2 Propanol Condensed Structural Formula 91

Hey there, science buddies and curious minds! Ever found yourself staring at a chemical formula and thinking, "What in the world is that supposed to be?" Yeah, me too. Especially when we start talking about things like "2 Propanol Condensed Structural Formula 91." Sounds a bit like a secret agent code, doesn't it? Like, "Agent 2-Propanol, your mission, should you choose to accept it, involves a cozy molecular embrace!" But don't worry, we're going to break this down together, nice and easy, like unwrapping a present. No fancy lab coats required, just your awesome brain and a willingness to have a little fun.
So, let's dive right into the mystery of 2-Propanol and its super-duper condensed structural formula. Think of it like this: sometimes in chemistry, we get a little wordy. We can draw out every single atom and bond, making a drawing that looks like a crazy spider web. But chemists, bless their efficient hearts, like to make things shorter, sweeter, and less prone to getting tangled. That's where "condensed structural formulas" come to the rescue!
Imagine you're texting your friend about a party. You wouldn't write out "I am going to attend the social gathering at my friend John's residence this evening." No way! You'd probably text something like, "Party @ John's tonite!" It's shorter, gets the point across, and is way easier to type on a tiny phone screen. Condensed structural formulas are the chemistry equivalent of that super-efficient text.
Must Read
Now, let's get to the star of our show: 2-Propanol. What is this critter? Well, the "ol" at the end is a big clue. It means we're dealing with an alcohol. Yep, like the kind you find in hand sanitizer (though probably not the exact same purity, and definitely don't go chugging this stuff – seriously, don't!). Alcohols have a special group of atoms attached to them called a hydroxyl group. It's basically an oxygen atom chilling with a hydrogen atom, and they love to hang out with carbon chains. Think of them as the life of the molecular party, always adding a little something extra.
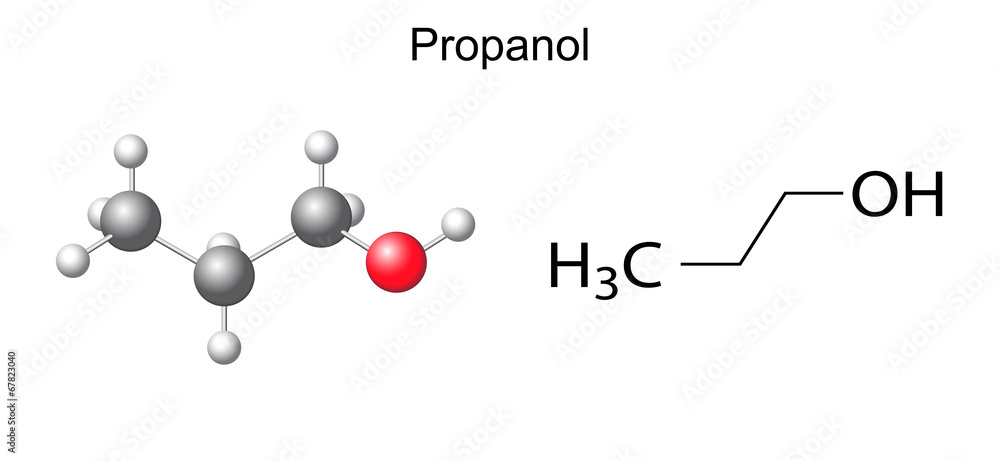
The "propan-" part of 2-Propanol tells us about the carbon backbone. "Prop" usually means there are three carbon atoms in a row. So, we've got a little chain of three carbons. Now, where does the "2-" come in? Ah, this is where the numbering starts! In organic chemistry, we number our carbon chains to tell us exactly where things are attached. The "2-" means that our special hydroxyl group (-OH) is attached to the second carbon atom in that chain of three.
So, picture this: three carbon atoms lined up like little soldiers. And on the middle soldier (that's carbon number 2, of course!), we've got our hydroxyl group, waving hello. The other two carbon atoms are just happily bonded to each other and to some hydrogen atoms. It’s a perfectly respectable, albeit small, molecule.
Now, let's talk about the "91" part. This is where things get a little tricky, or at least a little context-dependent. In most standard chemistry contexts, when you see "2-Propanol," that's it. There isn't a "91" hanging off it in the name itself. The "91" you might be referring to could be related to something else entirely, perhaps a specific batch number, a grade of purity, a catalog number from a chemical supplier, or even a typo! It’s like saying "my friend John 91" – what does the 91 mean? Is it his house number? His age? His IQ? We need more info!

