Classify Each Compound As Ionic Or Molecular

So, picture this: I'm standing in my kitchen, staring blankly at a jar of salt and a bottle of water. My mission? To make a truly epic cup of tea. Nothing fancy, just good old-fashioned comfort. But as I reach for the salt, a little voice in my head (okay, maybe it was just my inner chemistry nerd) pipes up: "Hey, you know that salt is ionic, right? And the water is molecular. Pretty neat, huh?" I swear, sometimes my brain just doesn't know when to switch off. It’s like having a tiny, overenthusiastic science professor living in my skull, constantly pointing out the molecular magic happening all around us.
It got me thinking, though. We all use these words – ionic, molecular – thrown around in science class, right? But what do they really mean? And more importantly, how can we tell the difference between a compound that’s one or the other? Because, let’s be honest, memorizing lists of compounds and their classifications feels a bit like trying to herd cats. Not exactly the most engaging pastime. But if we can understand the why behind it, suddenly things get a whole lot more interesting. It's like unlocking a secret code to how the world works, one compound at a time.
Think about it. We’re surrounded by a symphony of chemical interactions. That delicious crunch of a cookie? That refreshing sip of soda? That slightly alarming fizz when you mix vinegar and baking soda? It's all chemistry, folks! And at the heart of it all lies this fundamental distinction: whether a compound is built on the strong, electrostatic handshake of ionic bonds or the more subtle, shared whispers of covalent bonds, leading to molecular compounds.
Must Read
So, grab your metaphorical lab coat (or, you know, just a comfy chair), because we're about to dive into the fascinating world of classifying compounds. We’ll ditch the tedious memorization and instead focus on the superpowers and personalities of these tiny chemical building blocks. Because once you get the hang of it, it’s surprisingly intuitive. And who knows, you might even start seeing the world through a slightly more scientific, and dare I say, magical lens.
The Great Chemical Divide: Ionic vs. Molecular
Alright, let's get down to the nitty-gritty. At its core, the difference between ionic and molecular compounds boils down to how the atoms in them are held together. It’s like comparing a super-strong magnet to a couple of friends holding hands. Both involve a connection, but the nature of that connection is wildly different.
Imagine a world where some atoms are like generous givers, and others are like eager receivers. That's kind of what's happening in ionic compounds. We're talking about a dance between metals and nonmetals. Metals, bless their electron-donating hearts, tend to lose electrons pretty easily. They become positively charged ions, or cations. Nonmetals, on the other hand, are the electron hoarders. They love to grab those electrons, becoming negatively charged ions, or anions.
And what happens when you have a positively charged something and a negatively charged something hanging out together? Boom! Electrostatic attraction. It’s like a cosmic dance of opposites, where these charged particles are pulled towards each other with incredible force. This is what we call an ionic bond. It's not a sharing situation; it's a full-on, give-and-take transfer. Think of it as a chemical engagement, a permanent commitment born from a significant electron exchange.
So, when you see a compound made up of a metal and a nonmetal, you can pretty much bet your bottom dollar it's going to be an ionic compound. They form these beautiful, ordered crystal lattices – think of perfectly stacked LEGO bricks. This structure is a direct result of the strong attractions between all the positive and negative ions. It's a really stable, organized setup.

Now, let's switch gears to the other side of the track: molecular compounds. Here, we’re primarily dealing with nonmetals bonding with other nonmetals. But this time, the story is different. Instead of one atom completely snatching electrons from another, these atoms decide to share. They form covalent bonds, where they essentially pool their electrons together. It's like a chemical potluck, where everyone brings something to the table and they all benefit from the shared resources.
This sharing creates distinct, individual units called molecules. Unlike the giant, repeating structures of ionic compounds, molecules are like discrete little packages. They have their own identity, their own shape, and their own way of interacting with other molecules. It's a more flexible, less rigid arrangement than the ionic lattice.
The Sneaky Clues: How to Spot Them
Okay, so we've got the fundamental difference. But how do you actually look at a chemical formula and say, "Yep, that's ionic!" or "Ah, that’s definitely molecular!"? This is where the real detective work begins. Fortunately, there are some pretty reliable clues.
