Chapter 15 Review Acid Base Titration And Ph

You know, I remember this one time in high school chemistry. We were doing this lab, and I was trying to measure out exactly 50.00 mL of some solution. I had this fancy burette, all these stopcocks and markings, and I was convinced I was going to be the precision king of the lab. My lab partner, bless his heart, was a bit more… free-wheeling with his measurements. Let's just say his 50 mL looked suspiciously like 48 mL on a good day, and maybe 52 mL on a bad one. We were doing this thing where we were supposed to neutralize one liquid with another, and the difference in our results? It was wild. Like, my perfectly measured solution produced this pristine, neutral pH, and his? It was either way too acidic or way too basic. I was so smug about my accuracy, and he just shrugged and said, "Eh, close enough." Oh, the youthful ignorance! Little did I know, that seemingly simple act of mixing and measuring was just the tip of the iceberg when it came to something called acid-base titration. And trust me, it's way more interesting (and useful!) than I ever gave it credit for back then.
So, Chapter 15, huh? Acid-Base Titration and pH. If your eyes are glazing over already, I get it. The words themselves sound a bit… intimidating. Like something out of a science fiction movie where the mad scientist is mixing potions. But seriously, it's actually one of those concepts that, once you get it, you see it everywhere. Think about it. From the antacids you might take for heartburn to the way our bodies regulate their internal chemistry, this stuff is the unsung hero. It's all about figuring out the exact concentration of an acid or a base by carefully reacting it with another substance of known concentration. Pretty neat, right?
The Balancing Act: What's All the Fuss About Neutralization?
At its core, titration is a sophisticated way of playing "find the perfect match" in the world of chemistry. Acids and bases, as you probably remember, are like opposite sides of a coin. Acids have a surplus of hydrogen ions (H+), making them taste sour (think lemon juice!) and corrosive. Bases, on the other hand, have a surplus of hydroxide ions (OH-), making them feel slippery (hello, soap!) and bitter.
Must Read
When you mix an acid and a base, they react. This reaction is called neutralization. It's like they're having a polite little chemical argument, and the outcome is usually water (H2O) and a salt. The H+ from the acid teams up with the OH- from the base to form water, and the remaining ions from each form the salt. It’s a beautiful, orderly process. The ultimate goal of a titration is to find that sweet spot where all the acid has been neutralized by the base, or vice versa. No excess of either left hanging around. It's the chemical equivalent of a perfectly balanced meal.
But how do we know when we've reached that perfect balance? That’s where things get really interesting. We can't just taste it (please, don't taste your lab experiments!) or feel it (again, safety first!). We need a way to detect this neutralization point. This is where indicators come in.
Indicators: The Color-Changing Detectives
Think of indicators as little chemical cheerleaders. They're substances that change color depending on whether the solution they're in is acidic, basic, or neutral. It's their way of shouting, "We've reached the point!" They're usually weak acids or bases themselves, and their color is tied to their structure. When the pH of the solution changes, their molecular structure shifts, and bam! A different color appears. My personal favorite is phenolphthalein. It’s clear in acid, but in base, it turns this glorious, vibrant pink. It’s so dramatic, you almost expect a fanfare.
You add just a tiny amount of indicator to your solution, and as you add the titrant (the solution of known concentration), you watch the color. When you see that first persistent hint of the indicator's final color, you've likely hit the endpoint. Now, there’s a subtle, but important, difference between the equivalence point and the endpoint. The equivalence point is the theoretical point where the moles of acid exactly equal the moles of base. The endpoint is what you observe in the lab, which is when the indicator changes color. Ideally, a good indicator will have an endpoint that is very close to the equivalence point. Choosing the right indicator is crucial for an accurate titration. It's like picking the right detective for the case – you need someone who’s going to notice the slightest clue.

The Mighty Burette: Precision is Key
So, how do we actually do this titration thing? Well, you need some special equipment. The star of the show is often the burette. This is that long, graduated glass tube with a stopcock at the bottom that I was so proud of (and probably over-handled). The stopcock allows you to control the flow of the titrant, drop by drop. This is where that 0.00 mL precision I was aiming for comes in. You need to be able to accurately measure how much of the titrant you've added to reach the endpoint.
