Common Ion Effect On Acid Ionization Pogil
Hey there, fellow coffee enthusiast! Grab your mug, settle in. We’re about to dive into something that might sound a little science-y, but trust me, it’s actually pretty cool. Ever heard of the "common ion effect"? No? Well, buckle up, buttercup, because we’re going to chat about it, specifically when it comes to acids and their ionization. Think of it like this: we’re peeling back a little layer of chemistry, and it’s not as scary as it sounds. Promise!
So, you know how acids, like the ones in your lemon juice or that fizzy drink you love, love to dissociate? That’s just a fancy word for breaking apart into ions, right? They give up a hydrogen ion (H+) and become something else. It’s their whole jam. It’s how they do their acidic thing. But what happens when you, I don’t know, mess with the equilibrium? That’s where our buddy, the common ion effect, waltzes in. And it’s a bit of a drama queen, honestly.
Imagine you’ve got a perfectly balanced acid, just chilling in its water. It’s happily dissociating, making some H+ ions and some anion buddies. Everything is groovy. Then, BAM! You throw in another chemical that already has one of those ions. Like, if your acid breaks into H+ and A-, and you add a salt that’s got A- in it. Uh oh. Suddenly, there’s a party, but it’s a party with too many of the same people.
Must Read
This is where Le Chatelier’s principle, another one of those fancy chemistry names, really shines. Remember that? It’s basically the universe’s way of saying, “Hey, if you mess with something, it’s gonna try to push back!” So, if you’ve suddenly got a ton of A- ions hanging around, the acid is going to be like, “Whoa, hold up. Too much A- already. I’m not gonna make any more of that.” It’s like trying to sell ice cream on a freezing day. Nobody’s buying!
So, what does the acid do? It chills. It becomes less eager to dissociate. Its ionization goes down, down, down. It’s like it’s saying, “You know what? I’m good. I’ve got enough friends here already.” This is the common ion effect in action, my friends. It’s basically an ion, common to both the acid and whatever else you added, messing with the acid’s equilibrium. Sneaky, right?
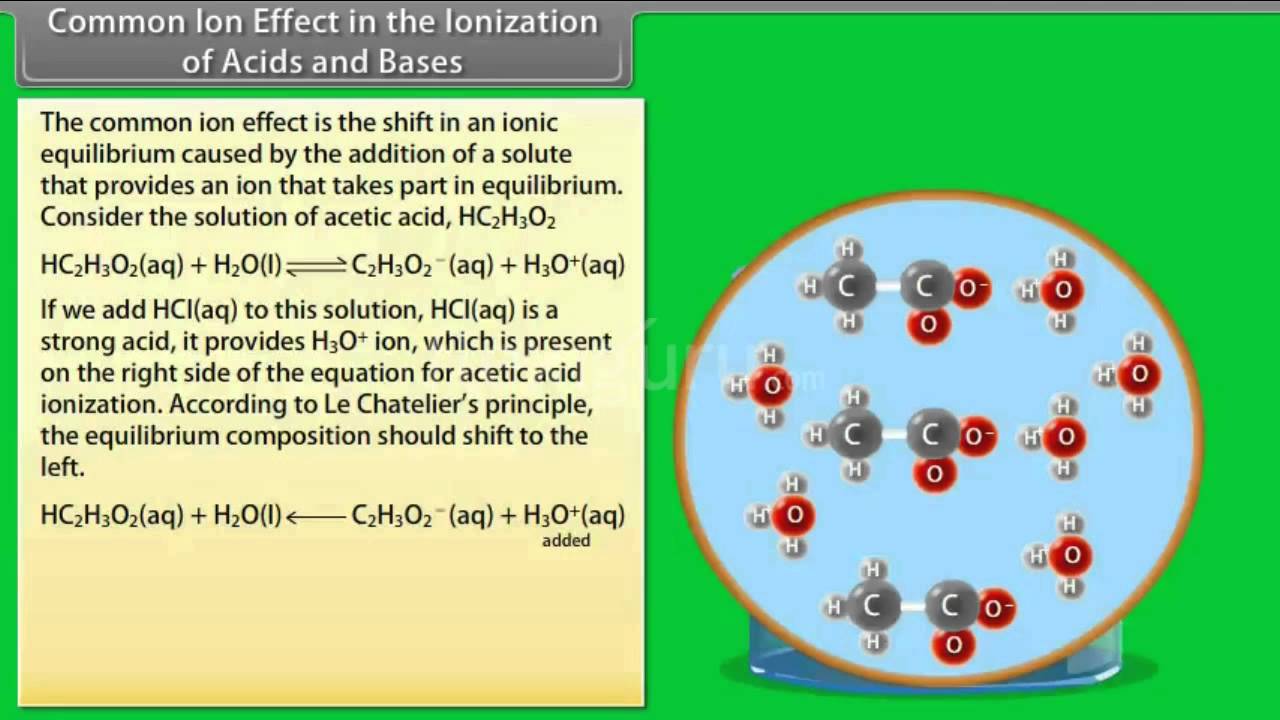
Think about acetic acid, the stuff in vinegar. It’s a weak acid, meaning it doesn’t break apart all the way. It’s got this equilibrium going on: CH3COOH (acetic acid) <=> H+ + CH3COO- (acetate ion). Pretty straightforward. Now, what if you add sodium acetate (CH3COONa) to your vinegar? Sodium acetate is a salt, and it dissociates completely. So, it’s like, “Here’s some Na+ and here’s a whole boatload of CH3COO-!”
Suddenly, the concentration of acetate ions (CH3COO-) in the solution shoots up. Remember our equilibrium? The poor acetic acid is looking at this massive influx of its own anion (acetate) and is like, “Seriously? I just made some of you! Now you’re here in such huge quantities?” Le Chatelier’s principle kicks in with a vengeance. The system wants to relieve that stress of too much product (acetate). So, the equilibrium shifts backwards.