The first and most obvious clue is the periodic table. It’s your best friend in this game. Remember that metals are generally found on the left side of the periodic table (think alkali metals, alkaline earth metals), and nonmetals are on the right (think halogens, noble gases, and those in the middle like oxygen, nitrogen, carbon, sulfur).
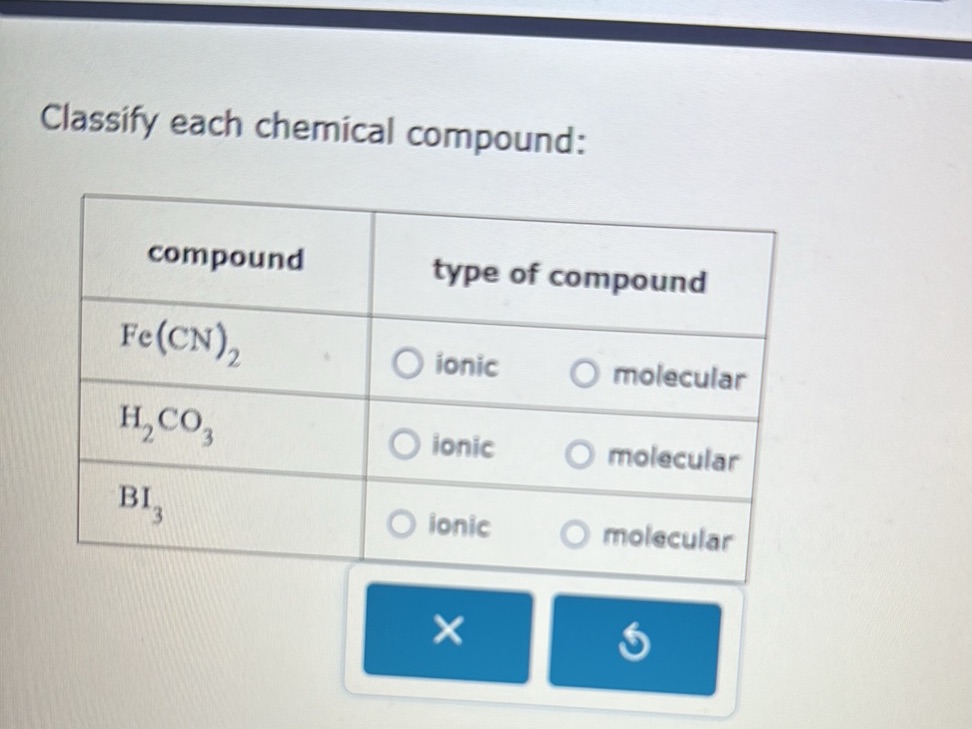
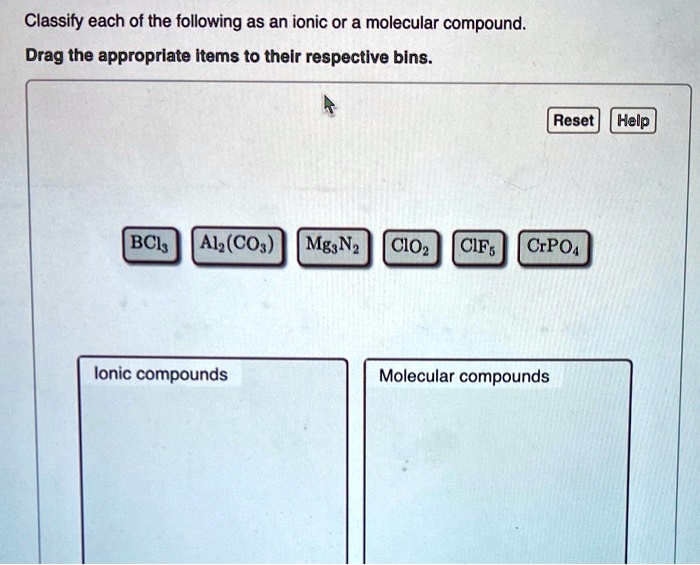
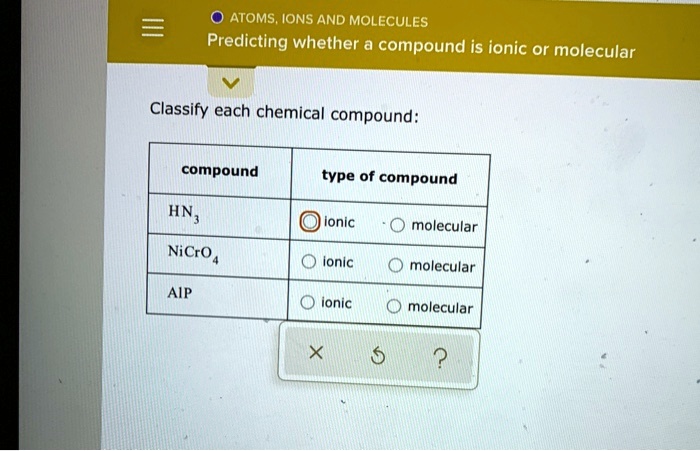
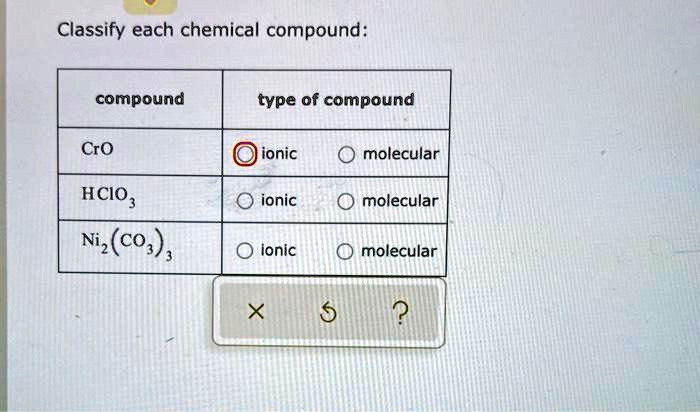
So, rule number one: If a compound is made up of a metal and a nonmetal, it’s almost certainly ionic. For example, sodium chloride, the fancy name for table salt (NaCl). Sodium (Na) is an alkali metal, and chlorine (Cl) is a nonmetal. Metal + Nonmetal = Ionic. Easy peasy, lemon squeezy. Another one: Magnesium oxide (MgO). Magnesium (Mg) is an alkaline earth metal, oxygen (O) is a nonmetal. Again, Metal + Nonmetal = Ionic. These compounds are characterized by their strong electrostatic attraction, leading to high melting and boiling points. They also tend to be brittle solids.

Now, for the molecular side of things. If a compound is made up of two or more nonmetals, it’s most likely molecular. Think of water (H₂O). Hydrogen (H) and oxygen (O) are both nonmetals. Covalent bonds are formed through sharing electrons, creating these discrete water molecules. Carbon dioxide (CO₂) is another classic. Carbon (C) and oxygen (O) are both nonmetals, forming molecules through covalent bonding. These compounds often exist as gases, liquids, or solids with lower melting and boiling points compared to their ionic counterparts. Their molecular structure means they don’t form those rigid lattices.
But wait, there's a little asterisk next to this rule. What about polyatomic ions? These are groups of atoms that have a charge, like sulfate (SO₄²⁻) or ammonium (NH₄⁺). When a metal bonds with a polyatomic ion, it’s still considered ionic, even though the polyatomic ion itself contains covalent bonds within it. So, sodium sulfate (Na₂SO₄) is ionic because sodium is a metal and sulfate is a negatively charged polyatomic ion. It’s like a metal cation is attracted to a whole molecule that carries a net negative charge. This can be a bit of a mind-bender at first, but once you recognize the polyatomic ion, you’ll know the compound is ionic.
Here's a handy trick for spotting common polyatomic ions: they usually end in "-ate" or "-ite" (like nitrate, sulfate, carbonate, sulfite, phosphate) or are the ammonium ion. So, if you see a metal followed by something ending in "-ate" or "-ite" (or ammonium), you’re probably looking at an ionic compound. The compound itself is held together by ionic bonds between the metal cation and the polyatomic anion.
The Tale of Two Bonds: A Deeper Dive
Let's get a little more granular, shall we? Understanding the nature of these bonds can shed even more light on why these compounds behave the way they do. It’s not just about classifying; it’s about understanding the underlying forces.