You have your solution of unknown concentration (let's say, an acid) in a flask below the burette. Then, you fill the burette with the solution of known concentration (your base, in this case). As you carefully add the base from the burette to the acid, you swirl the flask constantly to ensure everything mixes thoroughly. You're watching for that color change from the indicator. When it happens, you stop adding the base and record the volume you've used from the burette. This volume, along with the known concentration of the base and the initial volume of the acid, allows you to calculate the unknown concentration of the acid.
It sounds straightforward, but the practice is an art. You learn to recognize the faintest hint of color change. You learn to control that stopcock with a delicate touch. And you definitely learn to appreciate why your lab partner might have been a little less precise with his initial measurements – sometimes, things just don't go perfectly. But that’s the beauty of science, right? We refine, we learn, we try again.
The Math Behind the Magic: Stoichiometry Strikes Again!
Okay, so you've done the titration, you've got your volumes, and you've got your known concentration. Now what? This is where stoichiometry, the study of the quantitative relationships between amounts of reactants and products in chemical reactions, comes back to haunt (or help!) you. If you remember our good friend the balanced chemical equation, it tells you the mole ratio of reactants and products. For example, a balanced equation like:
HCl (aq) + NaOH (aq) → NaCl (aq) + H2O (l)

tells us that one mole of hydrochloric acid (HCl) reacts with one mole of sodium hydroxide (NaOH) to produce one mole of sodium chloride (NaCl) and one mole of water. This 1:1 ratio is super important in titrations where the moles of acid and base are equal at the equivalence point.
So, if you know the moles of base you added (calculated from the volume and concentration of the titrant), and you know the mole ratio from the balanced equation, you can figure out exactly how many moles of acid were in your original flask. And from there, calculating the concentration of the acid is a piece of cake. You're essentially working backward from the known to the unknown, using the balanced chemical reaction as your roadmap.
What if the mole ratio isn't 1:1? For instance, if you're titrating sulfuric acid (H2SO4) with sodium hydroxide (NaOH), the balanced equation is:
H2SO4 (aq) + 2NaOH (aq) → Na2SO4 (aq) + 2H2O (l)
Here, one mole of sulfuric acid reacts with two moles of sodium hydroxide. This 1:2 ratio is crucial! You can't just assume a 1:1 relationship anymore. You have to use the stoichiometry of the reaction to make your calculations correct. This is where many a student has stumbled – forgetting to account for that mole ratio. Don't be that student! Always check your balanced equation.

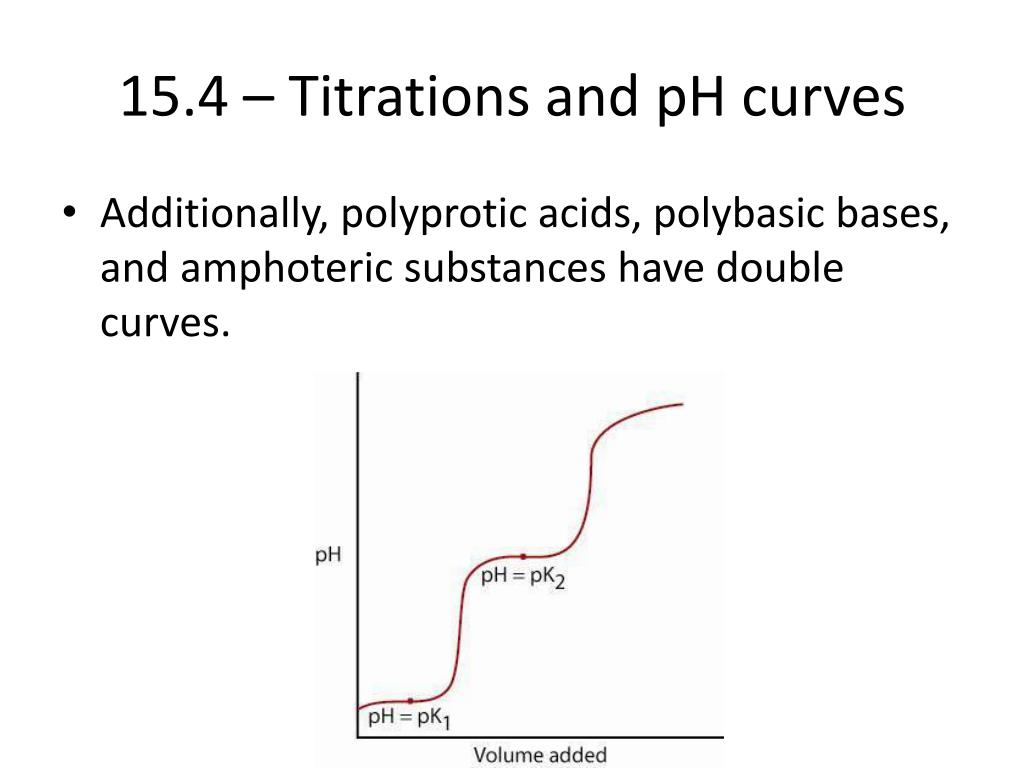
Beyond the Pink: Understanding pH Curves
