Basic Organic Nomenclature Packet Answer Key
Hey there, science explorers and curious minds! Ever find yourself staring at a string of letters and numbers that looks like a secret code, maybe something like "2-methylpentane" or "ethanol"? Yeah, me too. It's like the universe decided to speak in a secret language, and it’s all about the tiny, fascinating building blocks of life: organic molecules. And guess what? There’s a whole system behind those names, a way to decode them and even create new ones. Today, we’re going to take a peek behind the curtain at something called a "Basic Organic Nomenclature Packet Answer Key."
Now, before you run for the hills thinking this is going to be a dry, textbook-y lecture, hold up! Think of it more like a cheat sheet for understanding the language of chemistry. It’s not about memorizing a million rules; it's about unlocking a cool, logical system. And this answer key? It’s like the ultimate guide, showing you how the magic happens.
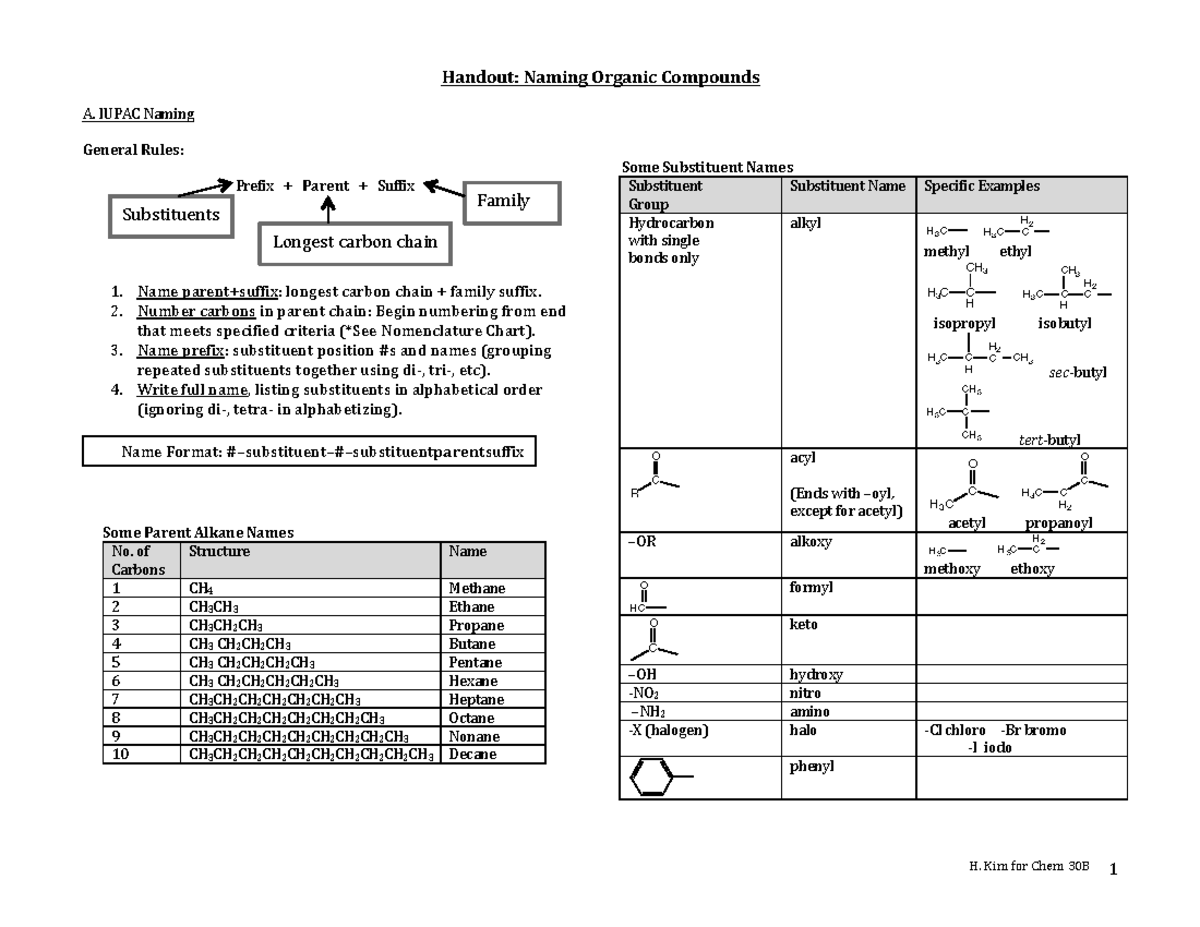
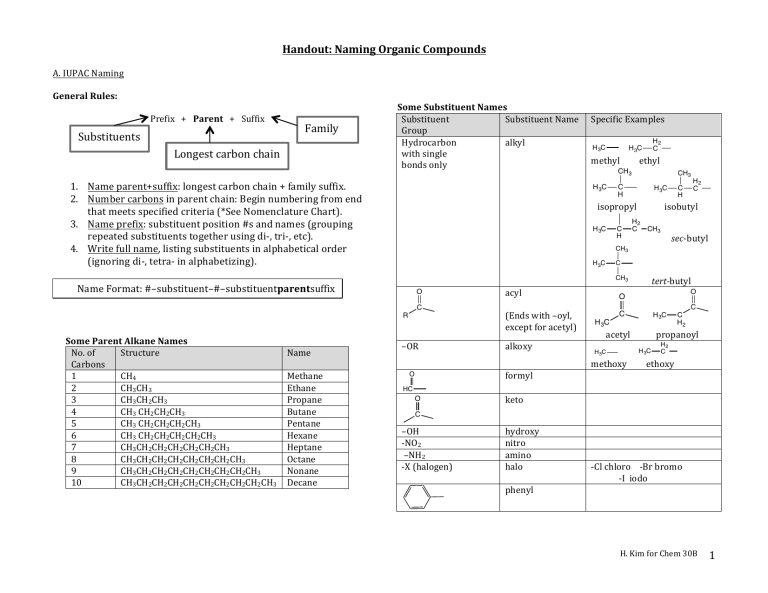
So, what is this "nomenclature" thing anyway? Basically, it's just a fancy word for naming things. In organic chemistry, where we’re dealing with molecules made primarily of carbon and hydrogen (and a few other bits and bobs), having a consistent way to name them is super important. Imagine if every car manufacturer named their models randomly. It would be chaos, right? You’d never know if you were asking for a “Sporty Sedan Deluxe” or a “Four-Door People Mover Plus.” Chemistry needs that same kind of order!
