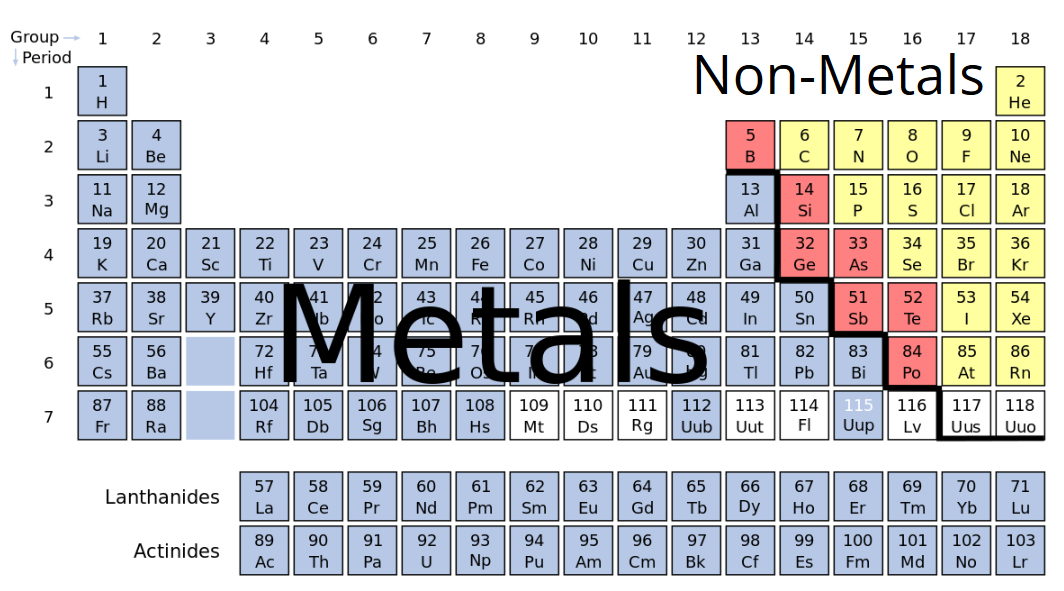
Classify Each Element As A Metal Or Nonmetal

Ever looked at a chart of the elements and wondered, "What's the deal with all these boxes?" It's like a giant cosmic recipe book, right? And within that book, there's a super fundamental way to sort everything: into metals and nonmetals. Pretty neat, huh? It sounds simple, but understanding this basic classification really unlocks a lot of the cool stuff happening all around us.
So, why bother classifying them at all? Well, imagine trying to organize your kitchen without putting the pots with the pans and the spices with the flour. It would be chaos! The periodic table is kind of like that, but on an atomic level. Grouping elements based on their properties helps scientists (and us curious folks!) predict how they'll behave. It's like knowing your apples are fruits and your carrots are veggies – you already have a general idea of what to expect.
The Shiny and the Strong: Meet the Metals
Let's start with the crowd-pleasers: metals. Think about what comes to mind when you hear "metal." Probably something shiny, right? Like the gleaming surface of a newly polished spoon or the sturdy frame of your bicycle. That lustrous (fancy word for shiny!) quality is a dead giveaway for most metals.
Must Read
But it's not just about good looks. Metals are also incredibly conductive. What does that mean? It means they're like superhighways for electricity and heat. Think about the wires in your house that power your gadgets – that's usually copper, a metal, doing its thing. Or how your frying pan gets hot really fast when you put it on the stove. Yep, metal doing its job again!
And then there's their strength. Most metals are pretty tough. You can hammer them, bend them, or stretch them without them just snapping. That's why we use them for everything from buildings to bridges to the phone you're probably reading this on. They're generally malleable (easy to hammer into thin sheets) and ductile (easy to stretch into wires). Pretty impressive resilience, wouldn't you say?
Think about gold, that super precious metal. It's shiny, it conducts electricity (though not as well as copper), and you can pound it into incredibly thin leaf. Or iron, the backbone of so much of our infrastructure. Even a common metal like aluminum, used in everything from soda cans to airplane parts, ticks all these boxes.

So, if you see an element that's shiny, good at conducting heat and electricity, and can be bent or shaped without breaking, chances are you're looking at a metal. It’s like the superhero of the element world, always ready for action and tough enough to handle it.
