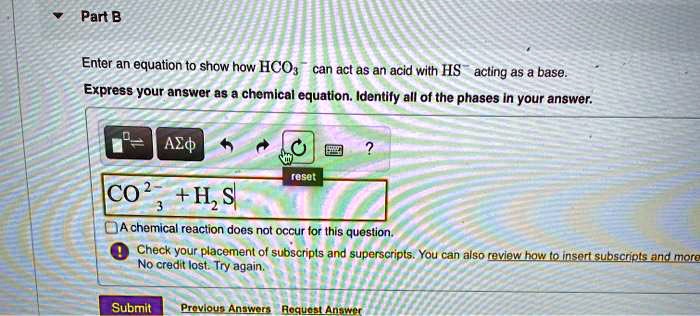
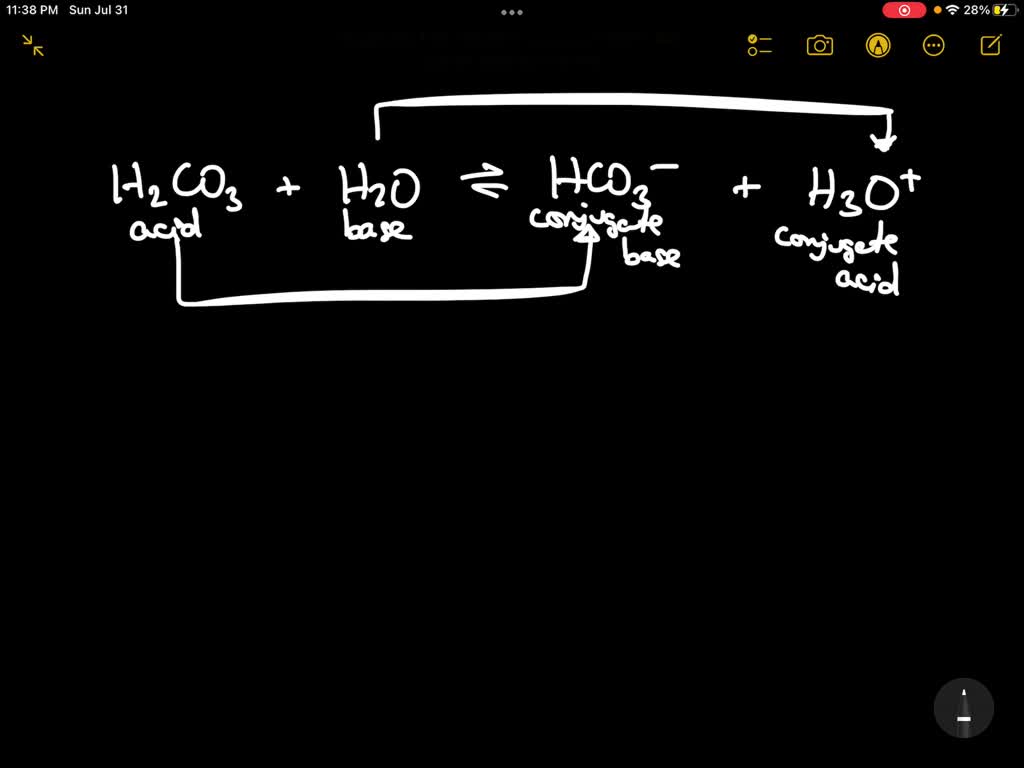
Choose The Answer That Best Describes Hco3