Instead of the acetic acid happily giving up more H+ ions and acetate ions, it’s now more likely to grab an H+ ion and reform the acetic acid molecule. It’s like a reverse party. Less dissociation, more recombination. The concentration of H+ ions in your vinegar solution actually decreases because of this. And what does a decrease in H+ ions mean? A decrease in acidity! Your vinegar is suddenly less…vinegary. Who would have thought?
The POGIL Way of Looking at It
Now, you might be thinking, “Okay, this is kind of neat, but why the POGIL part?” Ah, POGIL. Process-Oriented Guided Inquiry Learning. It’s this awesome way of learning science where you’re not just told stuff, you figure it out yourself. You work through activities, look at data, and make your own conclusions. It’s like a detective story for your brain!
In a POGIL activity about the common ion effect, you wouldn't just read this article (sadly for me, but good for you!). You’d probably be given some scenarios. Maybe some data tables showing pH values before and after adding a common ion. You’d be asked questions like, “What happened to the pH?” or “Why do you think the pH changed?” You'd be guided to see the pattern, to observe the decrease in ionization, and to eventually arrive at the concept of the common ion effect yourself.
It's all about building that understanding from the ground up. They might give you the equilibrium expression for a weak acid, then show you what happens when you add a salt of its conjugate base. You’d then use that data to deduce the shift in equilibrium. It's super empowering, really. You're not just memorizing a definition; you're understanding the 'why' behind it.

Imagine a POGIL worksheet. It’d start with simple things. Like, what does it mean for an acid to be “weak”? What does dissociation look like on paper? Then, it ramps up. You’d have diagrams showing equilibrium, arrows indicating shifts. It’s all very visual and interactive, even if it’s just on paper. They want you to think like a chemist. To hypothesize, to test, to refine your ideas.
Why Does This Even Matter? (Besides Making My Coffee Less Strong)
So, beyond your morning caffeine fix, why should you care about the common ion effect? Well, it has some huge implications in the real world. Think about buffers. You know, those solutions that resist changes in pH? Buffers are built on the common ion effect! They're usually made from a weak acid and its conjugate base (or a weak base and its conjugate acid).
Because you have both the acid and its conjugate base (the common ion!) present in high concentrations, the solution can absorb small amounts of added acid or base without a drastic pH change. It’s like a pH shock absorber! If you add H+, the conjugate base grabs it. If you add OH-, the weak acid neutralizes it. Super clever, and totally thanks to our common ion friend.
Your blood, for instance, has a sophisticated buffering system. It needs to maintain a very stable pH to keep you alive and kicking. Without the common ion effect and buffering systems, even a little bit of metabolic activity could send your pH spiraling. That's a quick trip to…well, not good places.

And what about chemical analysis? If you’re trying to precipitate out a specific metal ion, you might adjust the pH using a common ion to minimize the solubility of other metal ions. It’s all about controlling those equilibrium reactions. It’s a subtle but powerful tool in a chemist's toolkit.
Let’s get a little more technical, shall we? Remember that acid dissociation constant, Ka? It’s a measure of how strong an acid is. For a weak acid HA, the equilibrium is HA <=> H+ + A-. So, Ka = ([H+][A-])/[HA]. Now, if you add a common ion, say by adding a salt containing A-, the concentration of A- goes up. To keep Ka constant (it's a constant, after all!), the concentrations of H+ and HA must adjust. And guess what? The [H+] concentration goes down, and the [HA] concentration goes up. The equilibrium shifts left, just like we said!
This means that the effective strength of the acid decreases. It’s acting even weaker than it already was. It’s like it’s been demoted. Poor little acid.
Practical Examples: Beyond the Beaker
So, where else do we see this? Think about solubility of salts. Let's say you have calcium carbonate (CaCO3). It dissolves a bit in water: CaCO3(s) <=> Ca2+(aq) + CO32-(aq). Now, what if you add a solution that already has a bunch of CO32- ions in it, like sodium carbonate (Na2CO3)?

That extra CO32- is a common ion! According to Le Chatelier’s principle, the equilibrium will shift to the left, meaning less CaCO3 will dissolve. The solubility of CaCO3 goes down. This is super important in things like water treatment, where you might want to precipitate out certain minerals. Or, conversely, if you want something to dissolve, you’d want to avoid adding common ions!
It's all about manipulating those equilibrium constants. It’s like a chemical dance, and the common ion effect is the choreographer, telling everyone where to move. It’s not just some abstract concept in a textbook; it’s happening all around us, influencing everything from the stability of our biological systems to the processes in industrial chemistry.
Let’s think about it in terms of POGIL again. They might show you a solubility product constant (Ksp) for a salt. Then, they’ll present you with data where that same salt is added to a solution containing one of its ions. You’d observe the decreased solubility, and then you'd connect it back to the common ion effect and the principle of equilibrium shift. It’s a beautiful, interconnected web of chemical concepts!
And sometimes, it's just about making sure your lemonade isn't too tart. Or that your fizzy drink retains its sparkle. It's the unseen forces of chemistry, working their magic. So next time you're sipping on something acidic, or just thinking about how stable your body’s pH is, give a little nod to the common ion effect. It’s a true unsung hero of the chemical world.
Honestly, it’s the little things, right? The seemingly small interactions that have such a big impact. The common ion effect is definitely one of those. It’s a testament to how interconnected and responsive chemical systems are. And understanding it? Well, that just makes you a little bit more of a chemistry rockstar. So, keep those coffee cups full, and keep those curious minds buzzing. There's always more cool stuff to explore!





