Chemical Bonding Pogil Activity 4 Answer Key
Okay, so picture this: I'm digging through a box of my dad's old stuff, you know, the kind of box that smells faintly of mothballs and forgotten dreams. I stumble upon this really old, slightly beat-up chemistry textbook. Flicking through it, I see these diagrams of atoms holding hands, like, literally, with little lines connecting them. My initial thought? "Huh, did they really teach chemistry using stick figures?" Then, I remember a little POGIL activity we did back in the day, something about chemical bonding. And suddenly, those diagrams make a whole lot more sense. It’s not just stick figures, my friends, it’s the secret handshake of the universe!
You see, everything around us, from the air we breathe to the phone you’re probably scrolling on right now, is held together by these invisible forces. It’s like a giant, cosmic game of "keep away," but instead of a ball, it’s electrons. And this POGIL activity, specifically section 4, was all about cracking that code. For anyone who’s ever stared at a POGIL answer key with a mixture of relief and mild panic, this one’s for you. We’re diving into the nitty-gritty of Chemical Bonding, POGIL Activity 4, and, dare I say it, actually understanding it.
So, what is chemical bonding, anyway? Think of atoms as tiny, super-picky people. They don't like being alone if their outer electron shell isn't "full." It's like having a missing piece of a puzzle; they just feel… incomplete. This desire for completeness is what drives them to interact, to share, or even to straight-up steal electrons from each other. Wild, right? It's all about achieving a stable electron configuration, usually a full outer shell, like finally finding a matching sock.
Must Read
POGIL activities are designed to get you thinking, not just memorizing. They throw you into a scenario, ask you questions, and guide you towards the answers through observation and deduction. It’s like a scientific scavenger hunt. And POGIL Activity 4? That’s where things get really interesting. We move beyond just knowing that atoms bond to understanding how and why they choose certain partners and certain methods.
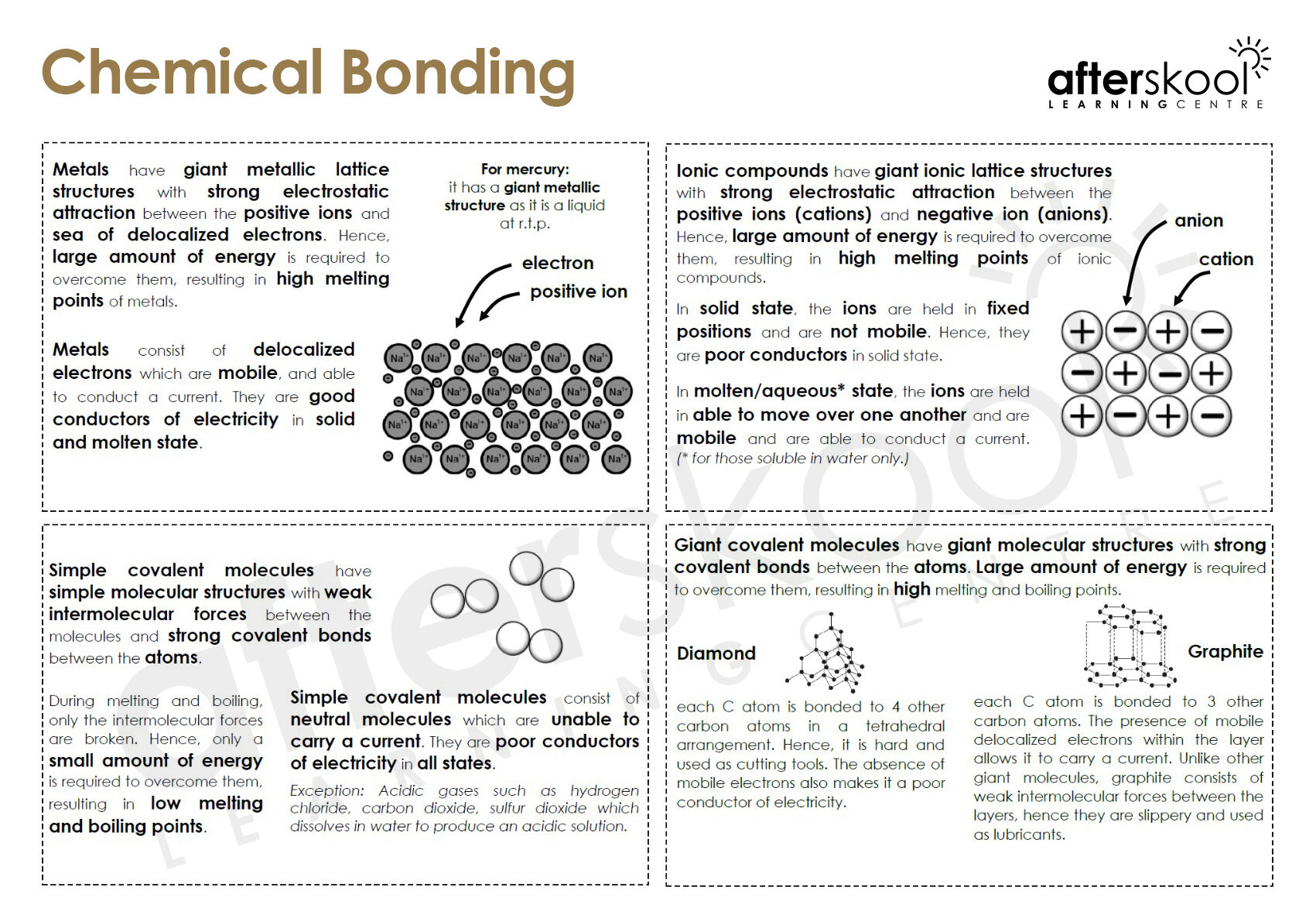
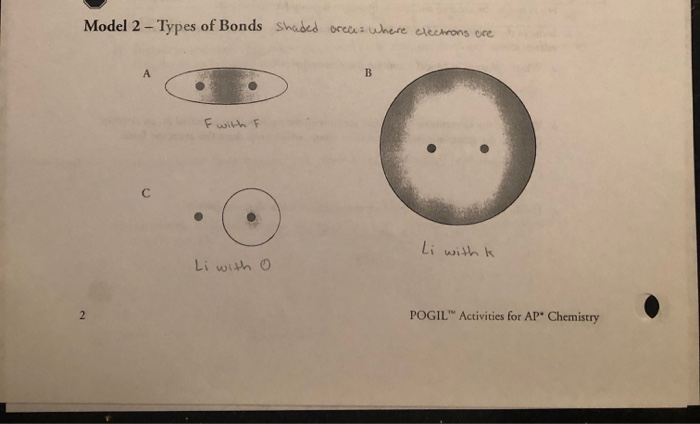
