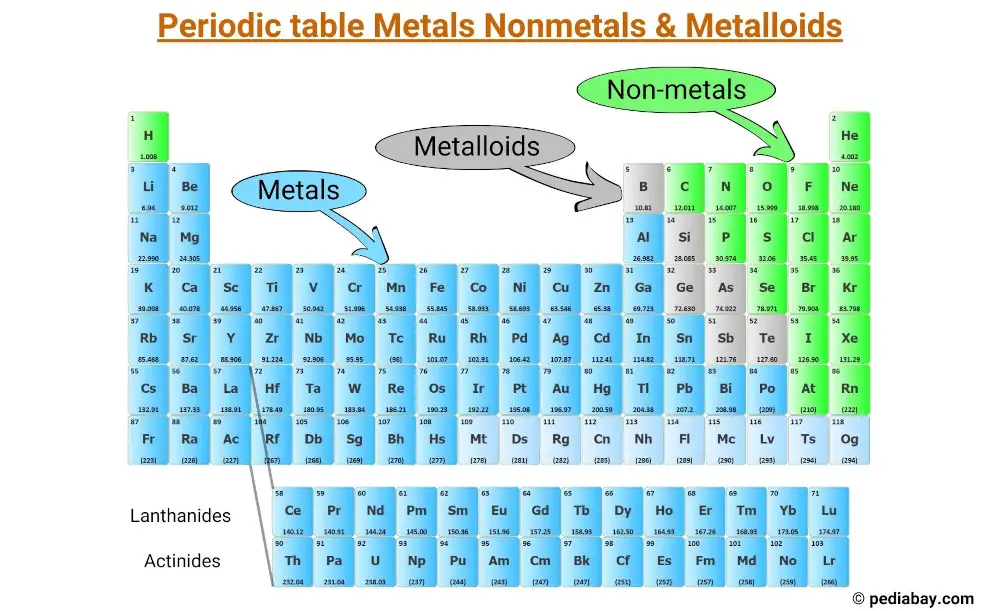
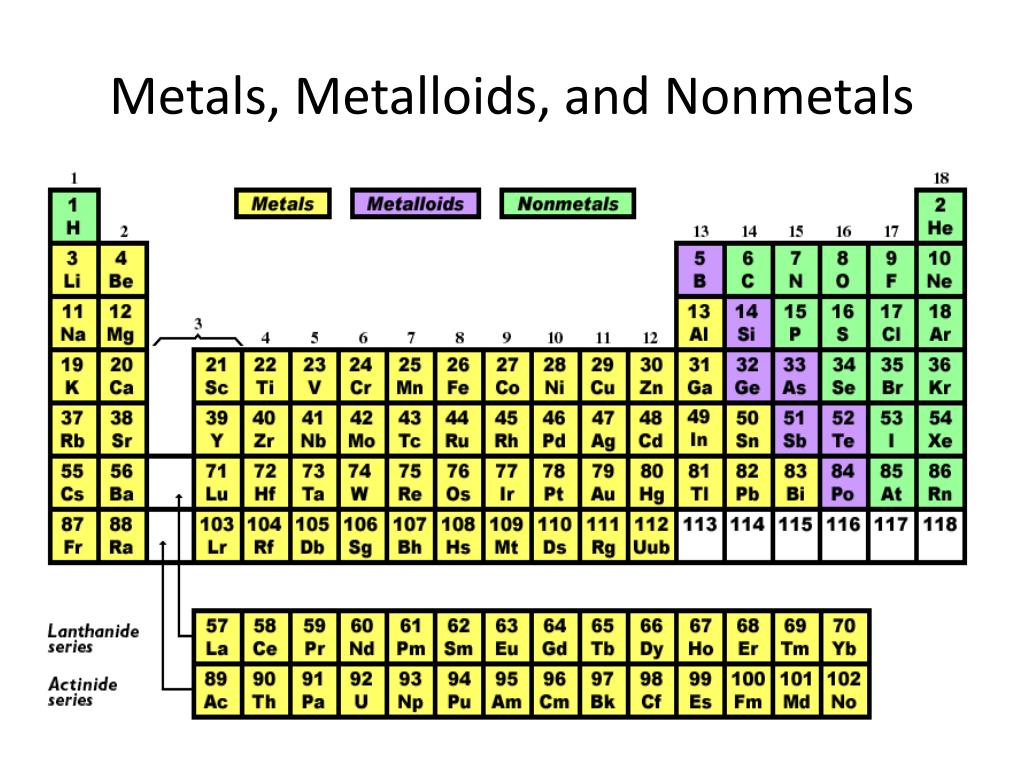
Compared To Nonmetals Metals Tend To Have
:max_bytes(150000):strip_icc()/metals-versusnonmetals-608809-v3-5b56348946e0fb0037001987.png)
You know, there are a lot of things out there that are kind of like people. Some are loud and boisterous, others are quiet and reserved. Some are shiny and get all the attention, while others are happy just doing their own thing in the background. Well, in the world of science, we've got a similar kind of vibe going on with our elements – the building blocks of everything! We’ve got the metals and the nonmetals, and they are, to put it mildly, chalk and cheese. And let me tell you, when you compare these two groups, the metals really tend to have a personality all their own, and it’s mostly the fun, flashy kind!