In ionic compounds, the bond is essentially an electrical attraction between oppositely charged ions. Imagine tiny magnets attracting each other. This attraction is incredibly strong and omnidirectional, meaning each ion is attracted to all the oppositely charged ions surrounding it. This is what leads to the formation of crystal lattices – highly ordered, repeating structures. These strong attractions mean that ionic compounds generally have very high melting and boiling points. It takes a lot of energy to break apart these strong electrostatic forces. Think about how much heat it takes to melt or boil salt! It’s not exactly something you can do on your kitchen stove. They are also typically hard but brittle, because if you try to shift the layers of ions, like charges will end up next to each other, causing repulsion and breaking the crystal.
When dissolved in water, ionic compounds often dissociate, meaning they break apart into their individual ions. This is why solutions of ionic compounds can conduct electricity – the mobile ions act as charge carriers. Remember that salty water you were thinking about for your tea? That's why it conducts electricity (though maybe not enough to power a small city, don't go experimenting with your toaster next to the kettle!).
Now, molecular compounds are formed by covalent bonds. Here, atoms share electrons. This sharing can be equal or unequal, depending on the atoms involved. If the sharing is equal, we have a nonpolar covalent bond. If the sharing is unequal, we have a polar covalent bond, where one atom pulls the shared electrons a bit closer, creating a slight positive charge on one end of the molecule and a slight negative charge on the other. This polarity can have a big impact on how molecules interact with each other and with solvents like water.
Because molecules are discrete units, the forces holding different molecules together (intermolecular forces) are generally weaker than the ionic bonds within ionic compounds. This is why molecular compounds often have lower melting and boiling points. They don’t need as much energy to overcome these intermolecular attractions and transition into different states. Think about ice melting into water – a relatively small change in temperature compared to melting rock salt.
Also, pure molecular compounds (unless they have polar bonds and are in water) don't typically conduct electricity because they don't have free-moving charged particles. They’re more like a collection of independent little balls, not a charged network.
Putting it All Together: Practice Makes Perfect!
So, let's recap the detective work. You see a chemical formula. Your brain goes into Sherlock mode. What do you look for?
Step 1: Identify the Elements
What atoms are present? Are they metals, nonmetals, or a mix? Your trusty periodic table is your guide here.

Step 2: Check the Metal/Nonmetal Combination
If you have a metal and a nonmetal, it's almost certainly ionic. Think: NaCl, KBr, MgO.
Step 3: Look for Nonmetal Combinations
If you have two or more nonmetals, it's most likely molecular. Think: H₂O, CO₂, CH₄.
Step 4: Beware of Polyatomic Ions!
If you see a metal bonded to a group of atoms that ends in "-ate" or "-ite" (or the ammonium ion, NH₄⁺), the compound is ionic, even though the polyatomic ion itself has covalent bonds. Think: Na₂SO₄, KNO₃, (NH₄)₂S.
Let’s try a few together, just for fun. Because, honestly, who doesn't love a good chemical puzzle? Imagine you’re presented with:
- Calcium chloride (CaCl₂): Calcium (Ca) is a metal, chlorine (Cl) is a nonmetal. Metal + Nonmetal = Ionic.
- Sulfur dioxide (SO₂): Sulfur (S) and oxygen (O) are both nonmetals. Nonmetal + Nonmetal = Molecular.
- Potassium nitrate (KNO₃): Potassium (K) is a metal. Nitrate (NO₃⁻) is a polyatomic ion. Metal + Polyatomic Ion = Ionic.
- Methane (CH₄): Carbon (C) and hydrogen (H) are both nonmetals. Nonmetal + Nonmetal = Molecular.
- Aluminum oxide (Al₂O₃): Aluminum (Al) is a metal, oxygen (O) is a nonmetal. Metal + Nonmetal = Ionic.
See? It's like a game of chemical bingo! Once you get the hang of spotting the combinations, it becomes second nature. You'll start looking at ingredient lists on food packaging and chemical labels with a whole new appreciation (and maybe a slight urge to classify everything).
It's truly fascinating how these simple differences in bonding can lead to such vastly different properties. From the solid, crystalline structure of salt to the gaseous nature of carbon dioxide, it all comes back to whether atoms are giving and taking or happily sharing. It’s a fundamental concept that underpins so much of the chemistry we encounter every day. So next time you're making that cup of tea, or just observing the world around you, take a moment to appreciate the ionic and molecular marvels at play. It’s a little bit of science magic that’s always, always happening. Happy classifying!