Now, while the color change of an indicator is a great way to get a quick idea of the endpoint, sometimes we need to be even more precise. This is where pH curves come into play. A pH curve is a graph that plots the pH of the solution as you add the titrant. You can actually measure the pH continuously with a pH meter as you titrate.
These curves have a characteristic "S" shape, and the steepest part of that "S" is where the equivalence point lies. It’s like a chemical rollercoaster, with a dramatic climb in pH (or drop, if you're titrating a base with an acid) right around the neutralization point. By analyzing the shape and the midpoint of this steep rise, you can determine the equivalence point with great accuracy. This is particularly useful when dealing with weak acids and weak bases, where the endpoint might not be as sharp or where the ideal indicator is harder to find.
The pH at the equivalence point tells you a lot about the strengths of the acid and base you're titrating. If you're titrating a strong acid with a strong base, the equivalence point will be at pH 7. If you're titrating a weak acid with a strong base, the equivalence point will be above pH 7 (because the salt formed will hydrolyze and make the solution slightly basic). Conversely, titrating a strong acid with a weak base will result in an equivalence point below pH 7. This is where understanding the interplay between acid strength, base strength, and pH really shines.
Applications, Applications Everywhere!
So, why do we bother with all this titration jazz? Well, it's not just for dusty chemistry labs. It's incredibly practical.
- Food and Beverage Industry: Ever wondered how they ensure your orange juice has the right amount of acidity? Titration! They use it to measure the concentration of acids in juices, wines, and other food products, affecting taste and preservation.
- Environmental Monitoring: Water quality testing often involves titrations to determine the acidity or alkalinity of water bodies. This is crucial for understanding the health of aquatic ecosystems and for treating wastewater.
- Pharmaceuticals: The exact concentration of active ingredients in medicines needs to be spot-on. Titration is a vital quality control tool in the pharmaceutical industry to ensure drug potency and safety.
- Medical Diagnosis: Certain medical tests, like blood gas analysis, involve principles related to acid-base balance and pH measurement that are underpinned by the concepts learned in titration.
- Agriculture: Soil pH is critical for plant growth. Farmers use titration methods to determine the acidity of their soil and decide whether they need to add lime or other amendments.
Seriously, the list goes on and on. It's one of those fundamental techniques that underpins so much of what we do in science and industry. It’s a way to quantitatively understand and control chemical reactions that are happening all around us, and even inside us.

