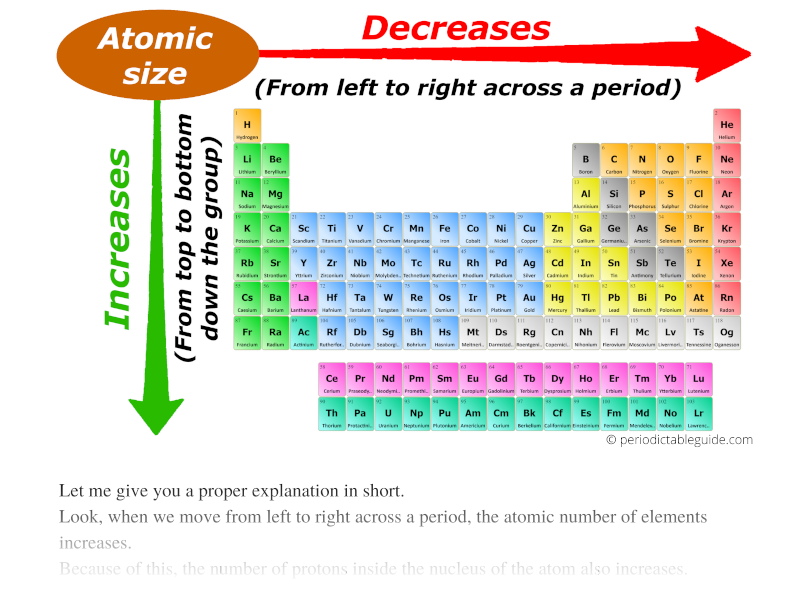
Atomic Radius Generally Increases As We Move
Imagine you're at a giant, cosmic party, and all the guests are tiny, invisible specks called atoms. Now, some of these atoms are a little shy, others are boisterous, but they all have something in common: a size! And just like at a real party, there are certain patterns to how these atoms hang out and how their sizes change.
We're going to talk about a really cool, and frankly, a little bit mind-bending, trend in the world of these tiny party-goers. It's like discovering a secret handshake or a hidden dance move that all the atoms seem to follow. This trend has to do with how big or small our atomic friends are, and it all starts with where they are positioned in this grand atomic ballroom. Think of it like a neighborhood map, but for atoms. We have rows, kind of like streets, and columns, like different districts within the city.
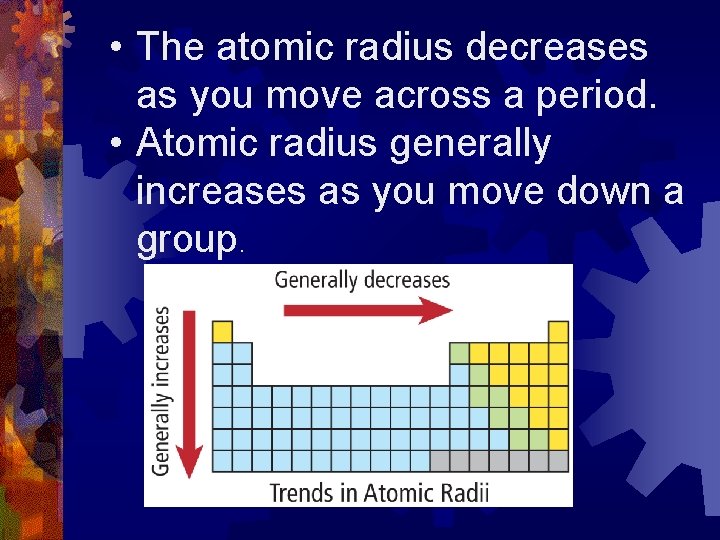
Now, here's where it gets interesting. As we take a stroll across this atomic map, moving from left to right, something rather predictable and, in its own way, quite delightful happens. The atomic radius, which is basically how much space an atom takes up, generally starts to get a bit… well, smaller. It’s like the atoms on the left side of the street are a bit more spread out, enjoying their personal space, while the ones further down the road are tucking themselves in a little tighter.
Must Read
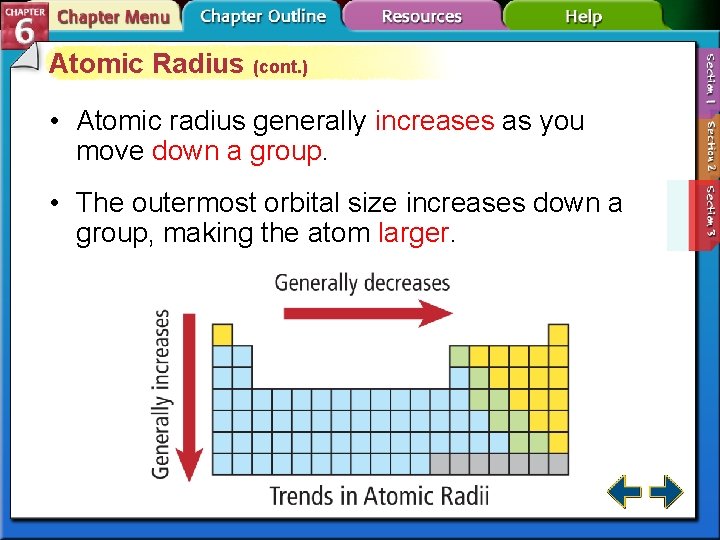
But the real magic, the heartwarming twist in this atomic tale, comes when we change our direction. If we decide to walk down a column, or as scientists call it, a group, the story takes a different turn. Imagine moving from the penthouse suite to the basement apartment. As we descend these atomic avenues, from the top of the column to the bottom, the atomic radius tends to increase. The atoms down below are bigger, more spacious! It’s like they’ve got more room to stretch out and be themselves.
