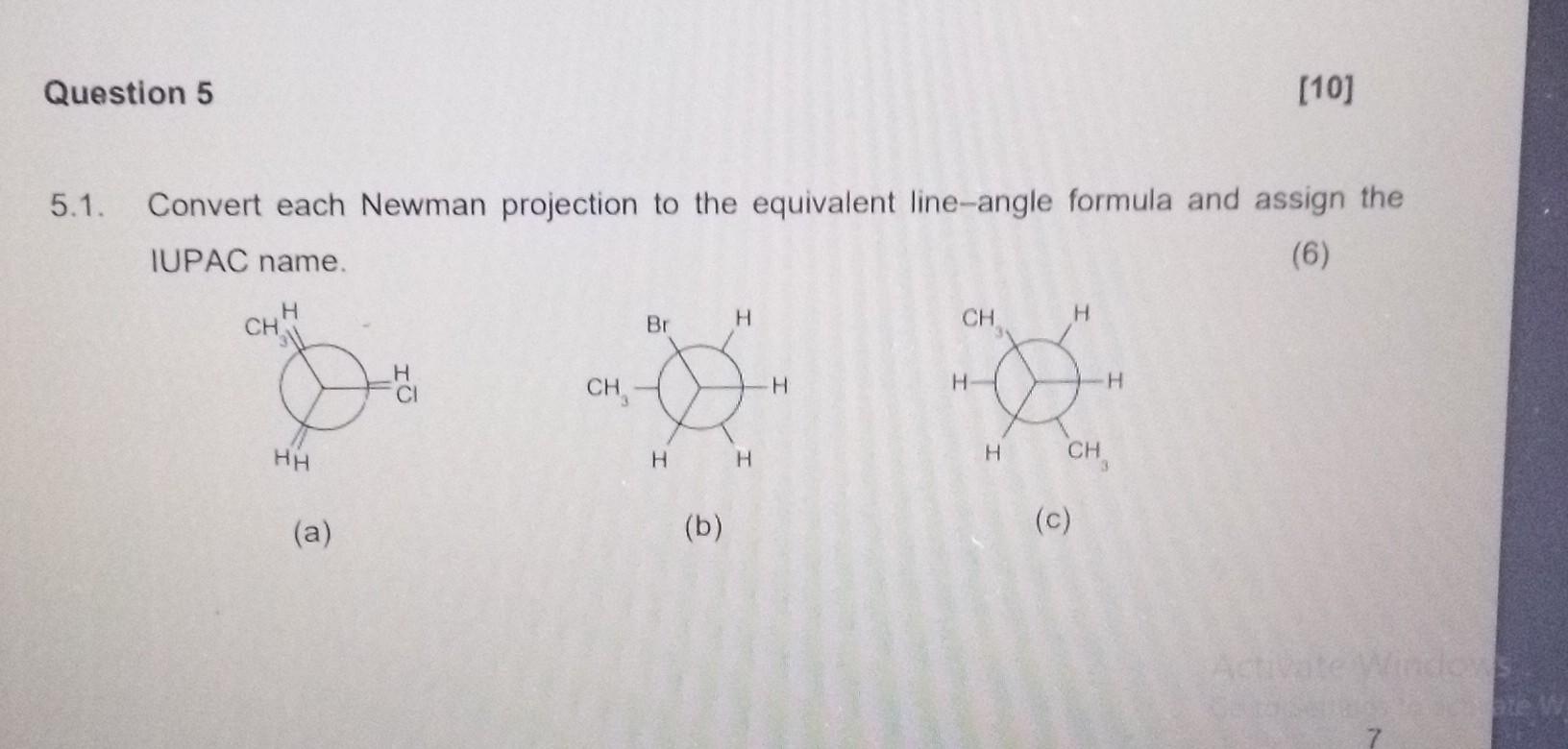
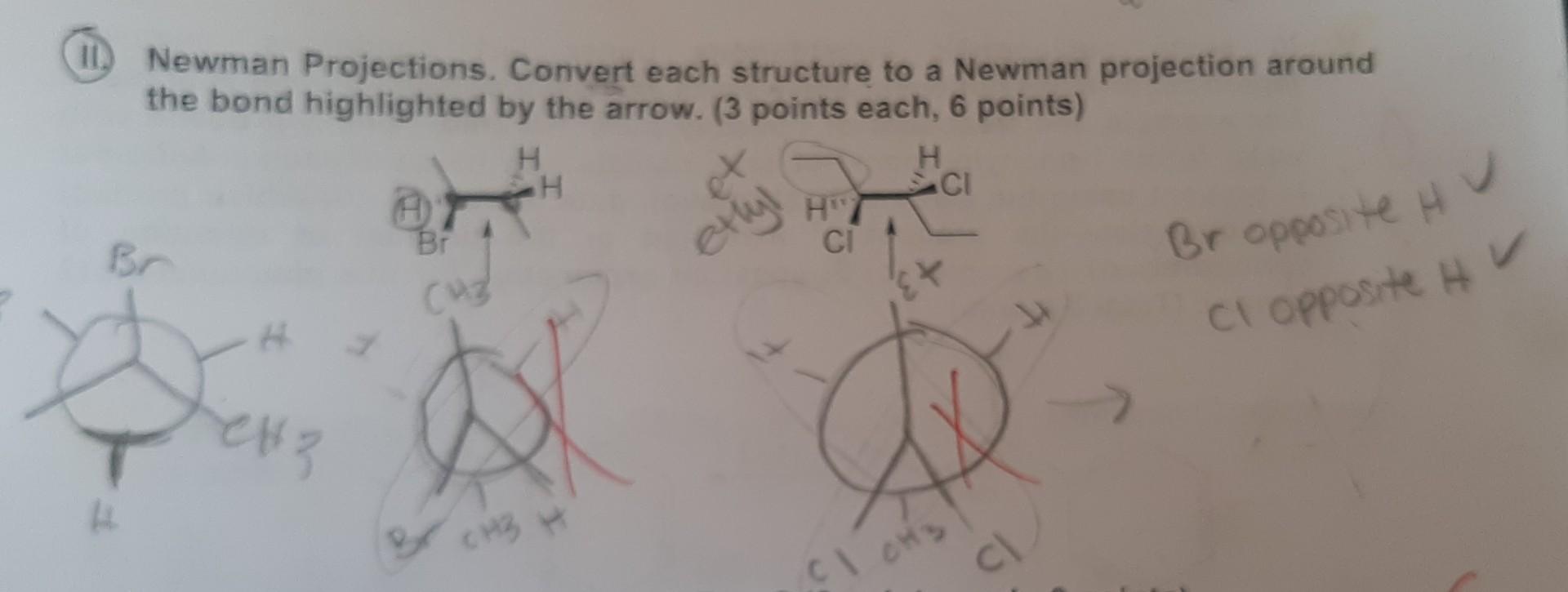
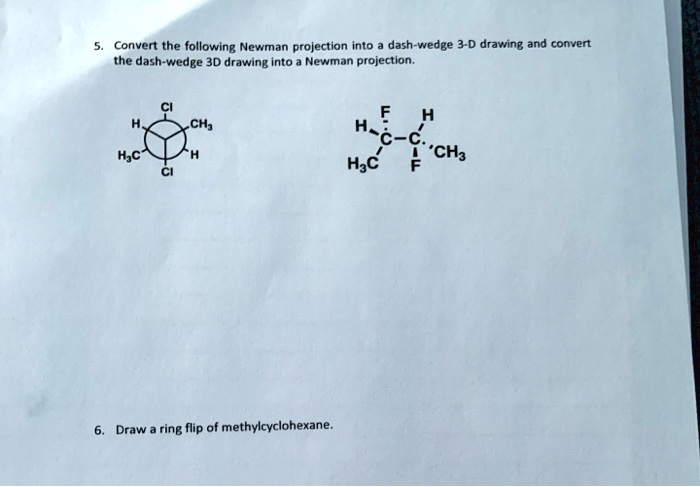
Convert Newman Projection To Line Drawing
Imagine you're a detective, and your mission is to crack the case of the squiggly lines and dots that chemists use to draw molecules. These aren't just random doodles; they're like secret codes that tell us how atoms are arranged in space. Today, we're going to learn how to translate one of these codes, the Newman projection, into a more familiar drawing style: the humble line drawing. It's like learning to read a secret diary and then rewriting it in a language everyone can understand!
