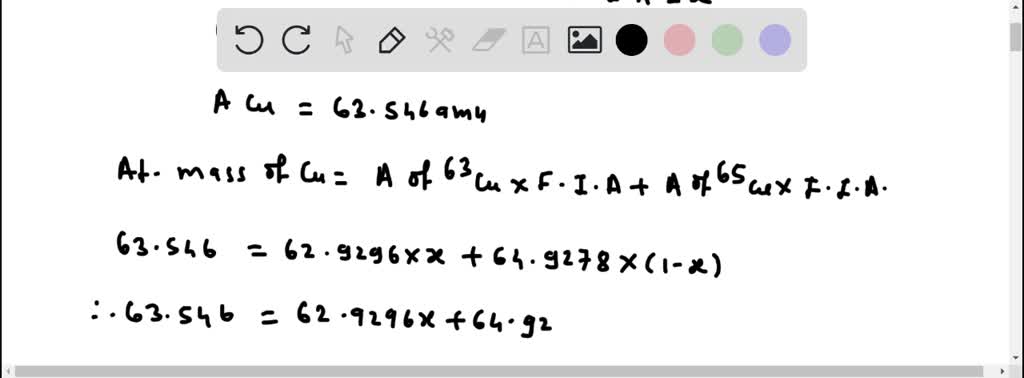
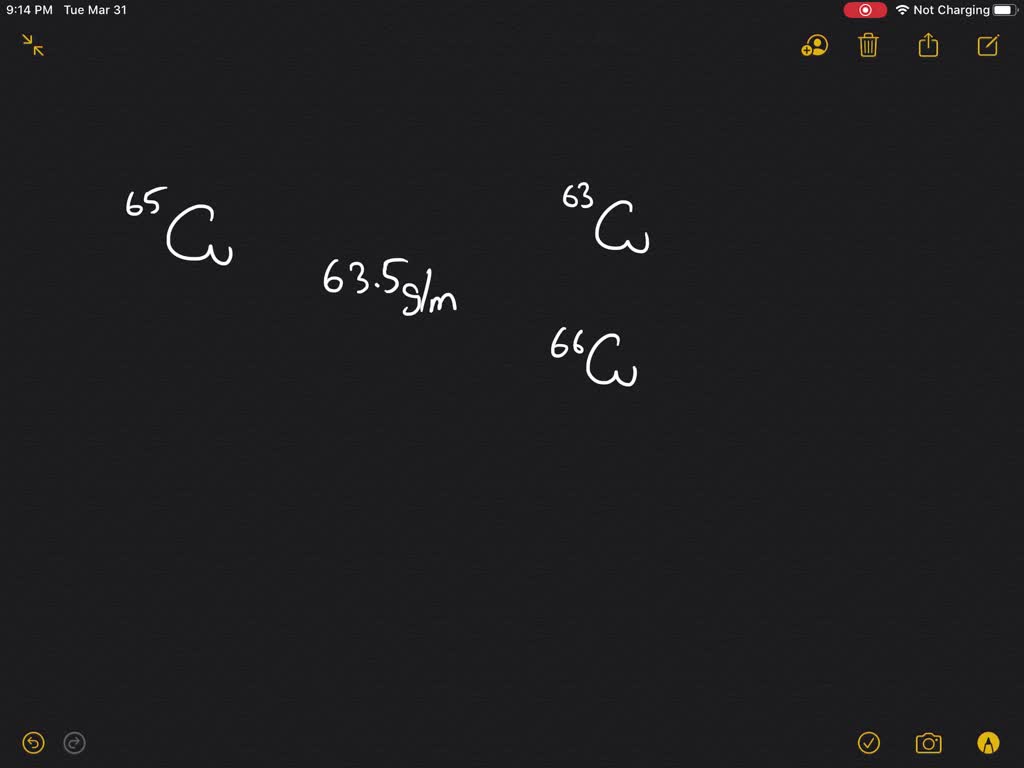
But did you know that this beloved metal isn't just one thing? It’s actually a bit of a two-for-one deal! Copper boasts two naturally occurring isotopes: copper-63 and copper-65. While this might sound like a science lesson, for artists, hobbyists, and even the casually curious, it’s a subtle secret that adds to copper's fascinating story and its creative potential. Understanding this duality, even on a basic level, can spark new ideas and appreciation for this enduring material.
For artists and crafters, this isotopic whisper can be a source of inspiration. While the difference isn't visually apparent in the finished product, knowing that the copper you're working with is a blend of these two stable siblings can add a layer of depth to your creative process. Imagine the ancient Egyptians, painstakingly crafting intricate bronzes and tools from copper – they were unknowingly working with this very same isotopic makeup! It connects us to a long lineage of makers. Hobbyists might find themselves drawn to copper for its beautiful patina that develops over time, a transformation that is both unpredictable and utterly captivating, regardless of which isotope is slightly more dominant. For casual learners, the mere fact that an element we encounter so often has such a neat scientific characteristic is a delightful discovery, proving that learning can be found in the most unexpected places.
Must Read
Consider the stunning array of copper creations: vibrant, hammered bowls that seem to glow from within; delicate filigree earrings that catch the light with every movement; even the iconic Statue of Liberty, slowly developing her signature green verdigris, a testament to copper’s enduring interaction with the environment. Whether it’s the bold statement of a copper sculpture or the subtle sheen of a handmade cuff bracelet, the beauty of copper is undeniable. You might see it incorporated into mixed-media art, providing a warm, earthy contrast to other materials, or used in rustic home decor, lending a touch of timeless elegance.

Thinking of dabbling with copper yourself? It's more accessible than you might imagine! You can find copper sheets and wire at most craft stores. For beginners, working with copper wire to create simple jewelry pieces like bracelets or keychains is a fantastic starting point. Try simple bending and shaping techniques using pliers. If you're feeling more adventurous, copper sheets can be cut and hammered (with appropriate safety gear, of course!) to create decorative elements. And don't be afraid of the patina! A little bit of natural oxidation can add wonderful character. There are also techniques like liver of sulfur (use with ventilation!) to artificially age copper for an instant antique look.
What makes working with or simply appreciating copper so enjoyable is its inherent warmth, its malleability, and its remarkable journey through time and science. Knowing that even something as elemental as its composition is a team effort between two isotopes only adds to its fascinating allure. It’s a metal that’s both ancient and ever-evolving, offering endless possibilities for creation and discovery.