This might sound a bit like a boring science fact, but let’s try to see it with a bit more flair. Think of a family. In the early generations, the ancestors might have been a bit more compact, perhaps living in a smaller home. But as the family tree branches out and grows, with new generations and bigger houses, there’s more space for everyone. The atoms are a bit like that, just on a much, much smaller scale. The earlier “generations” of atoms (those higher up in a group) are more tightly packed, while the later “generations” (those further down) have more “room” to expand.
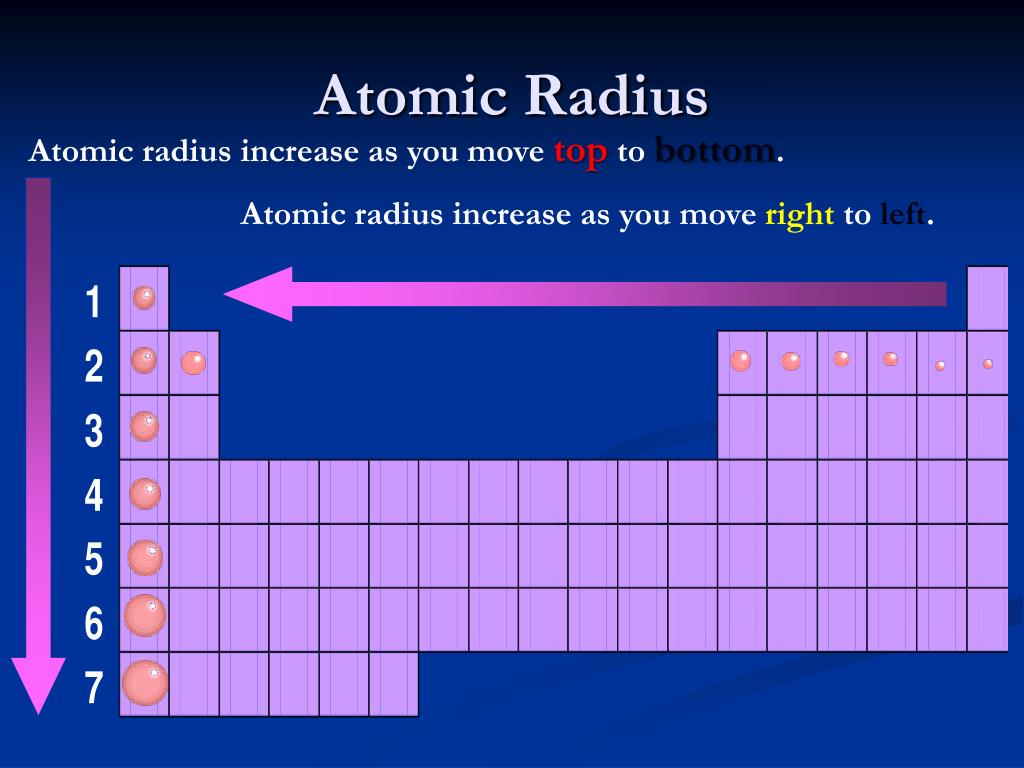
It’s also a bit like a game of musical chairs, but with electrons. These tiny, zippy things called electrons orbit the center of the atom, the nucleus. As we move down a column, each new row of atoms gets an extra layer of these electron shells. It's like adding more floors to a building. Each new floor pushes the outer walls of the building further out, making the whole structure larger. So, these larger atoms are not being greedy; they're just accommodating more of their electron residents!
The funny thing is, we can't even see these changes with our eyes. It's like knowing that your favorite song is getting a little louder as you walk towards the speakers, even if you can't consciously feel the sound waves getting stronger. It’s a subtle, yet powerful, shift in the universe of the very, very small.
All Periodic Trends in Periodic Table (Explained with Image)
And what about moving from left to right across a row, or a period? That’s where the nucleus, that central hub of the atom, starts to get a bit more “magnetic” in its pull on those electrons. It's like a really popular celebrity at a party, drawing everyone closer. Even though the number of electron shells stays the same as we move across, the increased pull from the nucleus causes the electrons to huddle a bit tighter. So, the atoms shrink!
This little atomic trend isn't just for scientists to marvel at. It helps us understand so much about how different elements behave. It’s the reason why some elements are happy to share their electrons, and others are a bit more possessive. It influences how atoms bond together to form the molecules that make up everything from the air we breathe to the chocolate we love to eat. It's the quiet architect behind so many of the wonders around us.
So, next time you're looking at something made of matter, whether it's a sparkling gem or a sturdy oak tree, take a moment to appreciate the intricate dance of the atoms within. Remember their sizes, how they stretch and contract, how they move across their cosmic ballroom. It’s a reminder that even in the tiniest, most invisible parts of our universe, there's a rhythm, a pattern, and a story waiting to be discovered. And who knows, maybe if you listen very, very closely, you can almost hear the atoms humming their own little tune as their radii change.
It's a beautiful, ordered chaos, this world of atoms. And the way their sizes change as they move around the periodic table? Well, that's just one more charming detail in their never-ending cosmic waltz. It's a testament to the elegance and predictability that underlies even the most fundamental building blocks of our reality. So, let's celebrate these expanding and contracting atomic sizes, a silent, yet profound, story unfolding all around us.