However, let's imagine, for the sake of fun and exploration, that this "91" is somehow related to the structure or a specific form of 2-Propanol. In some very niche or specialized databases, numbers might be appended to indicate specific isotopes, or perhaps a particular crystalline form. But for the general, everyday understanding of 2-Propanol, that "91" isn't usually part of its official chemical name or formula. So, for the rest of our chat, we'll focus on the awesome 2-Propanol and its condensed structural formula, leaving the mysterious "91" to its own adventures. Perhaps it’s off saving the world from a rogue molecule of pure evil!
Okay, back to our main squeeze, 2-Propanol. We've established it's a three-carbon chain with an -OH group on the second carbon. Now, how do we write this down without drawing all those little lines and circles? This is where the magic of the condensed structural formula comes in. It’s like a shorthand that shows you the atoms and how they're connected, but without being overly detailed.
Let's break down a typical condensed structural formula for 2-Propanol. Remember our three carbons? We'll write them out. And we know the -OH group is on the middle one. So, imagine the molecule like this:
The Carbon Chain Foundation
We have a carbon atom. What’s it attached to? In 2-Propanol, the first carbon atom is bonded to three hydrogen atoms. So, we can write that part as CH₃. Simple, right? It's like saying, "This carbon is hugging three hydrogens."

Then, we move to the next carbon, the middle one, our VIP (Very Important Propanol). This carbon is a bit more crowded. It's bonded to the first carbon (the CH₃ group), it's bonded to the third carbon, and it's bonded to that crucial hydroxyl group (-OH). So, what does it look like in terms of hydrogens? Well, it’s only bonded to one hydrogen atom directly, because the other two spots are taken by the carbons and the oxygen. So, this middle part can be represented as CH.
And finally, we have the third carbon atom. This one is at the other end of our chain. Just like the first carbon, it's bonded to only one other carbon atom (the middle one), so it has plenty of room to bond with hydrogen atoms. In fact, it's bonded to three hydrogen atoms. So, this end group is also written as CH₃.
Adding the Hydroxyl Group
Now, where does our hydroxyl group (-OH) fit in? Remember, it's attached to that middle carbon (the CH). So, we need to show that connection. In condensed structural formulas, we often put the groups attached to a central atom in parentheses.
So, if we take our three parts: CH₃ (the first carbon), CH (the middle carbon), and CH₃ (the third carbon), and we know the -OH is attached to the middle CH, we can combine them. The most common way to write the condensed structural formula for 2-Propanol is:

CH₃CH(OH)CH₃
Let's dissect that little beauty! * The first CH₃ is our first carbon, happily holding onto three hydrogens. * Then comes the central CH. This is our middle carbon, holding onto one hydrogen. * The (OH) immediately after the CH tells us that the hydroxyl group is directly attached to that specific CH (our middle carbon). The parentheses are a visual cue saying, "Hey, this group is hanging off this atom right here!" * And finally, the last CH₃ is our third carbon, also holding onto three hydrogens.
See? It’s like a mini-story of the molecule! It tells us which atoms are connected and in what order. It's much quicker to write than drawing out every single bond, and it still gives us a clear picture of the molecule's architecture.
Why is this important, you ask? Well, knowing the structure of a molecule tells us a lot about its properties. For example, the presence of that -OH group makes 2-Propanol a polar molecule, which means it can dissolve in water (unlike, say, plain old oil). This is why it's used as a solvent in many applications, from cleaning up messes to being an ingredient in some medicines and cosmetics. It’s like the friendly molecule that can bridge the gap between water-loving and oil-loving substances.

Sometimes, you might see slightly different ways of writing it, but they all mean the same thing. For instance, you might see it written as: * CH₃-CH(OH)-CH₃ (with dashes indicating bonds, though in condensed formulas, these are often omitted for brevity) * Or even something like (CH₃)₂CHOH. This version emphasizes that there are two CH₃ groups attached to the same central CH, which is also attached to the OH. It's like saying, "This central bit has two identical siblings and one wise elder (the OH)." It’s just another way to package the same information.
The key takeaway is that the condensed structural formula is a way to simplify the representation of a molecule while still retaining its essential structural information. It's a balance between drawing out every single atom and just giving a molecular formula like C₃H₈O, which doesn't tell you how the atoms are arranged at all. The condensed formula is that sweet spot!
So, the "91" remains a bit of an enigma in the standard chemical nomenclature. But the 2-Propanol part? That's our buddy, a simple, versatile alcohol with a clear and understandable condensed structural formula: CH₃CH(OH)CH₃. It’s a molecule that’s everywhere, quietly doing its job, from cleaning your hands to being a building block in more complex chemistry. It’s a testament to how even seemingly simple arrangements of atoms can have significant roles in the world around us.
And you know what? Understanding these little bits of chemical language, like the condensed structural formula for 2-Propanol, is pretty darn cool. It’s like unlocking a secret code that explains so much about the physical world. Every time you figure out one of these formulas, you're adding another piece to your knowledge puzzle. So, give yourself a pat on the back! You’re not just learning about chemicals; you’re learning to read the universe in a new way. And that, my friends, is a superpower worth celebrating!
So next time you see "2-Propanol," don't let it intimidate you. Just remember that friendly three-carbon chain with an -OH on the second guy, and its tidy condensed formula CH₃CH(OH)CH₃. You've got this! Keep exploring, keep learning, and keep that smile on your face. The world of chemistry is a fascinating place, and you're doing a brilliant job navigating it!