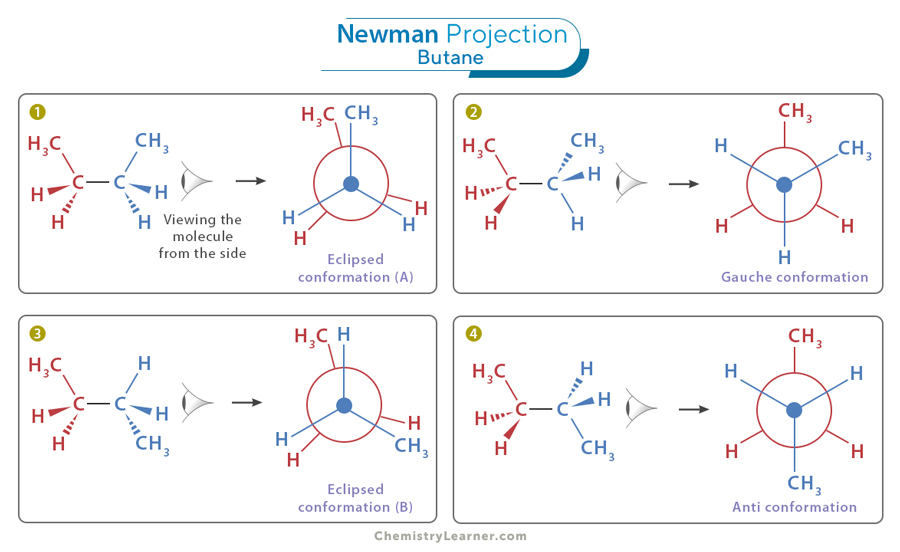
Think of a Newman projection as a very specific way of looking at a molecule – right down the middle of a bond, like peering through a telescope. You see a dot in the front, representing the atom closest to you, and a circle behind it, representing the atom further away. Attached to these are the other atoms or groups of atoms, like little arms sticking out. It’s like looking at two people standing one behind the other, and you can see the shoulders and heads of both. Kind of quirky, right? It’s a bit like trying to explain how you tied your shoelaces by looking directly at the knot from above.
Now, the line drawing is the more common way you see molecules drawn. It’s like a shorthand, where lines represent bonds between atoms, and the corners and ends of these lines are actually atoms. We usually leave the carbon atoms (the building blocks of most organic molecules) unlabeled, and any hydrogens attached to them are also invisible unless they're attached to something other than carbon. It's like a sketch artist’s quick impression rather than a detailed portrait. It’s efficient and, dare I say, a little bit chic in its simplicity.
Must Read
So, how do we make the leap from the intense gaze of the Newman projection to the elegant simplicity of the line drawing? It’s all about perspective and a little bit of spatial reasoning. Think of it as untangling a slightly messy kite string. The Newman projection shows you the twist and turns of that string from a very particular angle.
Let’s take a simple example. Imagine a Newman projection where you see a dot with three arms and a circle behind it with three arms. In the line drawing, you're going to focus on the central bond that the Newman projection is showing you. That bond becomes one of the lines in your line drawing. The atom represented by the dot in the Newman will be one end of that line in your line drawing. And the atom represented by the circle will be the other end. It’s like taking the two main points in your telescope view and connecting them with a single stroke.

It's kind of like a magic trick. One moment, you're staring down a microscopic tunnel, and the next, you've conjured a familiar, skeletal sketch!
Now, what about those little arms sticking out from the dot and the circle? These are the atoms or groups of atoms attached to your central bond. In the line drawing, these will become the other lines branching off from the ends of your central line. The key is to preserve the spatial arrangement. If an arm is pointing up on the dot in your Newman, it’ll likely be pointing up on the corresponding atom in your line drawing. If an arm is pointing down and to the left on the circle, you’ll draw a line pointing down and to the left from that atom in your line drawing. You’re essentially unfolding the view. It’s like taking a folded piece of origami and carefully flattening it out to see the whole shape.
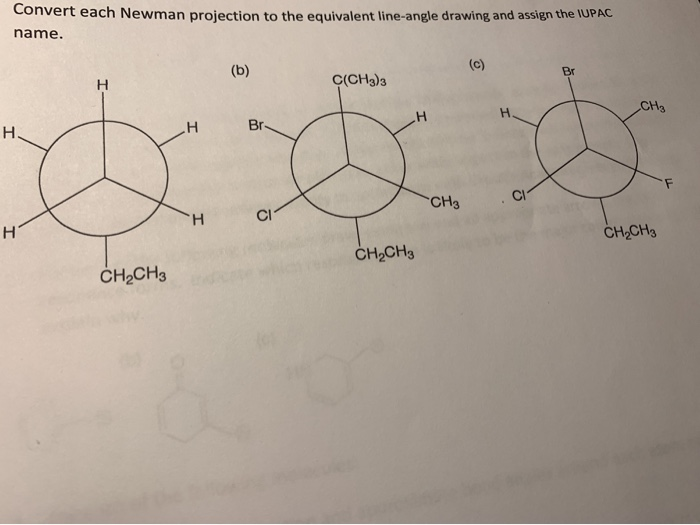
Sometimes, you might have a group of atoms, like a methyl group (which is a carbon with three hydrogens, often written as -CH3), attached to the central atoms. In a Newman projection, this might look like a single arm. In a line drawing, this -CH3 group is usually just represented by a single line extending from the atom it’s attached to. The carbon of the methyl group is at the end of that line, and the hydrogens are implied. It’s like saying, "This is a standard attachment, no need to draw every single detail."