Ever taken a sip of something fizzy and felt that delightful tingle on your tongue? That little tickle is often thanks to a superhero in disguise, a humble molecule known as HCO3. Now, that might sound a bit like a secret agent's code name, and in a way, it is! This amazing compound is constantly working behind the scenes to keep things balanced, both in our bodies and in the world around us.
Think of HCO3, or bicarbonate as it's more commonly called, like the ultimate peacekeeper. It’s a master diplomat, always ready to step in and mediate when things get a little too wild. Imagine a party where everyone is getting a bit too excited, shouting and bumping into each other. That's kind of what can happen inside us and in our environment if the acidity levels go haywire. Too much acid can be like a bunch of rowdy guests, causing trouble and making everyone uncomfortable. That's where our friend HCO3 swoops in, smoothing things over with its gentle, neutralizing touch.
It's like having a tiny, invisible bodyguard for your internal chemistry!
One of the most heartwarming jobs HCO3 has is keeping our blood just right. Our blood needs to be a very specific level of not-too-acidic and not-too-alkaline. If it swings too far one way or the other, it’s a big problem for our cells. HCO3 is like the vigilant doorman, making sure the pH of our blood stays in its happy, healthy zone. It’s so important that our bodies have clever ways of producing and recycling it constantly, like a super-efficient, internal recycling plant. It’s truly a testament to how much our bodies love us, constantly tending to our needs with molecules like HCO3.
Must Read
But HCO3 isn't just about keeping our insides tidy. It plays a starring role in some of our favorite treats and drinks! That satisfying fizz in your soda? That’s often the result of HCO3 reacting with acid to release tiny bubbles of carbon dioxide. It’s like a miniature fireworks show happening right in your glass! And speaking of baking, have you ever wondered how cakes and cookies get their fluffy, airy texture? Many baking ingredients, like baking soda, are sources of HCO3. When heated, they release those same little carbon dioxide bubbles, making your baked goods rise and become wonderfully light. So, every time you enjoy a perfectly risen muffin or a crisp cookie, you can give a little nod to HCO3 for its delicious contribution.
It’s quite funny to think that something so crucial can be so simple. Chemically speaking, HCO3 is just one carbon atom, three oxygen atoms, and one hydrogen atom. But oh, the magic they create together! It's a perfect example of how even the smallest things can have a massive impact. It's like finding out your quiet neighbor is actually a world-renowned chef who just prefers to keep to themselves. HCO3 is that unsung hero, quietly performing essential duties that keep us alive and allow us to enjoy so many things.
Beyond our bodies and kitchens, HCO3 is also busy in the natural world. It’s a major component of ocean water, helping to keep the vast seas at a stable pH. This is incredibly important for marine life, from tiny plankton to giant whales. Imagine the ocean as a giant bathtub; HCO3 is like the gentle soap that ensures the water is just right for all the aquatic creatures to swim and thrive. It’s a constant, silent guardian of the blue expanse.

Consider rivers and lakes too. HCO3 helps buffer these freshwater bodies, protecting them from sudden changes in acidity that could harm fish and other inhabitants. It’s like the natural filtration system that keeps our waterways clean and healthy, ensuring they can continue to be sources of life and beauty. It reminds us that even at the grandest scales, these tiny molecular interactions are shaping our planet.
So, the next time you hear the word HCO3 or see its chemical formula, don't think of it as some intimidating scientific jargon. Instead, picture a friendly, helpful molecule that's working hard to keep your body running smoothly, making your favorite fizzy drinks delightful, and contributing to the health of our planet. It's a tiny wonder, a true marvel of nature, and a little something to smile about. It’s the silent, often unacknowledged, star of so many everyday experiences, and understanding its role can bring a surprising sense of appreciation for the invisible forces that shape our world and keep us all going.

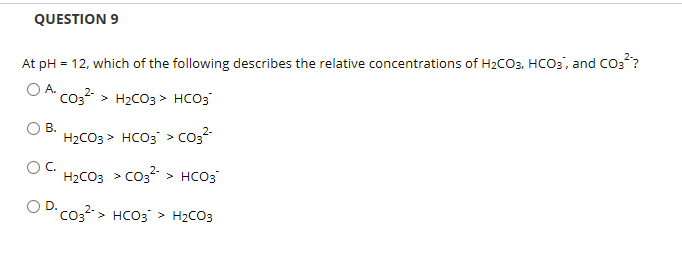

![[GET ANSWER] 3. Answer ALL parts a) – e). Reaction of H2O and CO2 to](https://cdn.numerade.com/ask_images/d5f4ddf998c54f75870b53fb7d1748ea.jpg)