The Diverse and the Different: Enter the Nonmetals
Now, let's switch gears and talk about the nonmetals. These guys are the wild cards, the eclectic bunch. They don't all look the same, they don't all act the same, and they definitely aren't as uniformly shiny and strong as their metal counterparts.
Nonmetals can be solids, liquids, or even gases at room temperature. Take oxygen, which we breathe every second – that's a gas. Then there's bromine, a liquid that looks like reddish-brown goo. And don't forget carbon, a solid that can form diamonds (super hard!) or pencil lead (pretty soft!). See what I mean about variety?
And when it comes to conductivity, nonmetals are generally the opposite of metals. They're usually insulators. That means they're not great at letting electricity or heat pass through them. Think about the plastic handles on your pots and pans, or the rubber casing around electrical cords. Those are often made from nonmetals specifically because they don't want the heat or electricity to travel!
Some nonmetals can be pretty reactive, like fluorine, which is super eager to grab electrons from other atoms. Others, like helium or neon, are incredibly lazy and prefer to hang out by themselves. It's a real spectrum of personalities!
Even though they're not as flashy as metals, nonmetals are absolutely essential. They make up a huge chunk of our bodies – think carbon, hydrogen, nitrogen, oxygen. They're in the air we breathe, the water we drink, and the clothes we wear. They might not be the strong, silent type, but they're definitely the backbone of life itself.