Think about it. When you picture a metal, what comes to mind? Probably something that gleams, right? Like a knight’s armor, a shiny new car, or even just the spoon you stir your coffee with. That’s one of the first big things metals tend to have that their nonmetal pals often don’t: a fantastic shine. Nonmetals? Well, some of them are gases, which you can’t even see! Others are… let’s just say they’re more like dull rocks or powdery substances. Not exactly red-carpet material, are they? But metals? Oh, they’re the divas of the element world, always ready for their close-up, reflecting light like they’ve just stepped out of a Hollywood studio.
But it's not just about looking good. Metals are also the strong, silent types when it comes to getting work done. Ever tried to build something substantial without metal? Good luck with that! Metals tend to be incredibly strong and durable. They can hold up skyscrapers, make sturdy bridges, and even form the very bones of our machines. Nonmetals are often brittle or just… well, not built for heavy lifting. Imagine trying to make a frying pan out of something like sulfur. You’d be scraping dust into your eggs! Metals, on the other hand, are the reliable workhorses. They’re the ones you can count on to not fall apart under pressure. They’ve got grit!
Must Read
And then there’s the way they behave with heat and electricity. Metals are like super-highways for these things. They’re excellent conductors. That’s why all your electrical wires are made of metal, like copper or aluminum. They let the electricity zoom through them without a fuss. And heat? They’re just as enthusiastic! Stick a metal rod in a fire, and the whole thing gets hot, fast. Nonmetals? Not so much. Many are insulators, meaning they’re quite happy to keep heat and electricity trapped, like a cozy sweater. This is super useful, of course – you don't want your oven mitts to conduct heat straight to your hands! But it’s just another way metals stand out: they’re the energizer bunnies of the element world, always ready to pass things along.

Metals love to share their energy, while many nonmetals prefer to keep it to themselves. It’s like a party where the metals are handing out snacks to everyone, and the nonmetals are hoarding them!
Let’s talk about shape. Metals are wonderfully malleable and ductile. What does that mean in plain English? It means you can hammer them into thin sheets (think of those beautiful tin foil rolls!) or stretch them into long, thin wires. They’re like the Play-Doh of the element world, but way, way tougher. Try doing that with a piece of coal, a nonmetal. You’ll just get dust. This amazing ability to be shaped is what allows us to create so many incredible things, from delicate jewelry to massive ship hulls. Metals are the ultimate shapeshifters, bending to our will to create whatever our imaginations can conjure.

Even the way they interact with other elements tells a story. Metals tend to be generous. They’re often happy to give away little bits of themselves (electrons, to be technical, but let’s not dwell on that!). This makes them eager to join up with other things and form new compounds. Think of how iron rusts – it’s just happily combining with oxygen. Nonmetals, on the other hand, are a bit more… grabby. They like to take those bits from the metals. It’s a bit of a dance, a push and pull, but metals are often the initiators, the ones saying, "Hey, let's make something new together!" They're the social butterflies of the periodic table.
And here’s a heartwarming thought: metals are the backbone of so much that we love and rely on. The very tools that allow us to create art, build homes, and connect with each other are often made of metal. From the sturdy frame of your bicycle to the intricate workings of your smartphone, metals are there, quietly doing their job, being strong, shiny, and conductive. They’re the unsung heroes, the reliable friends who are always ready to lend a hand (or in this case, a sturdy atom!). So next time you pick up something made of metal, give it a little nod. It’s not just an object; it’s a testament to the wonderful qualities that metals tend to have, qualities that make our lives easier, brighter, and a whole lot more interesting.