Let's talk about the main players: ionic bonding and covalent bonding. These are the heavyweight champions of chemical bonds. Think of ionic bonding like a wealthy benefactor and a person in need. One atom, typically a metal, has extra electrons it’s happy to get rid of. Another atom, usually a non-metal, is desperately looking to grab some electrons to fill its outer shell. So, the metal atom essentially gives an electron (or more!) to the non-metal atom.
This electron transfer creates ions – charged atoms. The atom that loses electrons becomes positively charged (a cation), and the atom that gains electrons becomes negatively charged (an anion). Now, here’s the magic: opposite charges attract! It’s the fundamental rule of the universe, folks. This electrostatic attraction between the positive cation and the negative anion is what holds them together in an ionic bond. It’s a powerful relationship, built on a foundation of electron generosity (or desperation, depending on your perspective).
Think about table salt, NaCl. Sodium (Na) is that generous donor, and chlorine (Cl) is the eager recipient. Sodium gives up an electron, becoming Na+, and chlorine snatches it up, becoming Cl-. Then, bam! They’re stuck together like glue. This is why ionic compounds, like salts, tend to form crystal lattices – ordered, repeating structures. They’re all just trying to get as close as possible to their oppositely charged neighbors.

On the other hand, we have covalent bonding. This is more like a cooperative partnership, a "let's share and be friends" kind of vibe. Instead of one atom completely giving up an electron, atoms in a covalent bond share electrons. They’re both looking for that full outer shell, and they realize that by pooling their resources, they can both achieve it. It's like a potluck dinner for electrons!
Covalent bonds typically occur between non-metal atoms. They can share one pair of electrons (a single bond), two pairs (a double bond), or even three pairs (a triple bond). The more pairs they share, the stronger the bond. Think of it like a tug-of-war where both sides are pulling equally. They're not letting go, but they're also not completely taking over. They’re in it together.
Water (H₂O) is a classic example of covalent bonding. Each hydrogen atom shares an electron with the oxygen atom, and the oxygen atom shares one electron back with each hydrogen. This sharing allows both hydrogen atoms to have a full outer shell (2 electrons), and the oxygen atom to have a full outer shell (8 electrons). It’s a beautiful dance of electron sharing, creating one of the most essential molecules for life.