Must Read
This is where the International Union of Pure and Applied Chemistry, or IUPAC for short, swoops in. They're the folks who come up with these naming rules. Think of them as the official rulebook creators for the molecular world. And a "Basic Organic Nomenclature Packet Answer Key" is essentially the solution manual for exercises designed to teach you these IUPAC rules. It’s like getting the answers after you’ve tried to solve some puzzles.
Why is this even a thing? Well, think about it. Every organic molecule has a unique structure. And that structure tells us a lot about how that molecule will behave. Will it be flammable? Will it be sticky? Will it be something that your body can use for energy? All of that comes down to its shape and the atoms it's made of. So, a correct name tells us exactly what that structure is. It's like having a molecular fingerprint!
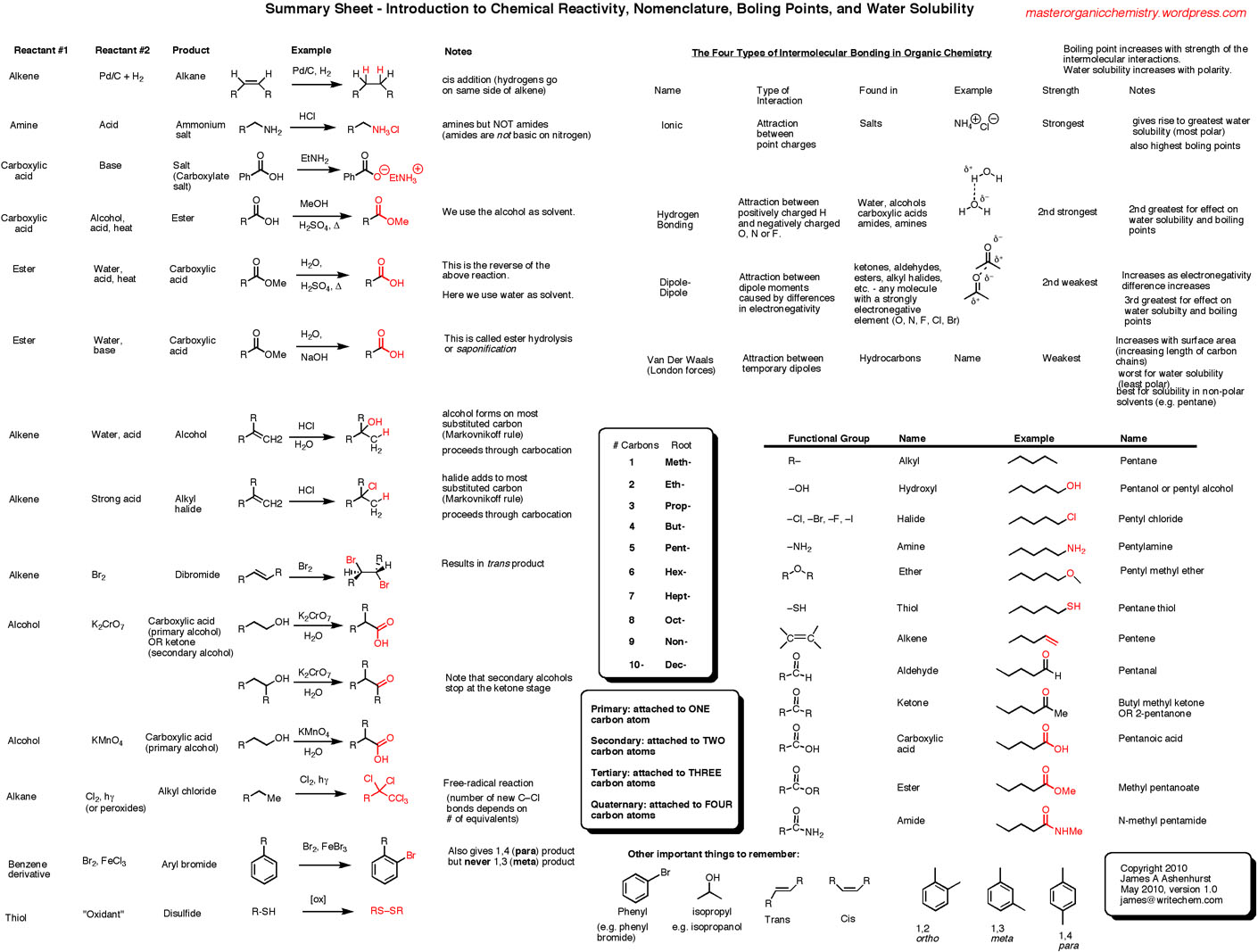
Let's break down what you might find in one of these packets and their answer keys. Usually, they start with the simplest types of organic molecules. We’re talking about alkanes, which are basically chains of carbon atoms bonded together with hydrogen atoms. Think of them as the plain spaghetti of the molecular world – simple and fundamental.