The real fun comes when you have different "views" of the same molecule. A molecule can be rotated around bonds, and each rotation gives a different Newman projection. This is where the "fun" aspect really kicks in. It’s like having a 3D puzzle and turning it this way and that to see all its facets. Each Newman projection is just one snapshot, and by translating it to a line drawing, you’re creating a more universal representation. You’re not losing information; you’re just changing the language.
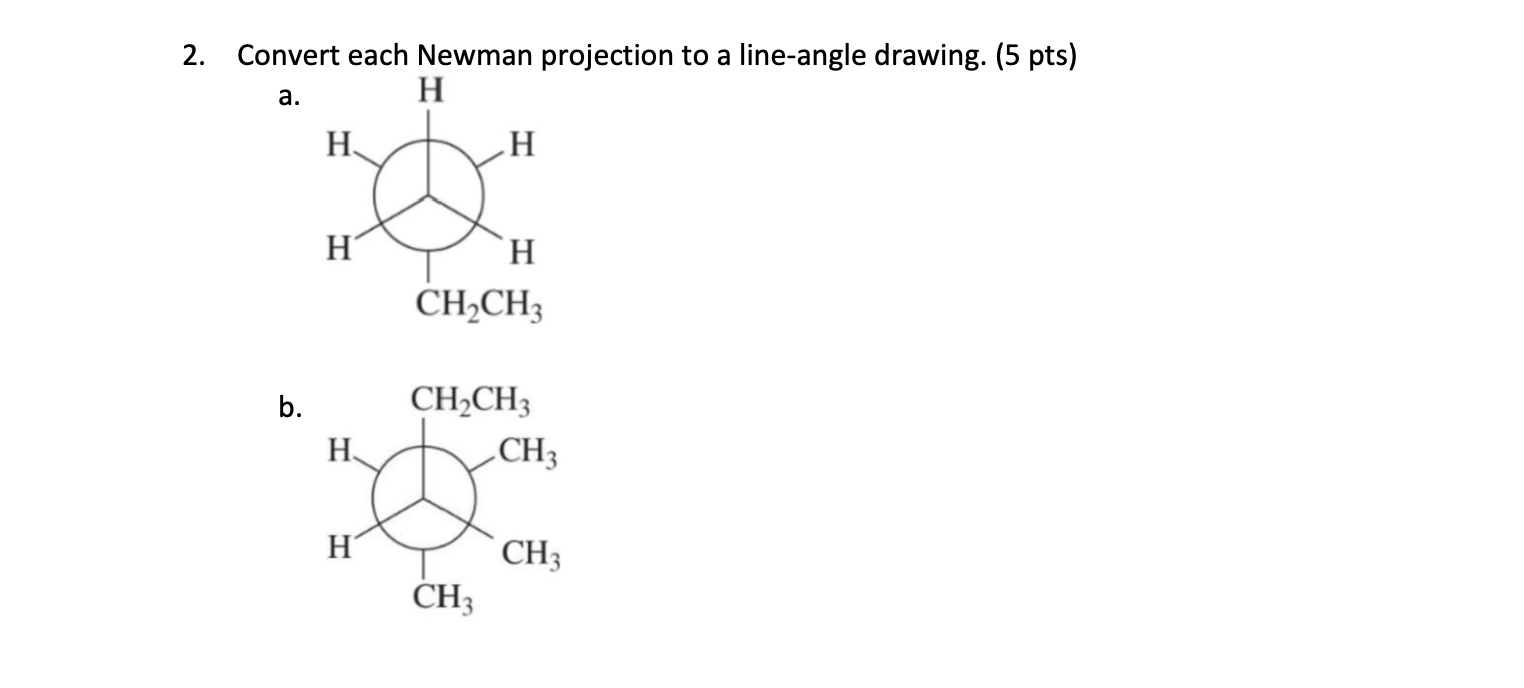
Think of the Newman projection as a very polite, formal introduction. It tells you exactly who’s where, and in what precise orientation. The line drawing, on the other hand, is like a casual nod from across the room. It gets the point across efficiently, and if you’re familiar with the people (or atoms), you know exactly who’s involved. It's less about the specific dance steps and more about recognizing the dancers and their general positions on the floor.
So, next time you see a Newman projection, don’t be intimidated by the dot and the circle. See them as your guideposts. Remember that the arms are your clues to where everything else is. And with a little bit of practice, you’ll be able to transform those sometimes-confusing views into the clean, crisp line drawings that are the everyday language of chemistry. It’s a skill that unlocks a deeper understanding, like finally figuring out the secret handshake. And who doesn't love a good secret handshake?