So, if you encounter an element that's dull (not shiny), doesn't conduct electricity or heat very well, and can exist in different states of matter, it’s probably a nonmetal. They’re the creative artists of the periodic table, showing off their unique talents in all sorts of ways.

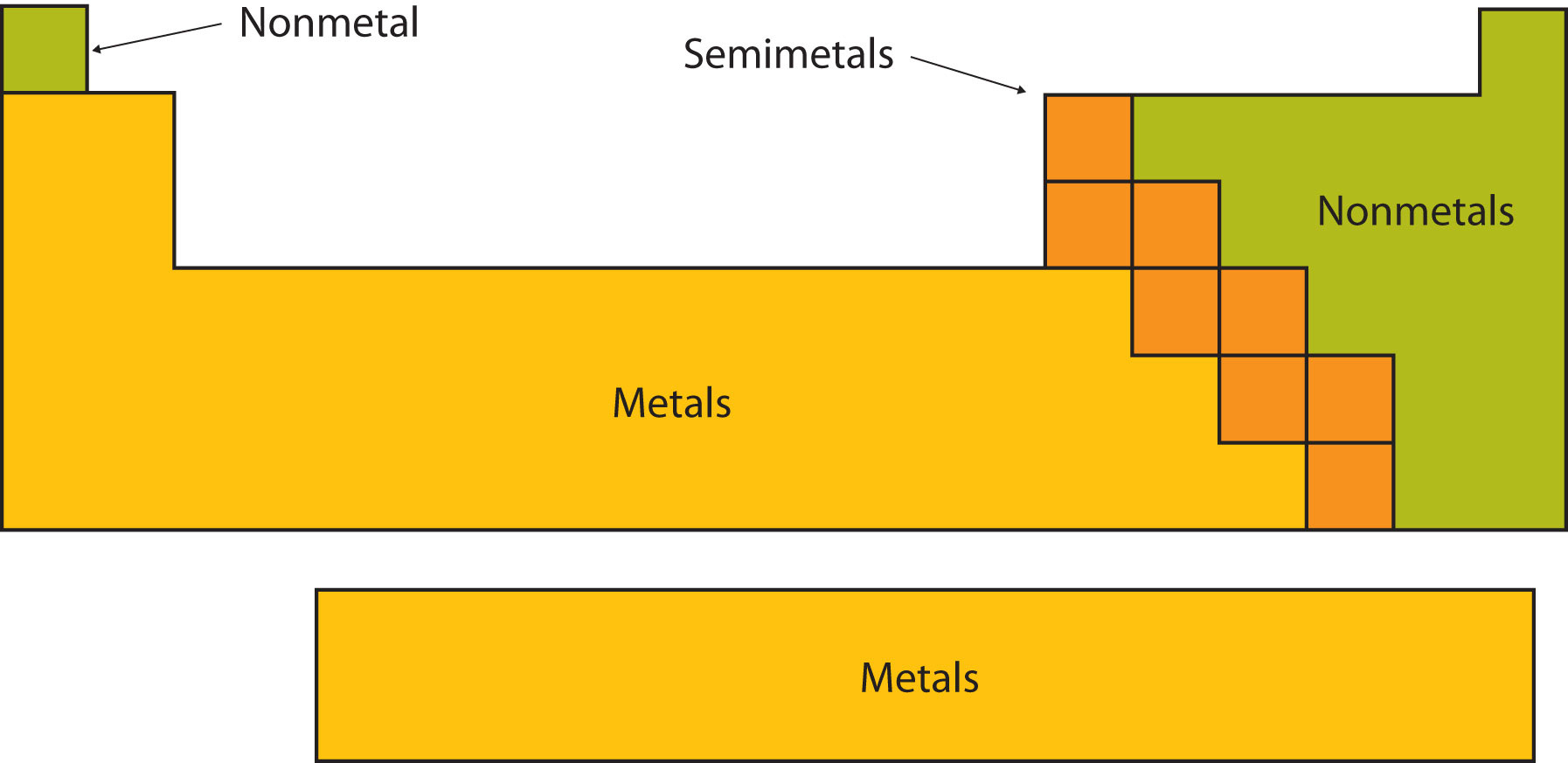
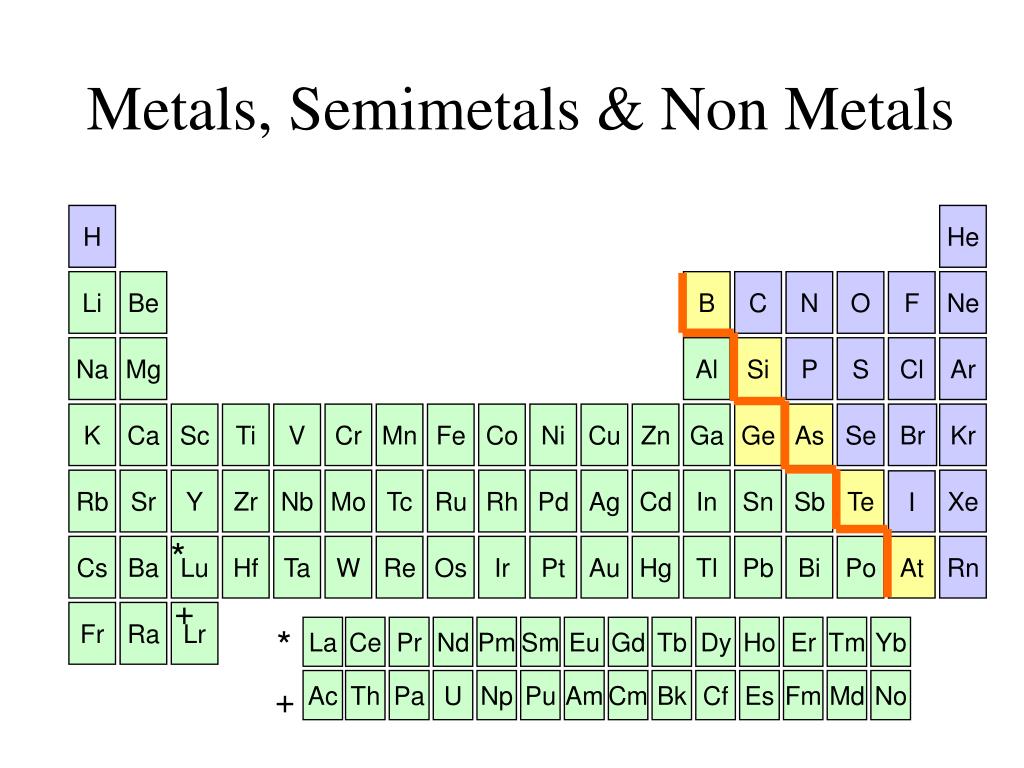
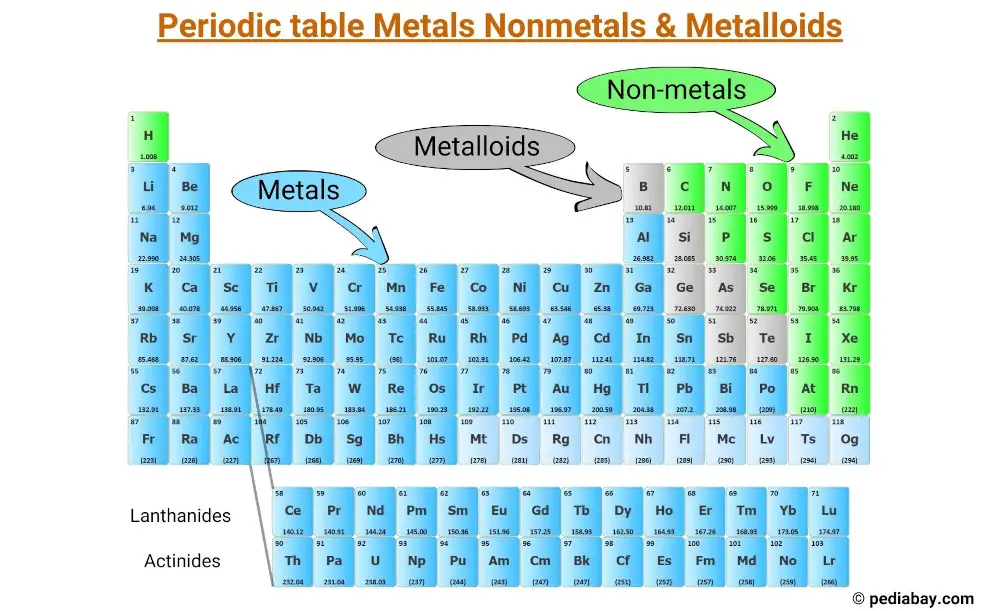
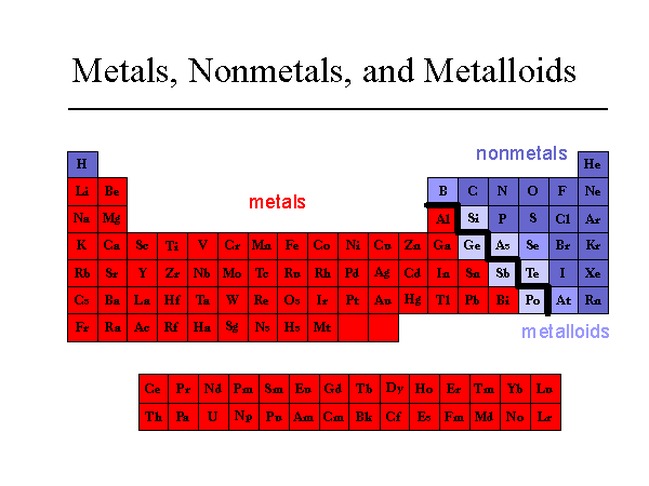
The Edge Cases: Metalloids
Now, things get even more interesting because nature loves to blur the lines. There's a third group, often called metalloids or semimetals. These elements are like the Switzerland of the periodic table – they have properties of both metals and nonmetals.
Take silicon, for example. It looks a bit shiny like a metal, but it's not as good at conducting electricity. However, under certain conditions, it can become a pretty decent conductor. This "in-between" nature is exactly what makes silicon so crucial for modern technology, especially in semiconductors and computer chips. Without these versatile metalloids, our digital world would be a very different place!
Other common metalloids include germanium, arsenic, antimony, and tellurium. They might not be as common in everyday conversation as iron or oxygen, but their unique properties are vital in many specialized applications, from electronics to medicine.
So, when you're looking at the periodic table, you might notice a diagonal "staircase" line that often separates the metals from the nonmetals. The elements sitting right on that line are usually the metalloids. They’re the perfect illustration that not everything in the universe fits neatly into just two boxes. Life is rarely that simple, and the elements are no exception!

Why Does It Even Matter?
So, why go through all this fuss? Classifying elements as metals, nonmetals, and metalloids isn't just an academic exercise. It's the foundation for understanding chemistry, materials science, and even biology.
Knowing if something is a metal or a nonmetal tells you how it will likely react with other elements. It helps predict whether it will be used to build a skyscraper or to make a fire-retardant fabric. It's like having a cheat sheet for how the universe works on its most fundamental level.
Think about the difference between a strong steel beam holding up a building (metal!) and the nitrogen gas filling your tires (nonmetal!). Or the copper wires carrying electricity (metal!) versus the glass in your windows (mostly made from a nonmetal, silicon dioxide!). These classifications are everywhere, shaping the world we live in.
The next time you see an element on the periodic table, take a moment to think about its category. Is it the shiny, conductive metal? The diverse, insulating nonmetal? Or the intriguing, in-between metalloid? Each one has its own story and plays a vital role in the grand, fascinating tapestry of matter.