The naming system for these is pretty neat. The number of carbon atoms in the longest continuous chain determines the "root" of the name. So, one carbon? That's "meth-." Two carbons? "Eth-." Three? "Prop-." Four? "But-." After that, it gets a bit more predictable: "pent- " for five, "hex- " for six, and so on. You probably recognize some of these prefixes from things you’ve heard before, right? Like methane (one carbon) is a common gas, and butane (four carbons) is what’s in lighter fluid.
Then comes the "–ane" ending. That tells you it's an alkane, meaning all the bonds between the carbon atoms are single bonds. So, a chain of five carbons with only single bonds? That's a pentane. Easy peasy, right? The answer key would simply show you that "C5H12" (the chemical formula) translates to "pentane" if it’s a straight chain.
But what happens when things get a little more interesting? Molecules aren't always just straight lines of carbon. They can have branches, like a tree with twigs sticking off the main trunk. This is where the "2-methylpentane" example comes in. The answer key would show you how to identify the longest chain (that's the "pentane" part) and then how to name and number the branches. The "methyl" part tells you there's a one-carbon branch, and the "2-" tells you where that branch is attached to the main chain.
It’s like giving directions to a house. Instead of just saying "a house," you say "the house on Elm Street, number 12." The name tells you the main road (pentane) and the specific location of the side street (methyl group on the second carbon). The answer key is like the map that shows you exactly how to draw it out and label it correctly.
Then, we move on to other types of organic molecules. We've got alkenes, which have at least one double bond between carbon atoms. The "–ene" ending tells you this. Think of a double bond as a slightly tighter, more reactive connection. Then there are alkynes, with triple bonds (–yne ending), which are even more reactive. These are like the more energetic, sometimes more volatile cousins of the alkanes.
And it doesn't stop there! We have molecules with functional groups attached. These are special clusters of atoms that give molecules unique properties. Think of an alcohol, which has an "-OH" group attached. That's where the "ethanol" name comes from – "eth-" for two carbons and "-ol" for the alcohol group. This little "-OH" group is what makes ethanol behave so differently from a simple alkane with the same number of carbons.
An answer key for these types of problems would guide you through identifying the main chain, pinpointing the functional group, and then putting it all together in the correct IUPAC format. It’s a step-by-step process, and seeing the answers makes it so much clearer. You can see how a seemingly complex name is built from simple, logical parts. It’s like solving a crossword puzzle where each letter has a specific meaning.
What’s really cool about learning organic nomenclature is that it’s not just about memorizing names. It’s about developing a visual understanding of molecular structures. When you see a name like "3-ethyl-2,4-dimethylhexane," your brain starts to paint a picture of that molecule. You can visualize the six-carbon chain, the ethyl group on the third carbon, and the two methyl groups on the second and fourth carbons. This ability to mentally construct molecules is a huge part of becoming proficient in chemistry.

The answer key serves as your trusty guide on this visualization journey. It shows you the correct way to translate between the name and the structure, and vice versa. It’s a powerful tool for reinforcing what you’re learning. You can test your understanding by trying to draw the structure from the name, then check the answer key to see if you got it right.
Think of it this way: learning nomenclature is like learning to read a map. Once you understand the symbols and the grid system, you can navigate anywhere. In chemistry, the IUPAC naming system is that map, and the answer key is the legendary atlas that helps you find your way. It’s not about knowing every single city, but about understanding the system to find them all.
And honestly, it’s kind of satisfying to be able to look at a chemical name and know exactly what that molecule looks like, or to be able to draw a molecule and give it its proper, scientific name. It feels like you've cracked a code, like you've unlocked a secret level in the game of chemistry. So, if you ever stumble upon a "Basic Organic Nomenclature Packet Answer Key," don't be intimidated! Think of it as your friendly guide to the amazing world of molecular naming. It’s a key to understanding the building blocks of everything around us, and that’s pretty darn cool, don't you think?