POGIL Activity 4 likely walked you through identifying these types of bonds based on the elements involved. You probably had to look at the periodic table, recall which elements are metals and which are non-metals. It's like having a cheat sheet for atomic personalities. Metals: generally like to lose electrons, forming positive ions. Non-metals: generally like to gain or share electrons.

One of the key concepts POGIL activities often explore is electronegativity. This is a fancy term for an atom's "electron-attracting ability." Some atoms are like magnets for electrons, while others are a bit more laid back. The greater the difference in electronegativity between two atoms, the more likely they are to form an ionic bond. If the electronegativity difference is small, they’re more likely to share electrons and form a covalent bond.
So, if you saw a big difference in electronegativity between two elements in POGIL Activity 4, you’d probably deduce it was an ionic bond. If the difference was small, ding ding ding, likely covalent. It’s like playing a game of "spot the difference," but the stakes are the very structure of matter!
The activity might have also introduced you to polar covalent bonds. This is where the sharing isn't perfectly equal. One atom pulls the shared electrons a little closer to itself, creating a slight negative charge on that end of the molecule and a slight positive charge on the other. It's like a covalent bond with a bit of a personality quirk, a slight bias.
Think of hydrogen chloride (HCl). Chlorine is more electronegative than hydrogen, so it pulls the shared electrons closer. This makes the chlorine end slightly negative ($\delta-$) and the hydrogen end slightly positive ($\delta+$). These are called dipoles, and they have a significant impact on how molecules behave and interact with each other. It’s the subtle nuances that make chemistry so fascinating, don’t you think?

Then there are nonpolar covalent bonds. In these bonds, the electrons are shared equally between the two atoms. This usually happens when the two atoms are the same element, like in O₂ or N₂. Since they have the same "electron-pulling power," the electrons hang out right in the middle, and there are no partial charges. It’s the ultimate in equal partnership.
The POGIL answer key for activity 4 would have solidified these distinctions. You might have been asked to draw Lewis structures, which are simple diagrams showing the valence electrons (the outermost electrons involved in bonding) and how they are shared or transferred. These diagrams are incredibly helpful for visualizing the bonds. They’re like the blueprints for molecular construction.
Let’s say you were asked to determine the type of bond in KBr. You’d recognize potassium (K) as an alkali metal (Group 1) and bromine (Br) as a halogen (Group 17). Metals and non-metals? That’s your cue for ionic bonding. Potassium wants to lose an electron to achieve a stable configuration, and bromine wants to gain one. Voilà! K⁺ and Br⁻, held together by electrostatic attraction.
Now, consider N₂ (nitrogen gas). Both nitrogen atoms are non-metals. They have similar electronegativity values. They’re going to share electrons. How many? Nitrogen needs 3 more electrons to achieve a stable octet. So, they share three pairs of electrons, forming a triple covalent bond. N≡N. Pretty neat, huh? It’s a very strong bond, which is why nitrogen gas is quite unreactive.

The POGIL activity likely had questions that required you to analyze these situations. Perhaps you had to predict the polarity of a bond or a molecule. This involves looking at the electronegativity differences and the molecular geometry. Even if a bond itself is polar, the molecule might be nonpolar if the polarities cancel each other out due to symmetry. It’s like having two people arguing on opposite sides of a seesaw; the seesaw stays balanced.
Understanding these bonding types is fundamental to understanding the properties of substances. Why is salt a solid at room temperature, while water is a liquid? Why does one dissolve in water and another doesn’t? It all comes back to how the atoms are connected. Ionic compounds tend to have high melting and boiling points because of the strong electrostatic forces holding the ions together. Covalent compounds, especially smaller ones, often have lower melting and boiling points because the forces between the molecules are weaker than the forces within the molecules.
The answer key served as your confirmation, your pat on the back saying, "Yep, you got it!" It’s a crucial step in the learning process, especially with POGIL, where you're actively constructing your understanding. Without the key, you might be left scratching your head, wondering if your deductions were right on the money or wildly off course. And trust me, we've all been there, staring at our answers, hoping for the best.
So, when you’re looking at that POGIL Activity 4 answer key, don't just see a list of correct answers. See the culmination of understanding how atoms interact, how they form the building blocks of everything. See the elegance of electron sharing and the power of electrostatic attraction. It’s not just chemistry; it’s the language of the universe, spoken in the silent, powerful bonds between atoms. Pretty cool, right?