The pH Scale: A Universal Measure of Acidity
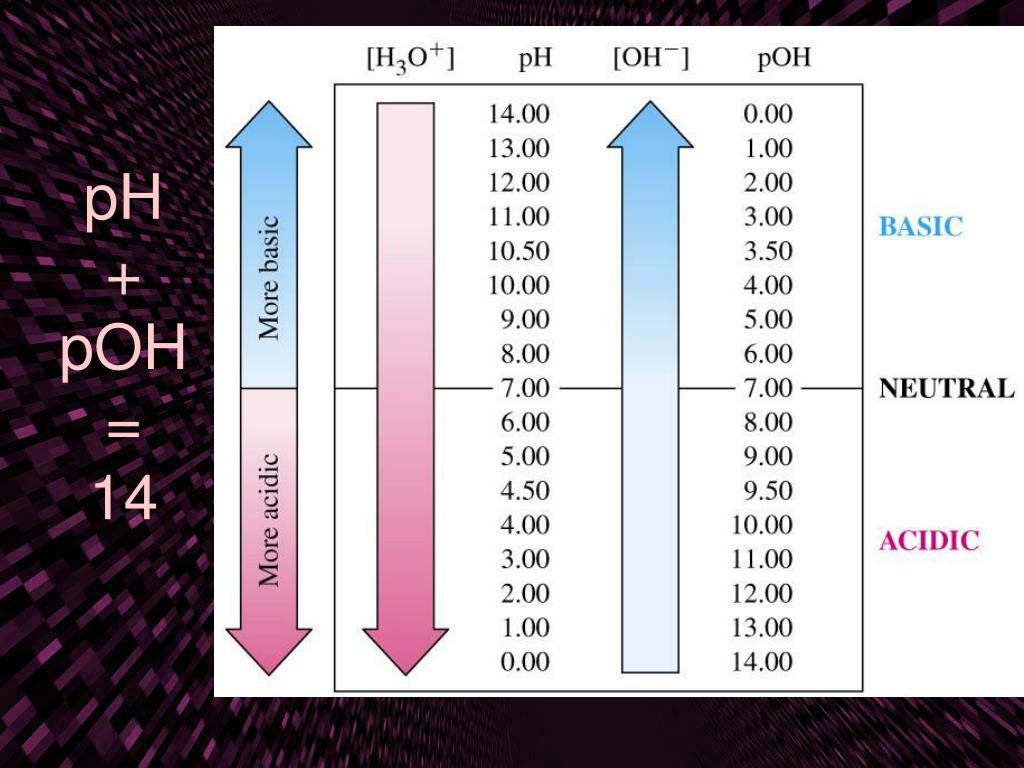
And of course, you can't talk about acids and bases without talking about pH. Remember that little number that tells you how acidic or basic something is? The pH scale runs from 0 to 14.
- A pH of 7 is neutral (like pure water).
- A pH below 7 is acidic. The lower the number, the more acidic.
- A pH above 7 is basic (or alkaline). The higher the number, the more basic.
Each whole number on the pH scale represents a tenfold change in acidity or alkalinity. So, something with a pH of 3 is 10 times more acidic than something with a pH of 4, and 100 times more acidic than something with a pH of 5. That's a pretty significant difference!
pH is mathematically defined as the negative logarithm (base 10) of the hydrogen ion concentration: pH = -log[H+]. This logarithmic scale is why those tenfold changes happen. It’s a convenient way to express very small concentrations of H+ ions. Our bodies, for example, work hard to maintain a very narrow pH range (around 7.35-7.45) in our blood. Even a small deviation from this can be dangerous, which is why we have buffer systems to help regulate it. It’s a constant, delicate balancing act.
Putting It All Together: The Big Picture
So, Chapter 15, acid-base titration and pH. It’s about understanding the power of neutralization, using indicators as our colorful guides, employing precise tools like the burette, and applying stoichiometric calculations to unravel the unknown. It’s about seeing the dramatic changes in pH during a reaction and using those changes to our advantage. It's about recognizing that this seemingly abstract chemistry is the foundation for countless practical applications that affect our daily lives.
It might have seemed like just another lab exercise back in high school, but when you look at it through the lens of what titration does, it’s pretty remarkable. It’s about precision, it’s about observation, and it’s about the elegant dance of chemical reactions. It's a reminder that even the most complex phenomena often have roots in simple, fundamental principles. So next time you take an antacid, or wonder about the acidity of your coffee, remember the science of acid-base titration and pH. It’s a world of precision and balance, happening all around you.