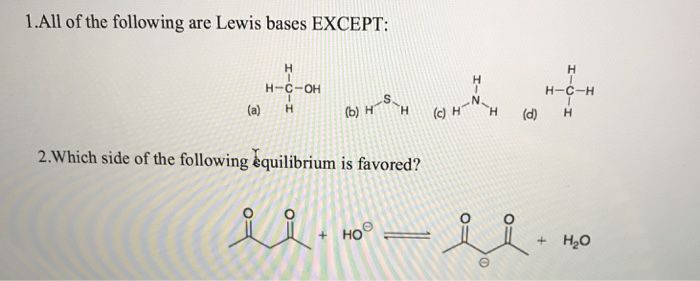
All Of The Following Are Lewis Bases Except:
Ever wondered what makes some things sticky and others slippery? Or how your cleaning supplies magically make grease disappear? A lot of that fascinating chemistry boils down to something called Lewis acids and bases. Now, that might sound a bit intimidating, but trust me, it's a really cool concept that pops up in unexpected places. Understanding it can be like unlocking a little secret to how the world around us works, and it's actually quite fun to see it in action.
So, why should you care about Lewis bases? For beginners in chemistry, it's a fundamental building block that helps explain many reactions. For families, you can use it to explain simple kitchen chemistry – like why adding baking soda to vinegar causes a fizz! Hobbyists, especially those into things like making soap or even brewing, will find this concept incredibly useful. It’s all about understanding how different molecules interact, and that knowledge can lead to better results in your projects.
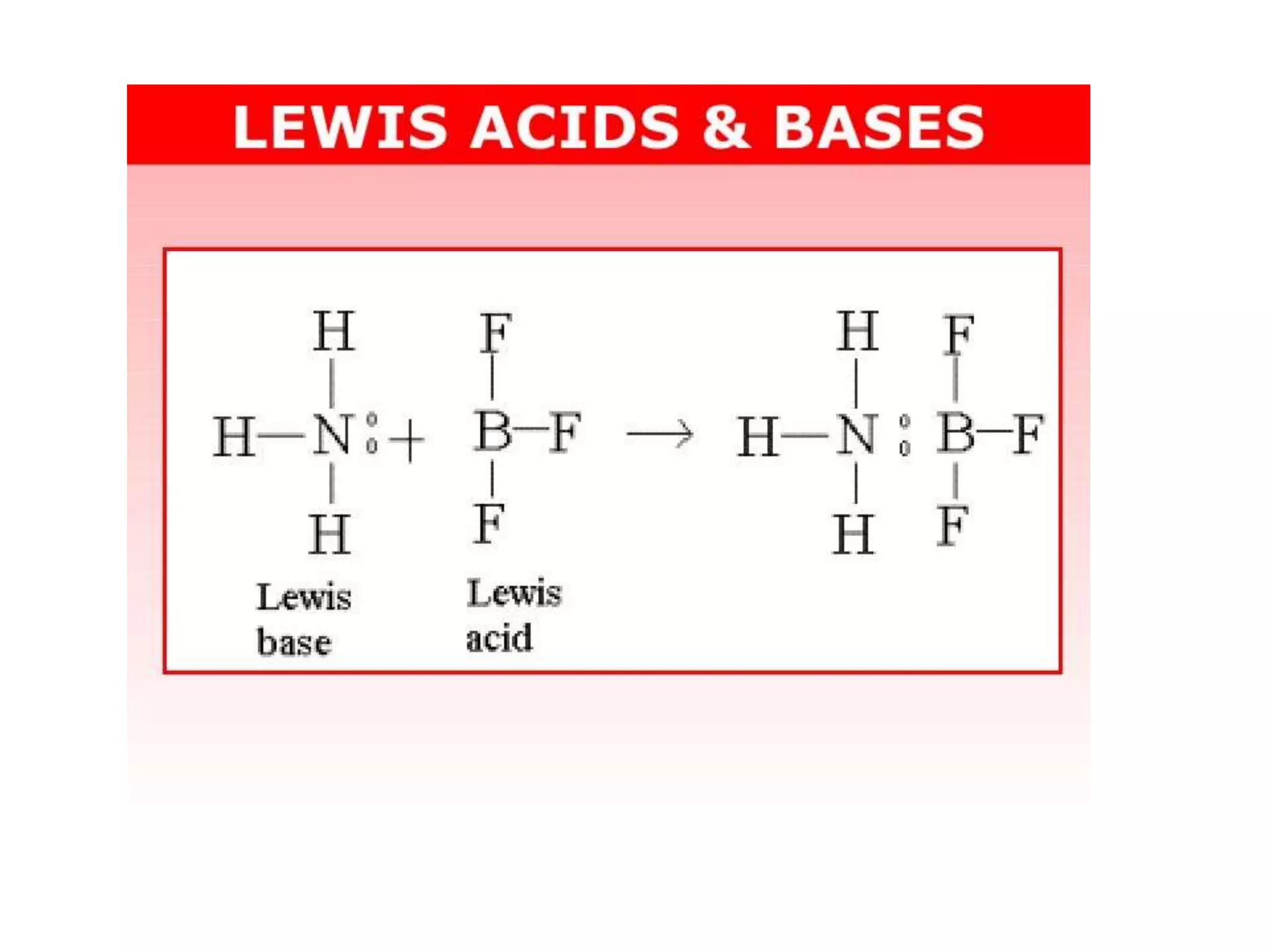
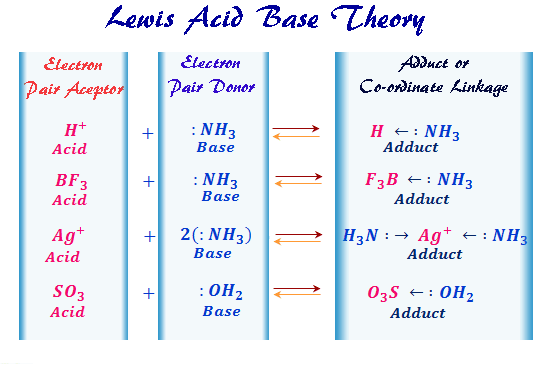
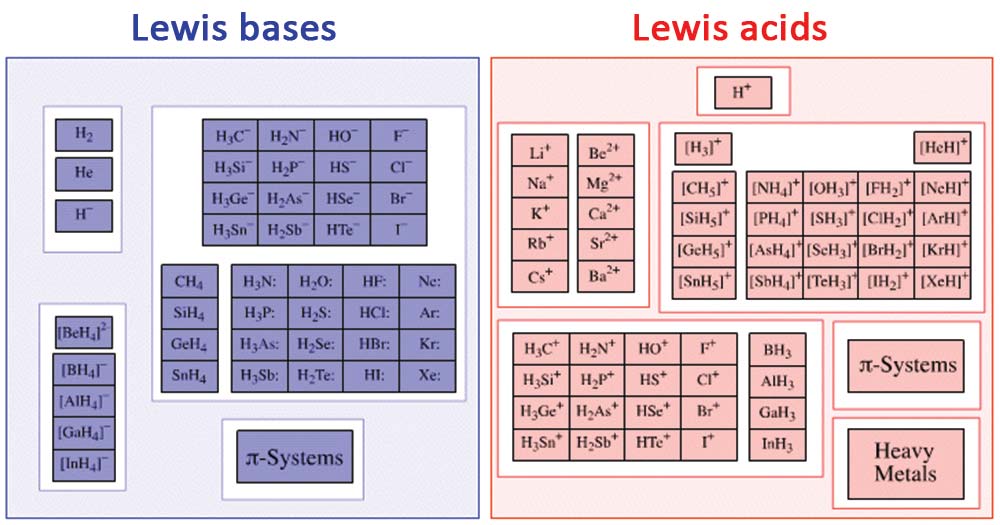
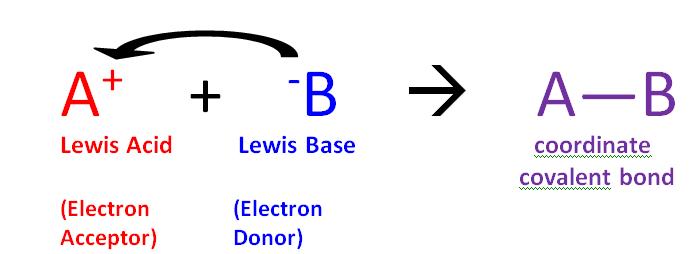
The core idea behind a Lewis base is pretty straightforward: it's a molecule or ion that has a pair of electrons it can donate. Think of it like offering a helping hand. A classic example is ammonia (NH₃). It has a lone pair of electrons on the nitrogen atom, just waiting to be shared. Other common Lewis bases include water (H₂O), which can donate electrons from its oxygen atom, and hydroxide ions (OH⁻), which are also electron-rich and ready to give. Even some simple anions, like chloride (Cl⁻), can act as Lewis bases because they carry extra electrons.
Must Read
Now, the question often comes up: "All of the following are Lewis bases except..." This is where we need to identify the odd one out. A Lewis acid, on the other hand, is a molecule or ion that can accept an electron pair. So, if you see something like boron trifluoride (BF₃) or aluminum chloride (AlCl₃) in a list, those are likely the Lewis acids, not bases. They have a "hungry" spot for those donated electrons. The key is to spot which molecule has those extra, available electrons to give away!

Getting started with understanding Lewis bases is easier than you think. You don't need a fancy lab. Start by looking at the chemical formulas of common substances around you. Try to identify atoms like nitrogen, oxygen, or anions with negative charges – these are often good indicators of potential Lewis bases. You can even search online for lists of common Lewis acids and bases to compare. Seeing the electron structures can really help visualize that "electron pair donation."
Ultimately, exploring Lewis acids and bases is a fantastic way to demystify chemistry. It’s not just about memorizing terms; it’s about grasping a fundamental principle that explains a vast array of chemical interactions. The next time you see a reaction, whether it's in a science book or your own kitchen, you'll have a better appreciation for the dance of electrons that’s making it all happen. It's a small piece of knowledge that can make the world of chemistry feel a lot more accessible and, dare I say, enjoyable!